AP Syllabus focus: ‘A buffer with more conjugate acid has greater capacity for added base, while a buffer with more conjugate base has greater capacity for added acid.’
Buffers resist pH change by converting added strong acid or base into a weaker conjugate partner. The direction of this protection depends on which buffer component is present in greater amount.
Core idea: “excess” determines what you can neutralise more of
A buffer contains a conjugate acid–base pair (such as HA/A− or HB+/B). When small amounts of strong acid or strong base are added, the buffer consumes them through essentially complete acid–base reactions.
Key terminology
Buffer capacity (directional) is the relative ability of a buffer to neutralise added strong base versus added strong acid, determined by which conjugate partner has more initial moles available to react.
A buffer does not have one single “capacity”; it has an acid-neutralising capacity (ability to consume added ) and a base-neutralising capacity (ability to consume added ). These are usually not equal unless the conjugate partners are present in equal amounts.
What reactions control capacity direction?
For an acid buffer containing HA (conjugate acid) and A− (conjugate base), the dominant neutralisation reactions are:
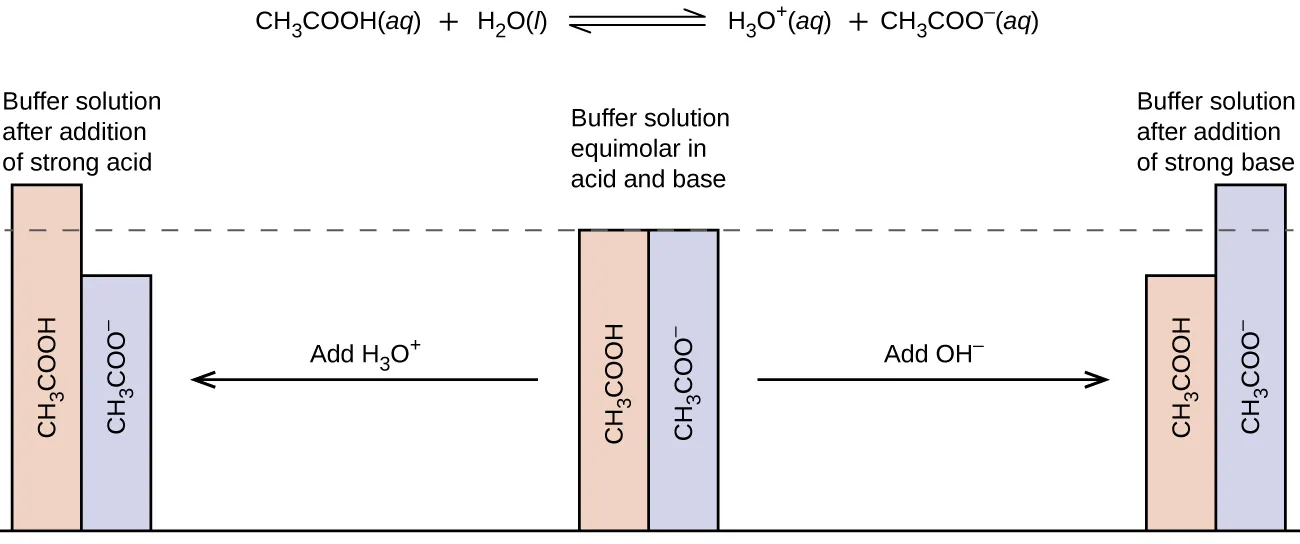
Bar charts compare the relative amounts of weak acid and conjugate base in a buffer before and after adding strong acid or strong base. The figure makes the stoichiometry visible: added converts HA to , while added converts to HA. This supports the “more initial moles = greater capacity in that direction” rule by showing which species is being consumed. Source
Added strong base is consumed by the conjugate acid:
HA + OH− → A− + H2O
Added strong acid is consumed by the conjugate base:
A− + H3O+ → HA + H2O
Because these reactions use up a specific buffer component, the component with more initial moles can react with (and therefore neutralise) more of the corresponding added strong reagent before the buffer begins to fail.
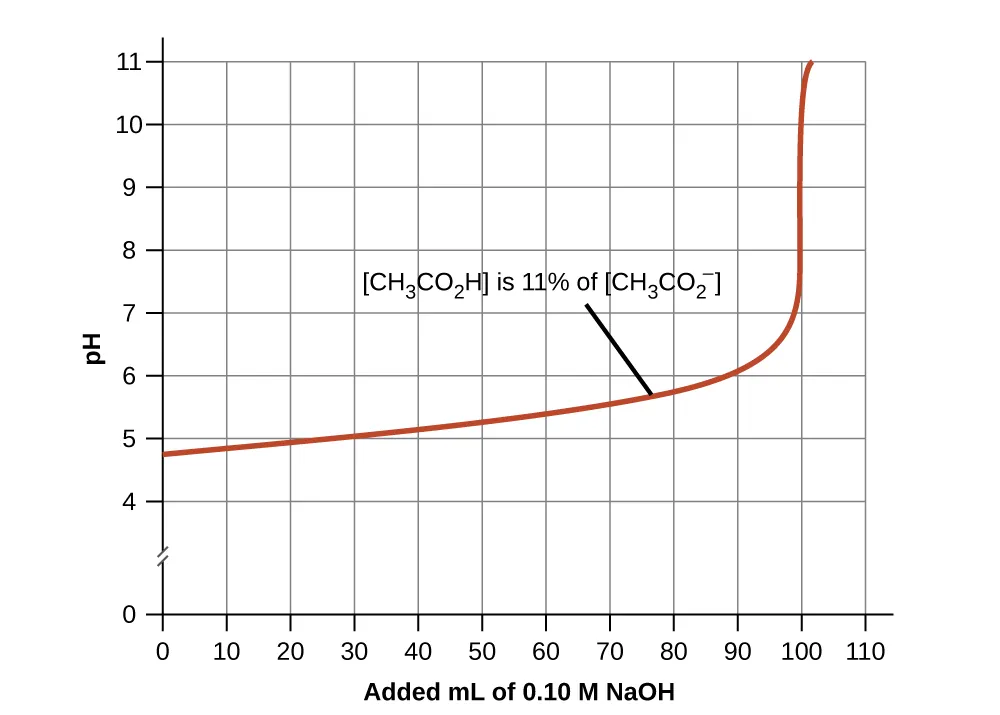
A pH-versus-added-base curve illustrates the buffered region (gradual slope) and the point where buffering capacity is exceeded (sharp pH increase). The gentle portion corresponds to substantial amounts of both conjugate partners still being present, while the steep jump reflects near-exhaustion of the component that neutralizes the added reagent. This connects buffer “capacity” to a limiting-reactant idea rather than to pH alone. Source
How to interpret “more conjugate acid” vs “more conjugate base”
If the buffer has more conjugate acid, HA
Greater capacity for added base (higher base-neutralising capacity)
Reason: there are more moles of HA available to react with incoming OH−, so the buffer can convert more OH− into water while forming A−.
Practical interpretation:
The buffer “handles base better” because HA is the reactant that removes OH−.
If the buffer has more conjugate base, A−
Greater capacity for added acid (higher acid-neutralising capacity)
Reason: there are more moles of A− available to react with incoming H3O+, so the buffer can convert more H3O+ into water while forming HA.
Practical interpretation:
The buffer “handles acid better” because A− is the reactant that removes H3O+.
Limiting-reactant view (why “excess” is the right concept)
Capacity direction is a stoichiometry question:
Each mole of HA can neutralise about 1 mole of OH− (for a monoprotic HA).
Each mole of A− can neutralise about 1 mole of H3O+.
So, whichever component starts with more moles is the one that will not run out first for its corresponding neutralisation reaction. The “weaker” side of the buffer (lower capacity direction) is controlled by the component present in smaller amount, because it becomes the limiting reactant sooner.
What counts as “more” in AP Chemistry context?
Use moles, not concentrations alone, when comparing buffer components:
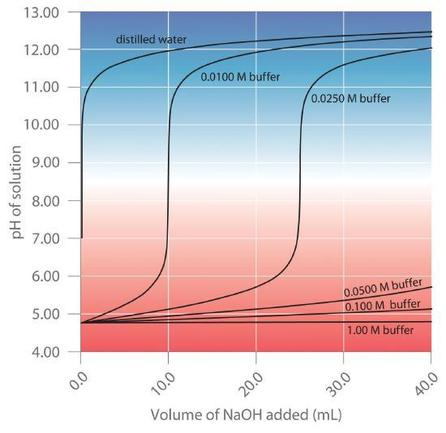
Overlaid curves show how the same addition causes a much smaller pH change when the buffer is more concentrated. The figure reinforces that buffer capacity increases with total amount of conjugate pair available to react, even when the acid:base ratio (and thus pH) may be similar. This is a graphical way to connect “capacity” to available moles in the mixture. Source
If volumes are the same, comparing concentrations is sufficient.
If solutions were mixed or diluted, compare initial moles of HA vs initial moles of A− present in the final mixture.
Extending the idea to basic buffers (HB+/B)
For a buffer made from a weak base B and its conjugate acid HB+:
HB+ neutralises added OH− (greater base-neutralising capacity when HB+ is in excess).
B neutralises added H3O+ (greater acid-neutralising capacity when B is in excess).
This is the same rule: the species that directly reacts with the added strong reagent controls capacity in that direction.
Common misconceptions to avoid
“A buffer with more HA has a lower pH, so it must resist acid better.” pH and capacity direction are different ideas; direction depends on which reactant is available to neutralise what is added.
“Equal concentrations always mean equal capacity.” Only true if the total volume context makes moles equal; otherwise, unequal moles give unequal directional capacities.
“The buffer stops working exactly when one component is gone.” In practice, buffering ability degrades progressively as the ratio becomes extreme, but the AP-level directional rule is based on which component is in excess.
FAQ
Compute moles of each component after mixing.
Then compare total moles of $HA$ and $A^-$ (or $HB^+$ and $B$), not their initial stock concentrations.
Yes; each acidic proton can contribute additional neutralisation steps.
Directional capacity depends on which relevant conjugate partners (e.g., $H_2A/HA^-$ vs $HA^-/A^{2-}$) are present in excess.
If one component is much smaller, the buffer will lose buffering action quickly in that direction.
This is crucial when you expect predominantly acid loads or predominantly base loads.
A rough upper bound comes from stoichiometry: available moles of the consuming species (e.g., moles of $A^-$ for added $H_3O^+$).
Equilibrium effects refine when “significant pH change” occurs.
They can slightly shift effective concentrations (activities), especially in very concentrated solutions.
However, the direction rule based on which component is in excess typically remains the dominant predictor at AP level.
Practice Questions
Question 1 (2 marks) A buffer is prepared with and . State whether it has greater capacity for added strong acid or added strong base, and explain briefly.
Identifies greater capacity for added strong base / base-neutralising capacity (1)
Explains that neutralises added and is in excess (more moles than ) (1)
Question 2 (5 marks) A student prepares two buffers of the same conjugate pair . Buffer X contains more than . Buffer Y contains more than . (a) For each buffer, state which added reagent is better neutralised: strong acid or strong base. (2 marks) (b) For Buffer X, write the net ionic equation showing consumption of added strong base. (1 mark) (c) For Buffer Y, write the net ionic equation showing consumption of added strong acid. (1 mark) (d) Using a limiting-reactant argument, explain why the less-abundant component determines which direction has the smaller capacity. (1 mark)
(a) X: better neutralises strong base; Y: better neutralises strong acid (2; 1 each)
(b) (1)
(c) (1)
(d) Explains that capacity in a direction is limited by the reactant consumed in that neutralisation reaction; fewer initial moles means it is limiting and is used up first (1)

