AP Syllabus focus: ‘Very large equilibrium constants indicate reactions that proceed essentially to completion, leaving mostly products at equilibrium.’
A large equilibrium constant signals that, at equilibrium, the mixture contains predominantly products. Understanding what “product-favored” really means helps you interpret equilibrium data and predict the qualitative composition of reacting systems.
What “large K” means at equilibrium
Equilibrium constant as a product-to-reactant measure
Equilibrium constant (K): A dimensionless number that describes the equilibrium ratio of products to reactants, each raised to their stoichiometric coefficients, for a specific reaction at a specific temperature.
A large K means the equilibrium “ratio” strongly favors products. In other words, the equilibrium state is reached when product amounts are much larger than reactant amounts (relative to the reaction’s stoichiometry).
= equilibrium constant (dimensionless)
= equilibrium concentrations (typically mol L)
= stoichiometric coefficients (unitless)
“Essentially to completion” is an equilibrium idea, not an absolute
When the syllabus states that reactions with very large K proceed “essentially to completion,” it means:
The equilibrium position lies far to the products side.
Only a small fraction of reactant remains at equilibrium (not necessarily zero).
The system still reaches equilibrium (it does not stop reacting); it reaches a state where additional net change is negligible on a macroscopic scale.
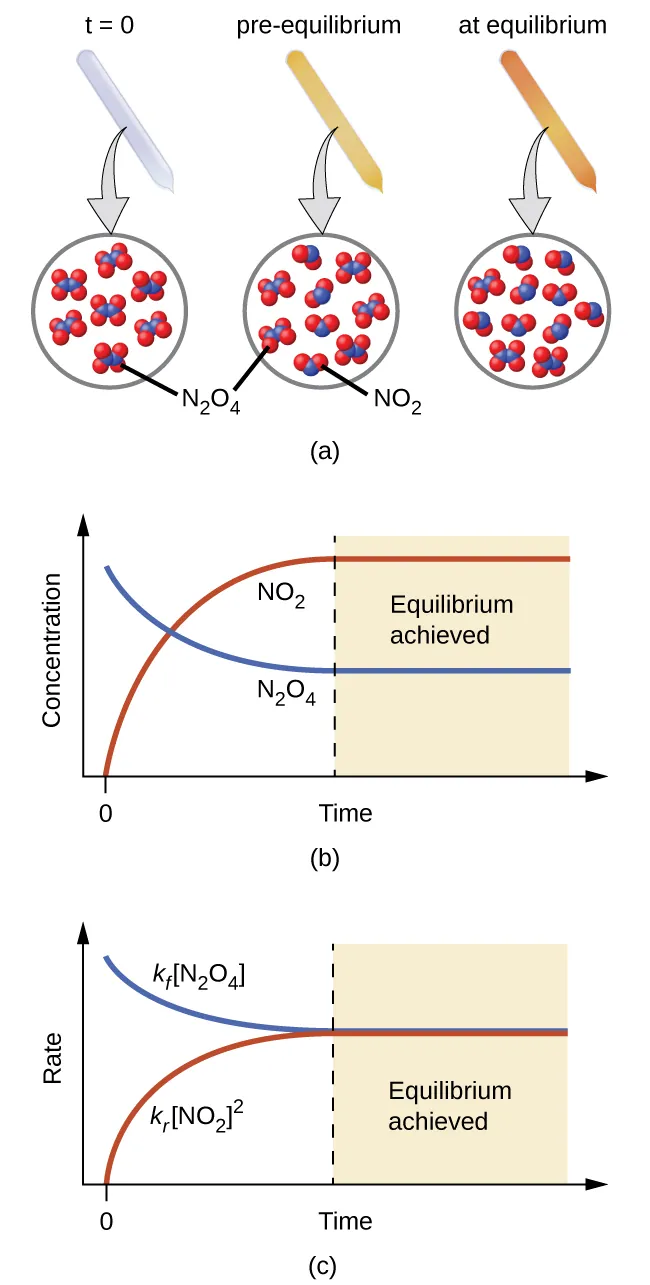
OpenStax figure illustrating dynamic equilibrium for : concentrations change over time and then plateau, while forward and reverse rates become equal. This clarifies that “essentially to completion” refers to the equilibrium position (final composition), not to reactions literally halting. Source
Connecting large K to equilibrium composition
Product-dominant mixtures
For a reaction with K much greater than 1:
The numerator of the K expression (products) must be large compared with the denominator (reactants).
At equilibrium, measured concentrations/partial pressures typically show:
High product amounts
Low reactant amounts
This interpretation is qualitative but powerful: you can look at a K value and anticipate whether equilibrium “looks like products.”
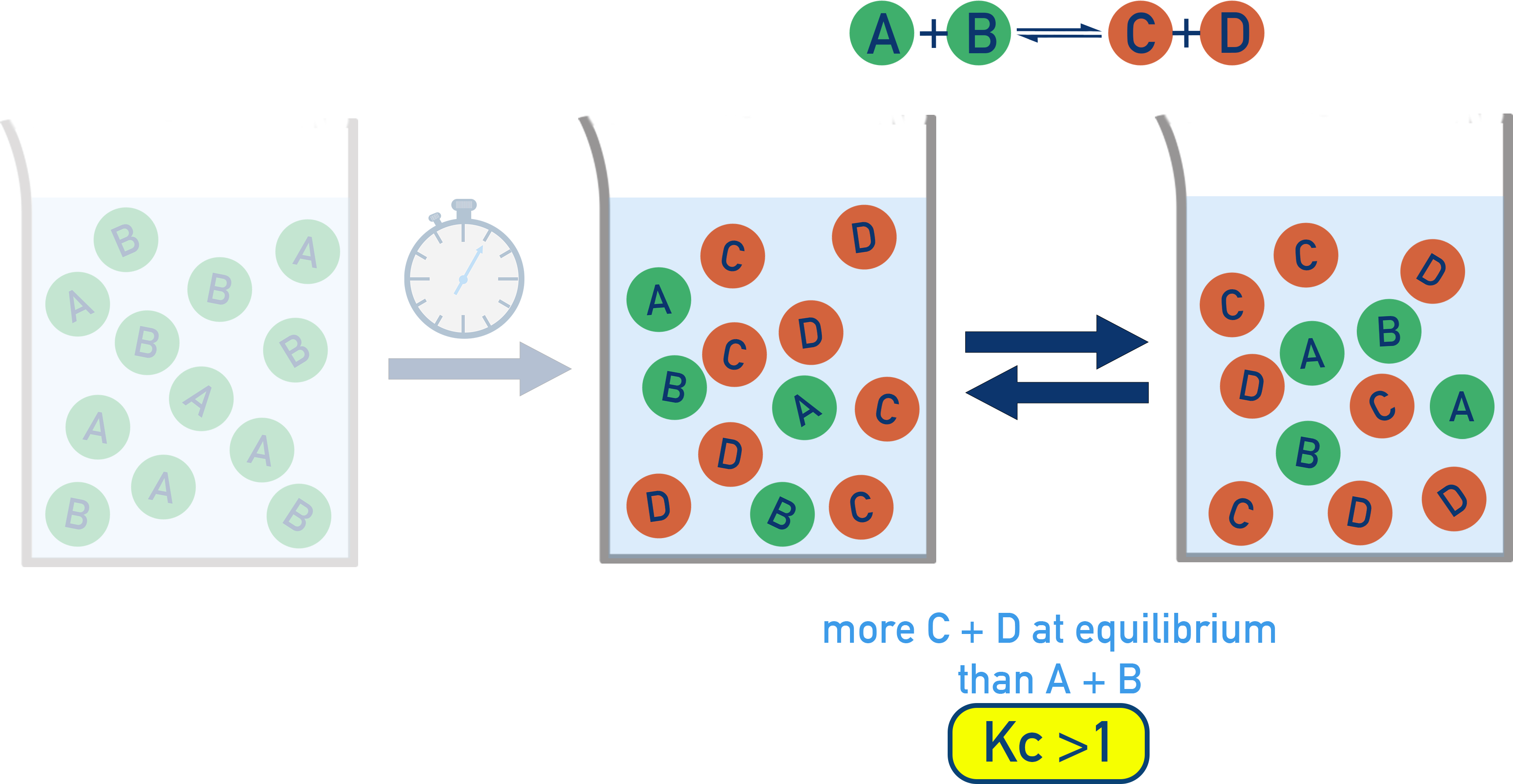
Particulate model for a reversible reaction showing the system evolving from mostly reactants to an equilibrium mixture with more products. The image connects the macroscopic idea “product-favored” directly to a particle-count representation, consistent with the meaning of . Source
Stoichiometry matters when interpreting “mostly products”
“Mostly products” should be interpreted in the context of coefficients in the balanced equation:
Because each species is raised to a power, small changes in certain concentrations can strongly affect K.
A reaction can have a large K even if one product concentration is not huge, provided reactant concentrations are extremely small (again, relative to the exponent structure).
What large K does and does not tell you
What it tells you
The equilibrium position is product-favored.
The equilibrium mixture is dominated by products under typical conditions that allow equilibrium to be established.
What it does not tell you
How fast equilibrium is reached (rate depends on kinetics, not K).
The exact equilibrium composition without additional information (such as initial amounts and the reaction expression).
Whether “complete reaction” occurs in a practical sense for every setup; physical constraints and initial conditions can limit how much product can form before a reactant is exhausted.
Temperature specificity
A K value (including a “very large” one) applies only at the temperature for which it is reported. Interpreting product-favored equilibria is always temperature-conditional.
FAQ
There is no single cutoff. In practice, values such as $10^{3}$ to $10^{6}$ or higher often indicate product-dominant equilibria, but the meaning depends on required purity and the stoichiometry of the K expression.
Yes. “Noticeable” depends on scale and detection limits. A tiny equilibrium fraction can still correspond to measurable moles in a large-volume system, even though the ratio implied by $K$ strongly favours products.
A rigorous treatment uses activities (effective concentrations relative to a standard state), which are dimensionless. Introductory problems often use molar concentrations as approximations, especially in dilute solutions.
Not necessarily. Side reactions, incomplete mixing, competing equilibria, separation losses, and practical constraints can reduce isolated yield even if the target equilibrium is strongly product-favoured.
Because exponents amplify effects. For example, if a reactant has a large coefficient, a modest decrease in its concentration can produce a large increase in the products-to-reactants ratio required by a large $K$.
Practice Questions
(2 marks) For a reaction at a fixed temperature, . State what this implies about the equilibrium mixture.
Recognises that is very large / much greater than 1 (1)
States equilibrium strongly favours products / mostly products present with very little reactant remaining (1)
(5 marks) Consider with a very large at a given temperature. Explain, using the meaning of , why the reaction is described as “essentially to completion,” and give two valid limitations of that description.
Links large to a much larger products-to-reactants ratio at equilibrium (1)
States that at equilibrium products predominate and reactants are present in very small amounts (1)
Explains “essentially to completion” as an equilibrium position statement, not requiring zero reactant (1)
Limitation: gives no information about reaction rate / time to reach equilibrium (1)
Limitation: is temperature-specific OR exact composition depends on starting amounts/constraints (1)

