AP Syllabus focus: ‘Equilibrium constants can be determined from experimental measurements of reactant and product concentrations (Kc) or gas partial pressures (Kp) at equilibrium.’
Equilibrium data let you quantify how far a reversible reaction proceeds. This page focuses on extracting or from measured equilibrium compositions and properly translating experimental values into an equilibrium-constant expression.
What it means to “determine K” from data
To determine an equilibrium constant, you use measured equilibrium amounts (concentrations for solutions or partial pressures for gases) and substitute them into the correct law of mass action expression for the balanced reaction.
Equilibrium constant, : A ratio (built from equilibrium concentrations or partial pressures) that is constant at a fixed temperature for a given balanced reaction.
A key skill is matching the form of the data to the form of : solution measurements lead to , while gas partial-pressure measurements lead to .
Building the correct equilibrium expression
Start with a balanced chemical equation
You must use the reaction as written (including stoichiometric coefficients) because exponents in the expression come directly from coefficients.
Use equilibrium values only
When determining , every substituted value must correspond to the system at equilibrium (not initial values, not changes).
= Equilibrium molar concentration in (M)
= Equilibrium partial pressure (commonly in bar or atm)
Choose when equilibrium data are given as molarities (typical for aqueous systems) and when equilibrium data are given as partial pressures (typical for gas-phase systems).
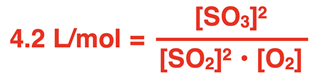
A labeled template for the gas-phase equilibrium constant , written as a product of partial pressures of products divided by those of reactants, each raised to their stoichiometric coefficients. This supports the “use the reaction as written” rule and makes the vs. data-type distinction concrete. Source
Check that the species match the data type
Use only for species with meaningful equilibrium concentrations in solution or gas concentration data.
Use only for gaseous species when equilibrium partial pressures are provided or can be obtained from the experiment.
Translating experimental measurements into equilibrium values
Equilibrium data are not always handed to you directly as the exact or needed for the expression; you may need to interpret what the experiment reports.
Common equilibrium measurements that directly give what you need
Equilibrium molarity from spectrophotometry or titration analysis (reported as )
Equilibrium partial pressures measured with pressure sensors or calculated from known gas composition and total pressure (reported as )
Measurements that require an intermediate step (conceptually)
If an experiment reports equilibrium moles in a container of known volume, those moles correspond to equilibrium and can be converted to concentrations conceptually by dividing by volume.
If an experiment reports total pressure plus equilibrium gas composition, each gas’s equilibrium partial pressure is conceptually tied to its fraction of the mixture and the total pressure.
When using such converted values, maintain consistent units throughout the calculation so the equilibrium expression is internally consistent.
How to ensure the value you calculate is truly K
Confirm equilibrium has been reached
Data must come from a state where macroscopic properties are constant over time (for example, stable color, stable pressure, or stable concentration readings).
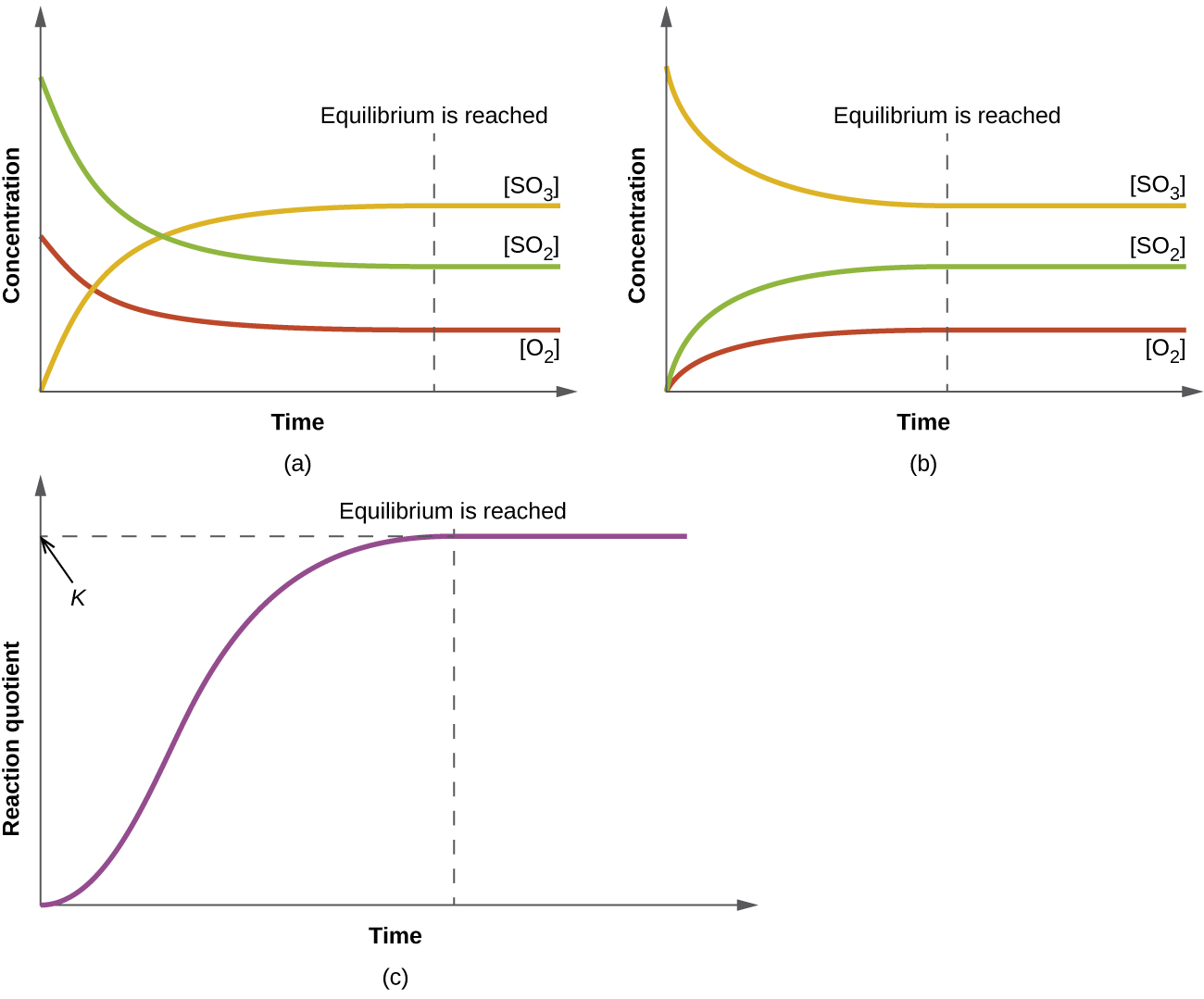
Time-course plots showing reactant/product concentrations leveling off as equilibrium is reached, along with a corresponding plot where the reaction quotient approaches a constant value at equilibrium (). This reinforces that plugging in non-equilibrium measurements yields , not the equilibrium constant. Source
If the system is still changing, substituting values would produce , not .
Temperature must be specified or controlled
Because is temperature-dependent, equilibrium data must correspond to a known temperature. If the temperature changes during measurement, the “constant” you compute may not represent a single value.
Use the correct reaction form
Determining is tied to the balanced equation provided. If you rewrite the reaction (for example, reverse it or scale coefficients), the numerical value associated with that new equation is a different constant for that new form, even though the underlying system is the same.
Report with appropriate significant figures
should reflect the precision of the equilibrium measurements. Since is computed from measured quantities raised to powers, rounding too early can noticeably change the final value.
FAQ
Often a single species is measured directly (e.g., by absorbance), then stoichiometry is used to infer the others.
Common approaches include:
Spectrophotometry for coloured species
Titration of one component followed by stoichiometric relationships
Small discrepancies come from measurement uncertainty and propagation through powers in the $K$ expression.
Typical contributors:
Sensor calibration limits (pressure, absorbance)
Volume or temperature drift
Rounding during intermediate steps
Use the constant that matches what is measured.
If the experiment provides equilibrium partial pressures, compute $K_p$ directly. If it provides equilibrium molar concentrations of gases, compute $K_c$ using those concentration values.
You must conceptually convert moles to the form required by the chosen equilibrium constant.
For $K_c$: use container volume to relate amount to concentration.
For $K_p$: relate gas composition to partial pressures using the reported total pressure and mixture information.
On AP-style reporting, $K$ is typically treated as unitless in practice, even though expressions built from $[X]$ or $P_X$ may suggest units.
A good convention is:
Report $K$ as a pure number
Use correct significant figures consistent with the equilibrium data
Practice Questions
(2 marks) For the reaction , write the expression for .
1 mark: Correct structure as products over reactants using partial pressures.
1 mark: Correct exponents: .
(5 marks) An experiment for reports equilibrium partial pressures at a fixed temperature: atm and atm. Determine for the reaction as written.
1 mark: Correct expression: .
1 mark: Substitution shown: .
1 mark: Correct evaluation setup (square applied correctly).
1 mark: Correct numerical value (allow appropriate rounding).
1 mark: States that pressures used are equilibrium values and temperature is fixed (validity condition).

