AP Syllabus focus: ‘Q and K omit solids and pure liquids because their concentrations are effectively constant. Be aware of whether Kc or Kp is used; converting between them is not assessed on the AP Exam.’
Equilibrium expressions are powerful only when written correctly. The most common errors come from including the wrong species, especially pure solids and pure liquids, or mixing up and .
What to omit from and
The core rule (heterogeneous equilibria)
In any equilibrium expression (both reaction quotient and equilibrium constant ), omit:
Pure solids (e.g., , )
Pure liquids (e.g., , )
Include species only when their amounts can change the “effective concentration” in a meaningful way, such as:
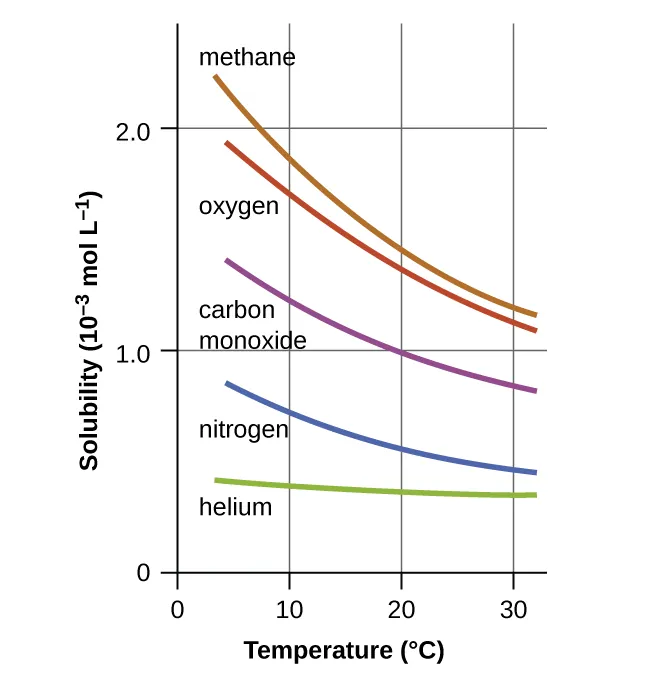
This graph shows that the solubility (concentration) of several gases in water changes substantially with temperature even at constant pressure. It visually reinforces why gaseous species are treated as composition-dependent terms in equilibrium expressions, unlike pure condensed phases whose activities are taken as approximately 1. Source
Aqueous solutes (use bracket concentration, e.g., )
Gases (use partial pressures for )
Why pure solids and pure liquids are omitted
Their “concentrations” do not meaningfully change as long as some of the phase is present:
A pure solid has essentially constant density, so its molar amount per unit volume of solid is constant.
A pure liquid similarly has nearly constant density (and thus constant molar concentration for the pure liquid).
These constant factors are mathematically absorbed into the constant itself, so the equilibrium expression is written only in terms of variable composition species (gases and dissolved solutes).
Activity viewpoint (what the rule is really saying)
Activity: An effective concentration used in equilibrium expressions; for a pure solid or pure liquid, activity is approximately 1 under typical conditions.
Because the activity of a pure solid or liquid is ~1, including it would multiply the expression by ~1 and change nothing, so it is omitted by convention.
How to write expressions without the omitted phases
Write or using only species whose measured amounts can vary in the mixture (typically aqueous and gaseous species), with exponents equal to stoichiometric coefficients.
= equilibrium molar concentration of an aqueous solute,
= partial pressure of a gaseous species (commonly in atm)
This same “include/omit” logic applies identically to and .
Common traps involving
Water is frequently present, so state symbols matter:
Omit when it is a pure liquid (even if it appears as a reactant or product in the balanced equation).
Include because it is a gas (its partial pressure can change).
Include only if it is being treated as a solute in a nonstandard way (rare in AP-level contexts; most often you won’t see water written as ).
A related idea: in reactions occurring in water, the solvent is not written into as a “reactant” because its effective concentration is treated as constant under the conditions assumed.
“Be aware of whether or is used” (AP exam note)
Always match the equilibrium constant form to the data provided:
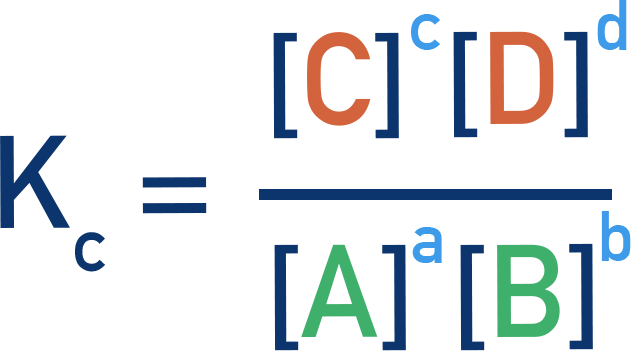
These two expressions highlight the structural parallel between and : both are products-over-reactants with exponents equal to stoichiometric coefficients. The key difference is the measurable quantity used—molar concentrations for versus partial pressures for . Source
If the problem gives molar concentrations or refers to species in solution, it is typically .
If the problem gives partial pressures for gases, it is .
Use consistent quantities within one expression (don’t mix and in the same or expression unless a problem explicitly defines a nonstandard approach).
Conversion note (what you are not required to do)
You should recognise that and are different forms, but converting between and is not assessed on the AP Exam. Your task is primarily to choose the correct form and write the expression correctly, omitting pure solids and pure liquids.
FAQ
They are in their standard state as a separate phase.
For a pure phase, the “effective concentration” does not change appreciably with the amount present, so equilibrium uses a reference value, giving activity $\approx 1$.
$K$ depends on temperature, not on how much solid is present.
Adding solid can change the rate of reaching equilibrium, but not the equilibrium ratio expressed by $K$, provided the solid phase remains present.
If water is not a pure liquid.
Examples include:
$\mathrm{H_2O(g)}$ in gas-phase equilibria (included via $P_{\mathrm{H_2O}}$)
Situations where water is a solute or its activity is not approximated as constant (beyond typical AP scope)
No—only that they are not variables in the expression.
Pure solids/liquids still participate and can limit how far the reaction proceeds (e.g., if one solid is completely consumed, the equilibrium situation changes).
Formally, $K$ is defined using activities, making it dimensionless.
In AP-style problems, you may see units implied by concentration-based forms, but you should treat $K$ as a number tied to a specific way of writing the expression and a specific temperature.
Practice Questions
(2 marks) For , write the correct equilibrium constant expression.
(1)
Omits solids and (1)
(5 marks) Consider in water.
(a) Write . (2 marks)
(b) A student includes in the denominator of . State whether this is correct and justify. (3 marks)
(a) (2: correct form (1), correct exponents/species (1))
(b) Not correct to include (1)
is a pure liquid with effectively constant concentration/activity (1)
Its factor is absorbed into so it is omitted by convention (1)

