AP Syllabus focus: ‘A salt’s solubility is pH sensitive when a constituent ion is a weak acid, a weak base, or the hydroxide ion.’
Solubility is often treated as a fixed property at a given temperature, but many salts dissolve to form ions that also participate in acid–base reactions. In those cases, changing pH can change how much dissolves.
Core idea: when pH can change “solubility”
A salt’s solubility depends on pH when at least one ion produced by dissolution is acid–base active (it reacts with or ). This couples the solubility equilibrium to acid–base equilibria.
pH-sensitive solubility — solubility that changes when solution pH changes because at least one dissolved ion is a weak acid, a weak base, or , and therefore reacts with or .
A useful anchor is that solubility equilibria are written in terms of the concentrations of dissolved ions.
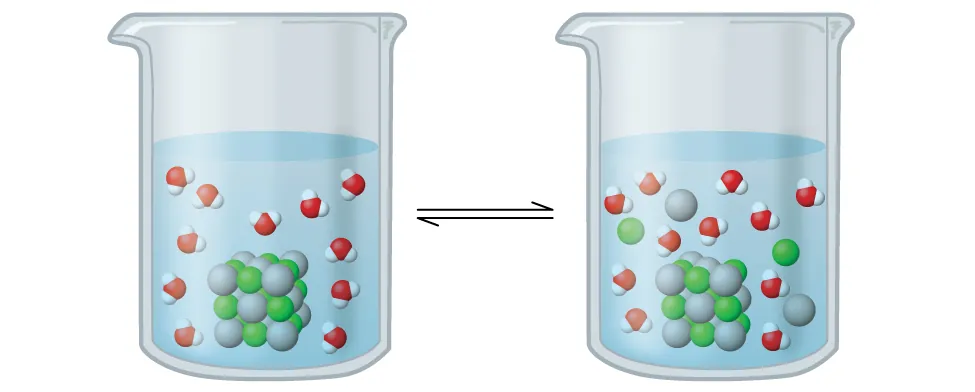
OpenStax schematic of the heterogeneous equilibrium for a sparingly soluble ionic solid (AgCl) dissolving to form aqueous ions. It emphasizes that dissolved ions exist in dynamic equilibrium with undissolved solid, which is the conceptual basis for writing and interpreting a expression. Source
= solubility product constant (unitless in AP-style treatments)
= equilibrium concentration of the cation, in
= equilibrium concentration of the anion, in
= coefficients from the balanced dissolution equation
If pH changes one of these bracketed concentrations through acid–base reaction, the dissolution position can shift, changing the amount that dissolves.
Case 1: the anion is a weak base (conjugate base of a weak acid)
Many slightly soluble salts contain anions that can be protonated by . These anions are weak bases (they are the conjugate bases of weak acids). Typical examples include , , , , , and .
Recognition rule
If the anion comes from a weak acid (not one of the spectator anions from strong acids like , , , , ), then the salt’s solubility is commonly pH sensitive.
Why pH matters (qualitatively)
Lower pH (more ) tends to remove by protonating it to , , etc.
Because is reduced by reaction, more solid can dissolve to re-establish the solubility equilibrium.
LibreTexts graph of CaF solubility versus pH showing a sharp increase in solubility at low pH. The figure connects the solubility change to fluoride speciation (HF vs. ), demonstrating how protonation of a weak-base anion reduces and drives additional dissolution to satisfy . Source
Case 2: the cation is a weak acid (conjugate acid of a weak base)
Some salts release cations that can donate protons to water (they are weak acids). This includes conjugate acids of weak bases such as (from ) and many small, highly charged metal aquo ions (often treated as acidic due to polarising O–H bonds in coordinated water).
Recognition rule
If the cation is the conjugate acid of a weak base (e.g., ), or a metal ion known to produce acidic solutions in water, then solubility can be pH sensitive.
Why pH matters (qualitatively)
Higher pH (more ) can react with acidic cations, reducing the free cation concentration (for example, by deprotonation of an acidic aquo complex).
Changing the free cation concentration can shift the dissolution equilibrium and therefore the solubility.
Case 3: hydroxide-containing salts (the ion is )
Metal hydroxides, , are classic pH-dependent solids because is explicitly in the solubility equilibrium.
Recognition rule
Any slightly soluble hydroxide (and many oxides that form hydroxides in water) will have solubility that depends strongly on pH.
Why pH matters (qualitatively)
Added acid consumes to form water, decreasing .
Because appears directly in , changing forces a shift in how much solid must dissolve to satisfy the equilibrium expression.
Practical checklist: decide quickly if pH dependence is likely
Use this identification workflow when you see a salt and are asked whether pH affects its solubility.
Write the ions produced by dissolution.
Ask: is any ion a weak acid or weak base?
Anion is a weak base if it is the conjugate base of a weak acid (common polyatomic bases, ).
Cation is a weak acid if it is the conjugate acid of a weak base (notably ) or an acidic metal aquo ion.
Ask: is present as an ion from the solid?
If “yes” to any, the solubility is pH sensitive; if the ions are all spectators from strong acids/bases (e.g., with ), solubility is much less likely to depend on pH.
FAQ
Amphoteric hydroxides can dissolve in both acidic and basic conditions.
In acid, $H_3O^+$ removes $OH^-$ groups; in strong base, complex ions may form (e.g., aluminate species), reducing free metal ion concentration and increasing dissolution.
Ionic strength mainly affects activities rather than the “yes/no” classification.
It can change apparent solubility slightly even without pH sensitivity, but pH sensitivity specifically requires coupling to acid–base reactions of weak acid/base ions or $OH^-$.
These anions are conjugate bases of strong acids, so they are negligibly basic in water.
Because they do not significantly react with $H_3O^+$, changing pH does not remove them through protonation in a way that strongly couples to dissolution.
Look for anions that commonly appear in weak acids:
Carboxylates (e.g., $CH_3COO^-$)
$F^-$ (from $HF$)
$CO_3^{2-}$, $SO_3^{2-}$, $PO_4^{3-}$, $S^{2-}$
If the corresponding acid is not among the standard strong acids, the anion is likely basic.
Complex-ion formation and redox side reactions can increase or decrease dissolved ion concentrations.
For example, ligand binding can lower free metal-ion concentration, allowing more solid to dissolve even at constant pH; this is distinct from pH sensitivity but can occur simultaneously.
Practice Questions
Question 1 (2 marks) State whether the solubility of depends on pH. Justify your answer by identifying the relevant ion property.
1 mark: Yes, it is pH dependent.
1 mark: is the conjugate base of the weak acid (so it can be protonated), making solubility sensitive to pH.
Question 2 (5 marks) For each solid below, state whether its solubility is expected to be pH sensitive and give a brief reason based on whether a constituent ion is a weak acid, a weak base, or : (a) (b) (c)
1 mark: (a) Not expected to be pH sensitive; and do not act as weak acid/base ions in the AP sense (chloride is conjugate base of strong acid).
2 marks: (b) pH sensitive; contains in the dissolution equilibrium and can remove to form water (any clear acid-consumption statement earns the second mark).
2 marks: (c) pH sensitive; is a weak acid (conjugate acid of weak base ) and reacts with / water in acid–base equilibria.

