AP Syllabus focus: ‘Solubility rules can be linked quantitatively to Ksp; in this framework, Ksp values greater than 1 correspond to salts that are classified as soluble.’
Solubility rules quickly predict whether an ionic compound dissolves, but they are qualitative. Using connects those rules to measurable equilibrium ion concentrations, giving a quantitative basis for “soluble” versus “insoluble.”
Solubility rules as equilibrium statements
Common “solubility rules” (e.g., “most nitrates are soluble”) are shorthand for a chemical equilibrium idea: ionic solids establish a balance between undissolved solid and dissolved ions. A salt that is labelled insoluble by rules still dissolves slightly; it simply produces very low equilibrium ion concentrations.
To connect rules to equilibrium, treat dissolution as a reversible reaction whose position is described by an equilibrium constant.
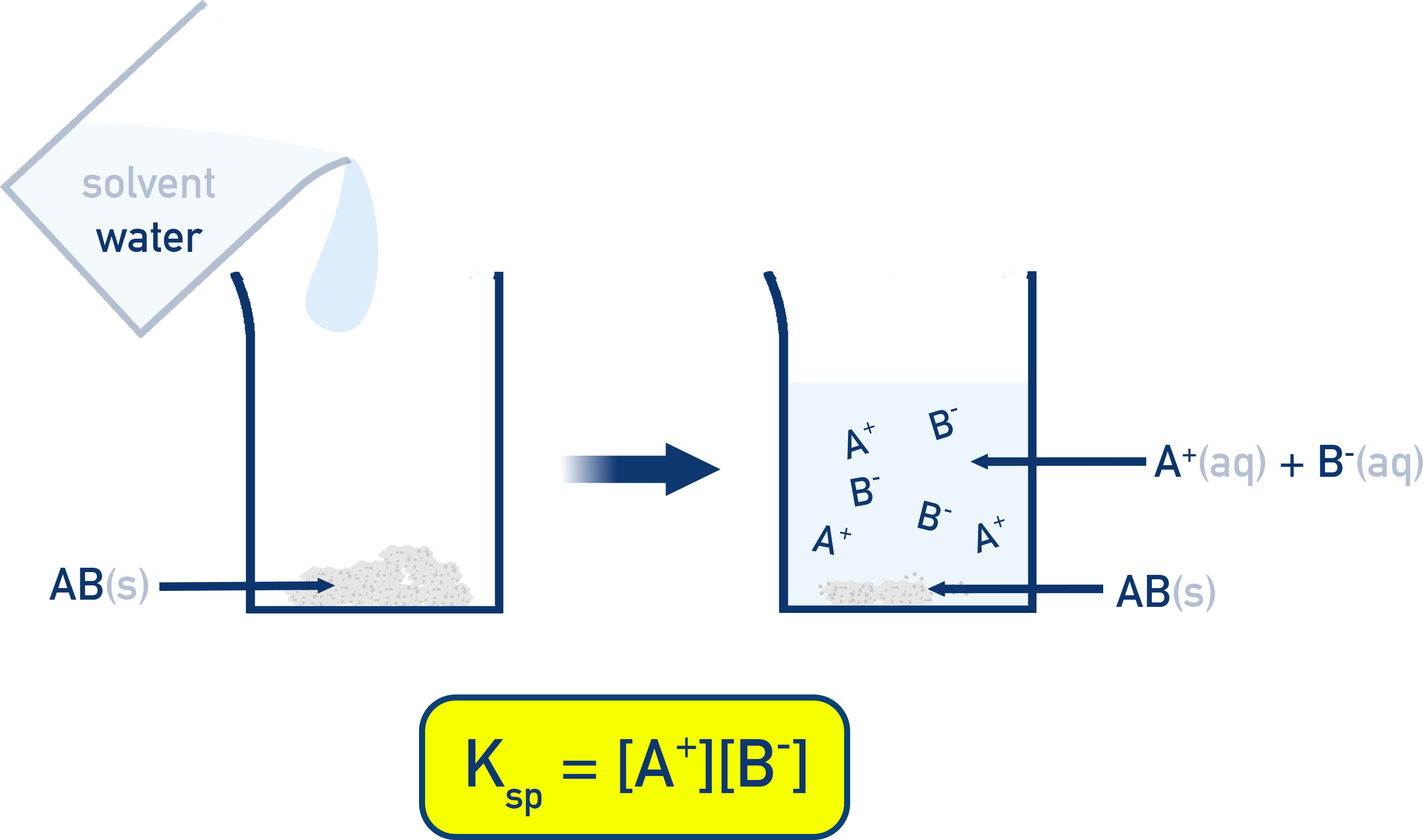
An ionic solid dissolves to form ions in water until the rates of dissolution and precipitation balance (dynamic equilibrium). At equilibrium, the solubility product is written from ion concentrations only, giving for a 1:1 salt. The solid phase is excluded from the expression because its activity is effectively constant while any undissolved solid remains. Source
The solubility-product constant,
Ksp (solubility-product constant): The equilibrium constant for the dissolution of an ionic solid into its constituent ions, written using equilibrium ion concentrations (or activities), with the solid omitted.
In AP Chemistry, is typically expressed using molar concentrations of ions at equilibrium, even though a more rigorous treatment uses activities. This approximation is what allows solubility rules to be discussed quantitatively with .
Writing to make solubility “quantitative”
For a generic ionic solid, the balanced dissolution reaction determines the exponents in the expression.

This ICE-table setup for shows how dissolution stoichiometry maps directly onto equilibrium ion concentrations. With initial ion concentrations of 0, each ion increases by , giving equilibrium values of and for and . Substituting these into makes the connection between the balanced equation and the equilibrium expression explicit. Source
= solubility-product constant (unitless by convention in AP)
= equilibrium molar concentration of the cation,
= equilibrium molar concentration of the anion,
= stoichiometric coefficients from the balanced dissolution equation
The solid is omitted because its “concentration” does not change as long as some undissolved solid remains; its effect is built into the constant value of at a given temperature.
Interpreting “soluble” vs “insoluble” using
Solubility rules classify salts into broad categories. refines that classification by indicating how far the dissolution equilibrium lies to the right.
Slightly soluble (rule-of-thumb “insoluble”)
Small corresponds to an equilibrium that lies far toward the solid.
This means only a small amount of solid dissolves before the ion product reaches .
Many salts labelled “insoluble” by rules have tabulated values specifically because their limited dissolution is experimentally distinguishable and important.
Soluble (rule-of-thumb “soluble”) and the idea
In the framework referenced in the syllabus, is associated with salts classified as soluble:
A large equilibrium constant indicates product-favoured dissolution, meaning the ions are strongly favoured relative to the undissolved solid.
Practically, “soluble” salts dissolve so extensively that the limiting factor is often the amount added, not the equilibrium position.
Because very soluble salts dissolve nearly completely, their values are not always tabulated in introductory tables; the equilibrium lies so far right that the concept is less useful for predicting a small equilibrium solubility.
Important cautions when linking rules to
magnitude is not the same as “molar solubility” for every salt
Even within this quantitative framework:
depends on stoichiometry through the exponents.
Two salts can have different values yet not follow the same ranking in “how many moles dissolve per litre,” because the number of ions produced per formula unit changes the equilibrium expression.
“Solubility rules” are conditional, but is specific
Solubility rules are broad patterns that can have exceptions.
is compound-specific (and temperature-specific), so it provides a more precise basis for calling something “soluble” or “insoluble” under defined conditions.
FAQ
If dissolution lies overwhelmingly to the right, the system behaves like near-complete dissociation under typical conditions, so “slightly soluble” equilibria are the main focus of $K_{sp}$ tables.
Yes. “Insoluble” groups many compounds with low solubility; $K_{sp}$ separates them quantitatively because each solid has its own equilibrium position.
Not always in practice, because observed dissolution can be limited by kinetics, particle size, mixing, and the amount of solvent relative to solute, even if equilibrium favours ions.
Different formulas produce different numbers of ions, changing the exponents in $K_{sp}$. The same “amount dissolved” can generate very different ion products depending on stoichiometry.
Using concentrations approximates activities and works best in dilute solutions. In more concentrated ionic solutions, activity effects can shift the apparent relationship between rule-based “soluble” and numerical $K_{sp}$.
Practice Questions
(2 marks) Explain why a pure ionic solid does not appear in the expression for its dissolution equilibrium.
Pure solid has effectively constant activity/concentration during equilibrium (1)
Therefore it is incorporated into the constant and omitted from the expression (1)
(5 marks) A student states: “If a salt is soluble, its must be greater than 1.” Using equilibrium ideas, assess this statement and explain how solubility rules can be linked quantitatively to .
Links solubility rules to dissolution equilibrium and as the equilibrium constant for that process (1)
Explains that larger means equilibrium favours dissolved ions (more product-favoured) (1)
States the syllabus framework: corresponds to salts classified as soluble (1)
Notes that depends on stoichiometry (exponents), so comparing “solubility” across different formulas is not purely by magnitude alone (1)
Recognises solubility rules are qualitative/approximate, whereas is quantitative and condition-specific (e.g., temperature) (1)

