AP Syllabus focus: ‘Weak acids react with water but only a small fraction ionizes, so most acid molecules remain un-ionized and [H3O+] is much smaller than the initial acid concentration.’
Weak acids behave differently from strong acids because their reaction with water does not go to completion. Understanding the equilibrium “picture” helps you predict which species dominate and why measured acidity is limited.
What “weak acid” means in water
A weak acid transfers protons to water only to a limited extent, producing a mixture of reactants and products at equilibrium rather than fully converting to ions.
Weak acid: an acid that partially ionizes in water, establishing a dynamic equilibrium between mostly un-ionized acid molecules and relatively small amounts of ions.
This partial ionization is the core reason weak acids produce less hydronium than a strong acid at the same initial concentration.
The equilibrium reaction and the equilibrium picture
For a generic monoprotic weak acid, , placed in water, the key process is proton transfer to water.
Water acts as the base (proton acceptor), forming hydronium.
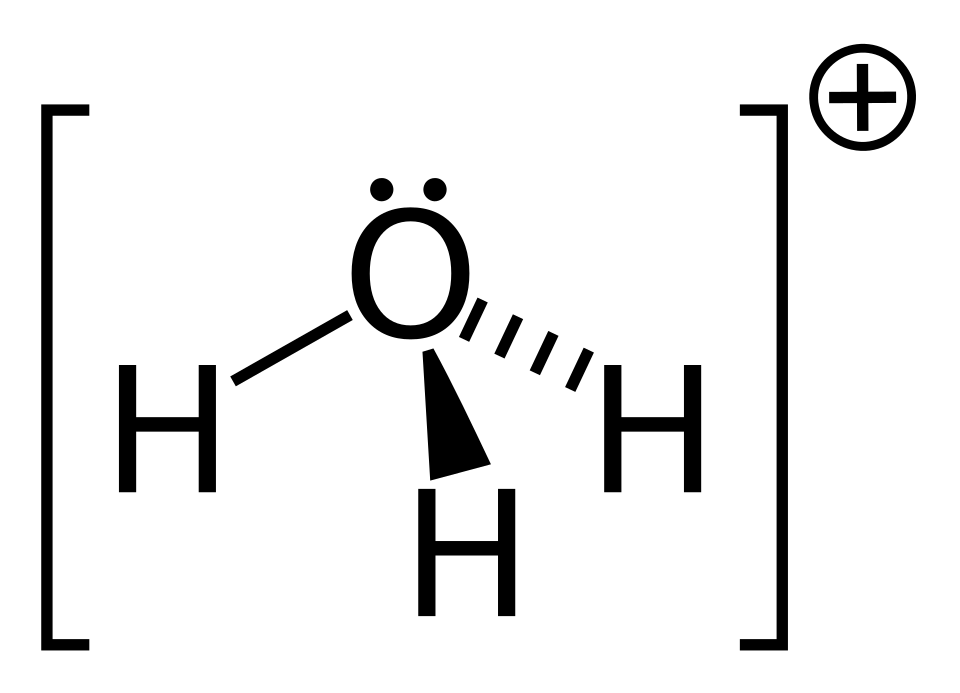
Structural formula of the hydronium (hydroxonium) ion, , showing oxygen bonded to three hydrogens with an overall positive charge. This emphasizes that the “acidic particle” in water is best represented as (a solvated/protonated water molecule) rather than a bare . Source
= un-ionized weak acid (aqueous)
= hydronium ion (mol L)
= conjugate base of the acid (mol L)
Because the arrow is reversible, both the forward reaction (ionization) and reverse reaction (recombination of with ) occur simultaneously.
What “only a small fraction ionizes” looks like
At equilibrium in a weak acid solution, the dominant species is typically , not ions.
3D van der Waals-style model of that helps connect the symbolic ion to a real molecular geometry (trigonal pyramidal around oxygen). Seeing the ion as a discrete species can make the “minor amount of ions” idea more concrete when comparing weak-acid solutions to strong-acid solutions. Source
The “equilibrium picture” is therefore:
Many neutral molecules dispersed in water
A comparatively small number of ions
A similarly small number of ions
An enormous background of molecules (the solvent)
A useful qualitative check is that weak acid solutions contain far fewer charge carriers than strong acid solutions of the same initial molarity, consistent with partial ionization.
How concentrations compare (qualitatively)
The syllabus emphasis is that the equilibrium hydronium concentration is much smaller than the initial acid concentration. In symbols, if the solution is prepared with an initial analytical concentration :
Most acid remains un-ionized: is close to
Product ions are minor species: is small compared with
Stoichiometric link between hydronium and conjugate base
Every successful forward ionization event produces one and one . Therefore, the increase in these product concentrations from ionization occurs in a 1:1 ratio.
= hydronium produced by the weak acid (mol L)
= equilibrium conjugate base concentration (mol L)
In real solutions, water itself contributes a tiny baseline amount of , but for the equilibrium picture of a typical weak acid solution, the key idea is that the ions formed from appear together and remain small compared with un-ionized .
Why equilibrium limits ionization
Ionization slows because products build up and drive the reverse reaction:
As increases, it more frequently collides with , reforming
As increases, it acts as a base toward , also reforming
Eventually, the system reaches dynamic equilibrium, where forward and reverse rates are equal (concentrations are constant, but molecules still react)
Common student pitfalls with the equilibrium picture
Assuming the acid “stops reacting” at equilibrium; instead, both directions continue at equal rates.
Treating weak acids like strong acids and setting ; for weak acids, this is not true because ionization is incomplete.
Forgetting that the major species is typically the molecular acid , not .
Interpreting “much smaller than the initial acid concentration”
The specification statement is much smaller than the initial acid concentration is a conceptual guide for setting expectations:
The solution is acidic because some forms.
The acidity is limited because most stays intact.
Any reasoning about pH trends should begin from the idea that weak acid solutions are mostly plus water, with minor amounts of and .
FAQ
Water is shown to make clear that it acts as a Brønsted–Lowry base, accepting a proton to form $H_3O^+$.
In dilute aqueous solutions, water is in vast excess, so its concentration changes negligibly even though it participates.
Individual molecules are continuously reacting in both directions.
At equilibrium, the rates of $HA$ ionising and $H_3O^+$ recombining with $A^-$ are equal, so the concentrations remain constant.
Electrical conductivity depends on mobile ions.
Weak acids generate relatively low concentrations of $H_3O^+$ and $A^-$, so there are fewer charge carriers compared with a strong acid, which produces many ions.
A particle diagram would show many neutral $HA$ particles and only a few $H_3O^+$ and $A^-$ ions.
The ions should appear in matched numbers (each ionisation event produces one of each).
If you include all sources of $H_3O^+$, pure water contributes a small background amount.
Also, side reactions (very concentrated solutions, non-ideal behaviour, or additional equilibria) can slightly disturb the simple 1:1 bookkeeping, though the basic stoichiometric link from $HA$ ionisation still applies.
Practice Questions
Question 1 (1–3 marks) Explain why a solution of a weak acid does not have .
States that a weak acid partially ionises in water / does not ionise completely. (1)
Mentions the establishment of an equilibrium between and ions (forward and reverse reaction). (1)
Concludes that most remains un-ionised, so is much smaller than the initial acid concentration. (1)
Question 2 (4–6 marks) A monoprotic weak acid is added to water to make a dilute aqueous solution.
(a) Write the equilibrium equation for the reaction of with water.
(b) Describe the relative amounts (major/minor species) of , , and present at equilibrium.
(c) State and justify the relationship between the amounts of and formed from ionisation of .
(a)
Correct reversible equation: . (1)
(b)
States is the major species (most remains un-ionised). (1)
States and are minor species present in relatively small amounts. (1)
Links to the idea that is much smaller than the initial . (1)
(c)
States that ionisation produces and in a 1:1 ratio (equal amounts formed). (1)
Justifies using stoichiometry of the equilibrium equation (one of each produced per ionisation event). (1)

