AP Syllabus focus: ‘Changes in concentration affect Q only, while a change in temperature changes K. In both cases, the system shifts until Q and K are equal again.’
Chemical equilibrium problems often hinge on separating what changes the reaction quotient (Q) from what changes the equilibrium constant (K). This distinction lets you predict the direction of shift after a stress without doing full calculations.
Q vs K: what each represents
Reaction quotient (Q): A ratio of product to reactant amounts (using current concentrations or partial pressures) that describes the system’s position at any moment.
Q is calculated from the instantaneous mixture, so it can change immediately when you change what’s in the container.
Equilibrium constant (K): The value of Q at equilibrium at a specific temperature; it is fixed for a given reaction only at that temperature.
Because K is tied to temperature, most “disturbances” do not change K, even though they can force the system to shift.
The shared algebra of Q and K (why comparing them works)
For a general reaction, Q and K have the same mathematical form; only the input values differ (current vs equilibrium).
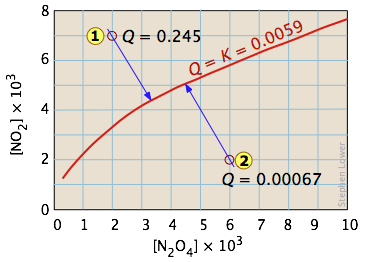
Graphical representation of equilibrium for , where points on the curve correspond to compositions satisfying . Points off the curve represent non-equilibrium mixtures, and the arrows indicate how the system’s composition changes as it approaches the equilibrium condition. This makes the statement “equilibrium means ” visually literal. Source
= molar concentration (mol L)
= stoichiometric coefficients from the balanced equation
If the system is at equilibrium, .
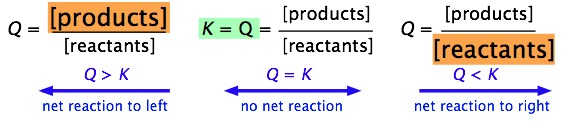
Schematic summary showing how the sign of predicts the net reaction direction. When , the mixture is reactant-heavy relative to equilibrium and the forward reaction proceeds; when , the mixture is product-heavy and the reverse reaction proceeds. This visual reinforces the idea that the system shifts until returns to (at constant temperature). Source
After a stress, Q typically changes first; then the reaction shifts until Q returns to equal K (or until K changes, if temperature changed).
Stresses that change Q (but not K)
Changing concentration (or partial pressure)
Adding reactant/product increases its term in Q; removing decreases its term.
This alters Q immediately, while K stays the same (temperature unchanged).
The system then shifts until Q becomes equal to K again:
If Q is made smaller than K, the net reaction proceeds forward to raise Q.
If Q is made larger than K, the net reaction proceeds reverse to lower Q.
Changing volume/pressure for gas-phase equilibria
For reactions involving gases, changing container volume changes partial pressures, so it changes Q (because Q depends on current partial pressures).
K does not change if temperature is constant.
The shift direction is the one that restores after Q is disturbed (often described qualitatively as favouring the side with fewer/more moles of gas, but the Q/K logic is the core idea).
Dilution (for aqueous systems)
Adding solvent reduces solute concentrations, so Q changes immediately.
K remains unchanged (again, assuming constant temperature).
The system shifts until the concentration ratio returns to match K.
The one stress that changes K: temperature
Why temperature is different
A temperature change alters the underlying energetics of the equilibrium, so K itself changes.
After a temperature change:
The system may momentarily have the old equilibrium concentrations, but the new K applies.
The system shifts until Q equals the new K.
Directional idea (no memorisation-heavy details needed)
For an endothermic forward reaction (heat as a reactant), increasing temperature increases product-favouring tendency, so K increases.
For an exothermic forward reaction (heat as a product), increasing temperature decreases product-favouring tendency, so K decreases. Regardless of sign, the syllabus-critical point is: temperature changes K; concentration changes Q only; the system shifts until Q and K match again.
Practical checklist (what to say on AP-style explanations)
State whether the stress changes Q or K.
If it changes Q only: say K is constant and the system shifts until .
If it changes temperature: say K changes and the system shifts until .
FAQ
Neither. A catalyst changes reaction rates, helping the system reach equilibrium faster, but it does not change the equilibrium composition, so it does not change $Q$ (by itself) or $K$.
At constant volume, partial pressures of reacting gases do not change, so $Q$ is unchanged.
At constant pressure, volume increases and partial pressures can change, so $Q$ may change.
Because $K$ depends on temperature through thermodynamics. Changing $T$ changes the energetic balance between reactants and products, so the equilibrium ratio must change.
Because $Q$ is computed from current concentrations/partial pressures. Unless temperature changes, the equilibrium condition (encoded by $K$) is unchanged, so only the system’s position relative to equilibrium changes.
Treat it as two effects: temperature changes $K$; pressure/volume changes $Q$ (for gases). Then predict the shift by comparing the new $Q$ to the new $K$.
Practice Questions
A system is at equilibrium for at constant temperature. More is injected. Does this disturbance change , , or both? State what must happen to re-establish equilibrium.
changes (1)
does not change because temperature is constant (1)
The system shifts until again (forward shift as was added) (1)
A sealed equilibrium mixture for is at equilibrium. The temperature is increased. Explain, in terms of and , what changes immediately and what drives the shift to a new equilibrium.
Temperature change alters (1)
becomes a new value at the higher temperature (1)
Immediately after heating, concentrations/partial pressures have not yet re-equilibrated, so initially reflects the old mixture (1)
Therefore (1)
The reaction shifts in the direction that makes move toward (1)
A new equilibrium is reached when (1)

