AP Syllabus focus: ‘Dissolution of a salt is a reversible process; its extent is described by the solubility-product constant, Ksp, for the dissolution reaction.’
Many ionic solids do not simply “dissolve and disappear.” For slightly soluble salts, dissolution and precipitation occur simultaneously, creating a dynamic equilibrium that can be quantified using the solubility-product constant, Ksp.
Dissolution as a reversible (equilibrium) process
When an ionic solid is placed in water, ions can separate from the crystal and become solvated (hydrated). At the same time, dissolved ions can collide and re-form the solid lattice. For many salts, especially sparingly soluble ones, these two opposing processes establish an equilibrium.
What “saturated” means at the particle level
Saturated solution: a solution in which the dissolved ions are in dynamic equilibrium with undissolved solid, so the dissolved-ion concentrations remain constant at a given temperature.
In a saturated solution:
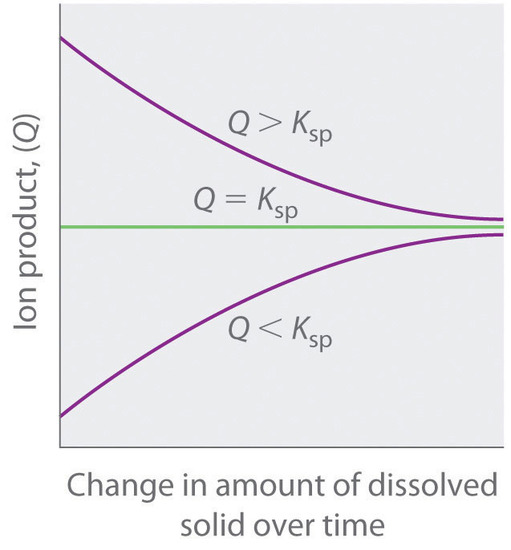
This graph summarizes how the ion product () compares to and predicts the direction of change: net dissolution when , no net change at (saturation), and precipitation when . It provides a compact visual way to connect “dynamic equilibrium” with the equilibrium-condition test used in precipitation problems. Source
Both undissolved solid and aqueous ions are present.
The rate of dissolution equals the rate of precipitation.
The ion concentrations are constant over time (though particles are still moving and reacting).
Writing the dissolution equilibrium (Ksp reaction)
A dissolution equilibrium is written as a reversible reaction between a pure solid and its aqueous ions. For a salt with formula :
The solid appears as a reactant: it is the source of ions.
The aqueous ions appear as products: they are formed by dissolution.
The equilibrium constant for this specific dissolution reaction is called the solubility-product constant, Ksp.
Solubility-product constant (Ksp): the equilibrium constant for the dissolution of a slightly soluble ionic solid, written in terms of the equilibrium concentrations of its ions.
The Ksp expression (what it includes and omits)
The law of mass action is applied to the dissolution reaction to write Ksp. Importantly, pure solids are omitted from equilibrium expressions because their effective concentration (activity) is constant as long as some solid is present.
This omission is not “ignoring” the solid; it reflects that changing the amount of pure solid does not directly change the equilibrium ratio of ion concentrations.
= solubility-product constant for the dissolution reaction (at a specific temperature)
= equilibrium molar concentration of the cation in solution,
= equilibrium molar concentration of the anion in solution,
= stoichiometric coefficients from the balanced dissolution equation
Because the exponents come from stoichiometric coefficients, Ksp depends on how the dissolution reaction is written. Each solid has its own Ksp for its own dissolution equilibrium.
Interpreting Ksp conceptually
Ksp describes the extent of dissolution at equilibrium:
A system with a small Ksp has an equilibrium that lies mostly toward undissolved solid (very few ions in solution).
A system with a larger Ksp (for the same type of stoichiometry) corresponds to a greater extent of ion formation at equilibrium.
Ksp is an equilibrium constant, so it is a temperature-dependent quantity. If temperature changes, the equilibrium constant for dissolution changes, meaning the equilibrium ion concentrations for a saturated solution must also readjust to satisfy the new Ksp.
Key conditions for using Ksp correctly
To use Ksp as intended, keep these conditions in mind:
Equilibrium must be established (typically a saturated solution with undissolved solid present).
Concentrations used in Ksp are equilibrium ion concentrations in solution, not initial values.
The Ksp expression contains only aqueous species (no solid term).
Ksp applies to the dissolution reaction as written; if the reaction is reversed (precipitation), the equilibrium constant would be the reciprocal.
FAQ
In equilibrium thermodynamics, pure solids have (approximately) constant activity under the conditions of the equilibrium.
This constant factor is absorbed into the value of $K_{sp}$, so including a variable “concentration of solid” would be meaningless unless the solid’s composition changes.
In a rigorous (activity-based) treatment, equilibrium constants are dimensionless.
In introductory chemistry, $K_{sp}$ is often reported using concentrations, so it may appear to have derived units (e.g., $(\text{mol L}^{-1})^{a+b}$). This depends on the stoichiometry and convention used.
A saturated solution is prepared at a controlled temperature and allowed to reach equilibrium.
The equilibrium ion concentration(s) are then determined, commonly by:
gravimetric methods (mass of precipitate formed from a known volume)
titration of one ion
instrumental methods (e.g., ion-selective electrode)
The $K_{sp}$ expression’s powers depend on stoichiometry, so the same numerical value can correspond to different equilibrium ion concentrations.
Meaningful comparisons typically require relating $K_{sp}$ to equilibrium ion concentrations under the relevant dissolution stoichiometry.
In non-dilute solutions, ions interact, so concentration is not an exact measure of “effective concentration.”
Ionic strength affects activity coefficients, shifting the relationship between measured concentrations and the thermodynamic equilibrium constant. This is why $K_{sp}$ values are most reliable under defined conditions (often near-dilute solutions).
Practice Questions
(2 marks) Write the expression for for the dissolution of calcium fluoride:
(1)
Correct exponent of 2 on and no term for (1)
(5 marks) A beaker contains a saturated solution of at a fixed temperature with some solid present. (a) State what it means, in terms of particle processes, for this system to be at equilibrium. (2 marks) (b) Predict what happens to and after each change, assuming the temperature is unchanged: (i) some solid is removed but solution remains in contact with solid, (ii) additional solid is added. (3 marks)
Forward dissolution and reverse precipitation both occur (1)
Rates are equal so there is no net change in ion concentrations (1) (b)
(i) No change in and provided some solid remains to maintain saturation (1)
(ii) No change in and ; adding more pure solid does not change expression or equilibrium concentrations (1)
Clear statement that the solid is omitted from the equilibrium expression because it is a pure solid with constant activity/concentration (1)

