AP Syllabus focus: ‘Because total energy is conserved, the net thermal energy transferred equals the sum of thermal energy transfers for the individual steps.’
Energy changes in chemistry often occur through multiple conceptual “steps.” This page explains why you can add those stepwise enthalpy changes to get an overall , rooted in energy conservation.
Energy Conservation and “Adding” Enthalpy Changes
At the core of thermochemistry is energy conservation: energy can be transferred and transformed, but the total energy of the universe does not change. In lab problems, we track energy moving as heat (and sometimes work), and we report reaction energy changes using enthalpy.
Why energy conservation implies additivity
If a process is carried out in several steps that start at the same initial state and end at the same final state, then:
The overall energy change depends only on the initial and final conditions, not on the pathway.
Each step transfers some amount of energy.
By conservation of energy, the energy transferred over the full pathway must equal the sum of the energy transferred in the individual steps.
This is why thermochemical “step” equations can be combined: the energy bookkeeping must remain consistent.
Enthalpy as a State Function (Why Path Doesn’t Matter)
The reason this works specifically with is that enthalpy behaves as a state function under typical AP Chemistry conditions.
State function: A property whose value depends only on the current state (conditions) of the system, not on how the system reached that state.
Because enthalpy is a state function, its change for a given start and end state is fixed.
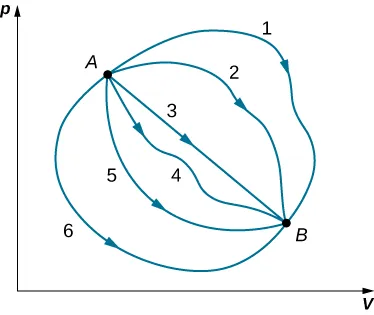
This p–V diagram shows many different thermodynamic paths connecting the same initial state A and final state B. The figure is used to emphasize that heat and work can vary with the path, while the change in a state function between A and B is fixed. This provides a strong visual analogy for why is path independent when initial and final states are the same. Source
Whether you imagine one direct reaction or multiple intermediate reactions, the start and end states (reactants/products in specified states) determine the same total .
A useful mental model is to treat enthalpy like “altitude”: you can climb a hill directly or via switchbacks, but the net altitude change from bottom to top is the same; the individual rises add to the total rise.
Mathematical Statement of Additivity
When you split an overall process into steps and assign an enthalpy change to each step, the overall enthalpy change is the sum of the step enthalpy changes. This is a direct consequence of energy conservation applied to a state function.
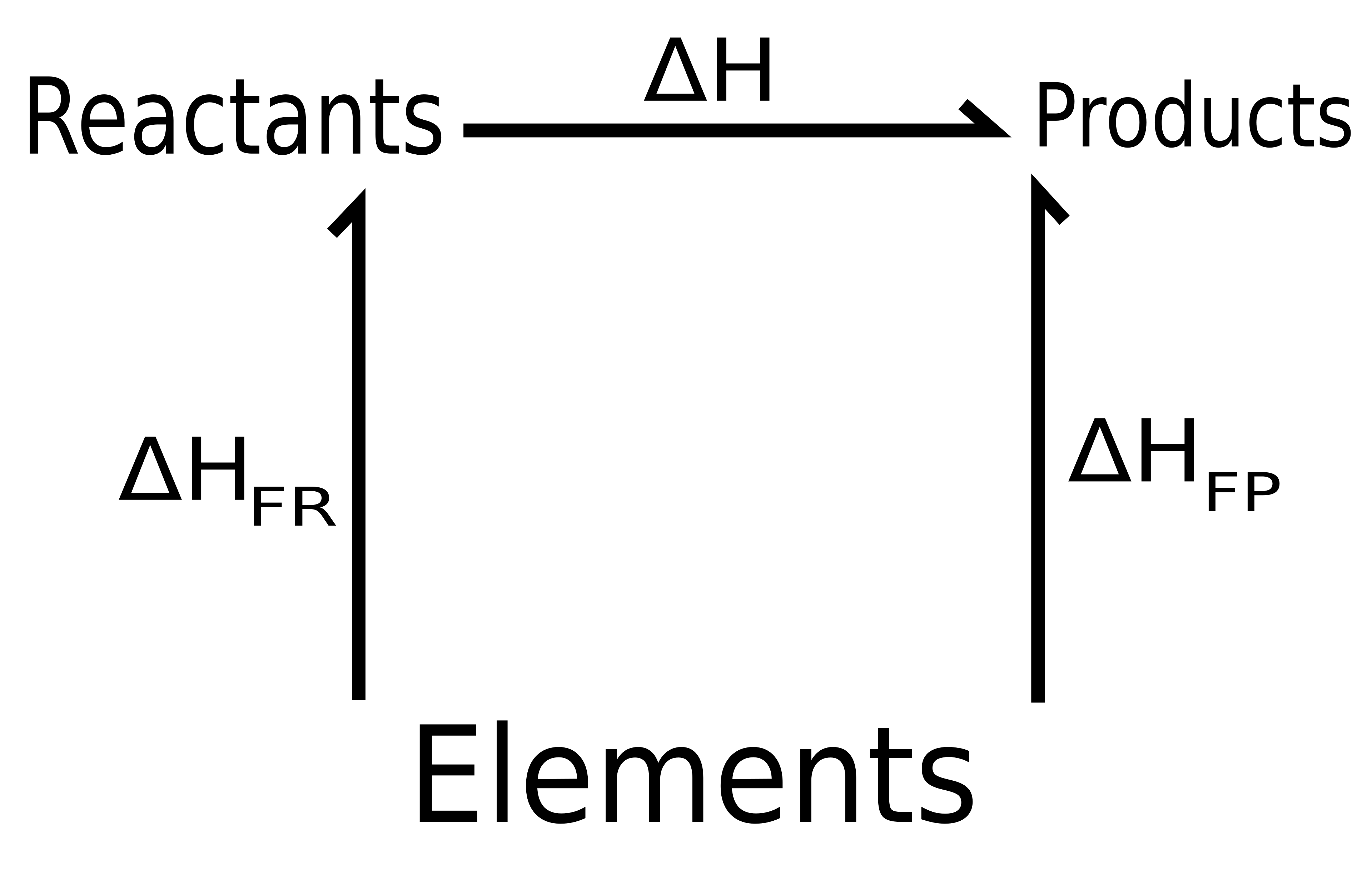
This Hess cycle schematic organizes reactions into an energy cycle connecting reactants, products, and elements in their standard states. It emphasizes that enthalpy changes along alternative routes between the same endpoints must be consistent, so stepwise values can be summed to obtain the net . The cycle layout also supports the bookkeeping idea that intermediate transformations are accounted for even when species cancel algebraically. Source
= enthalpy change for the net process (kJ or kJ/mol as reported)
= enthalpy change for step (kJ or kJ/mol as reported)
= “sum over all steps used to build the net process”
To use this relationship correctly, every step must be defined so that, when added together, the intermediate species cancel and only the desired net reactants and products remain.
What Must Stay Consistent for the Sum to Be Valid
Same initial and final states
The “initial” and “final” states include:
Chemical identity (which substances)
Physical state (s, l, g, aq) and conditions when specified
If the states differ, you are not describing the same overall change, so the summed enthalpy would not correspond to the target process.
Units and interpretation
values may be reported as kJ for a stated amount of reaction, or kJ/mol relative to a balanced equation.
When combining steps, the enthalpy values must match how each equation is written (i.e., “per reaction as written”).
Heat transfer perspective
Even though is a system property change, it is experimentally connected to heat transfer at constant pressure. If a pathway is broken into steps, each step may transfer heat; energy conservation requires the net heat transferred over the full pathway equals the sum of heat transfers for the steps (under the same pressure basis). This is the physical basis for adding enthalpy changes.
Common Conceptual Pitfalls (Energy-Conservation View)
Confusing temperature with : temperature is not a state function in the same way; tracks energy change of the system, not “how hot it gets.”
Forgetting that “path independence” is about states: if an intermediate changes phase or concentration differently, you may have changed the state and therefore changed the enthalpy change you’re summing.
Assuming cancellation implies zero energy: species can cancel algebraically while the corresponding values still add; energy bookkeeping follows the equations’ stated transformations, not the written presence/absence of intermediates.
FAQ
Steps let you combine measurable or tabulated reactions to infer an overall $\Delta H$ that is difficult to measure directly.
This is a bookkeeping strategy, not a claim that the real mechanism occurs in those steps.
No. Conservation means the system’s energy decrease equals the surroundings’ energy increase (and vice versa).
$\Delta H$ can be positive or negative; it is not required to be zero.
When the initial or final states are not truly identical, for example:
different phases (g vs aq)
different allotropic forms
different specified conditions tied to the state description
In AP Chemistry, additivity is applied to $\Delta H$ because it is a state function.
Heat $q$ can depend on the path in general, but at constant pressure for reaction processes, $\Delta H$ provides a consistent state-based measure to sum.
Cancelling intermediates confirms that the algebraic sum of steps represents the same net change of state.
Energy conservation then implies the corresponding enthalpy changes must add to the net $\Delta H$ for that same overall change.
Practice Questions
(2 marks) Explain, using energy conservation, why the overall enthalpy change for a reaction equals the sum of the enthalpy changes for a set of steps that add to the same overall reaction.
States that energy is conserved / total energy change must be the same for the same initial and final states. (1)
States that therefore the net equals (or equivalent wording). (1)
(5 marks) A student proposes two different multi-step pathways that both convert reactants to the same products (same physical states). Using the idea that is a state function and energy conservation, justify whether the two pathways must give the same . Include a statement using symbols (e.g., ).
Identifies that enthalpy change depends only on initial and final states (state function). (1)
Links state-function behaviour to energy conservation/path independence. (1)
States both pathways must yield the same if initial/final states are identical. (1)
Includes a correct symbolic relationship such as . (1)
Notes requirement that the steps sum to the same overall reaction (intermediates cancel) / states must match (including physical states). (1)

