AP Syllabus focus: ‘Molar enthalpy of fusion and vaporization can be used to calculate energy absorbed during melting/boiling and released during freezing/condensing.’
Phase changes involve large energy transfers without changing temperature.
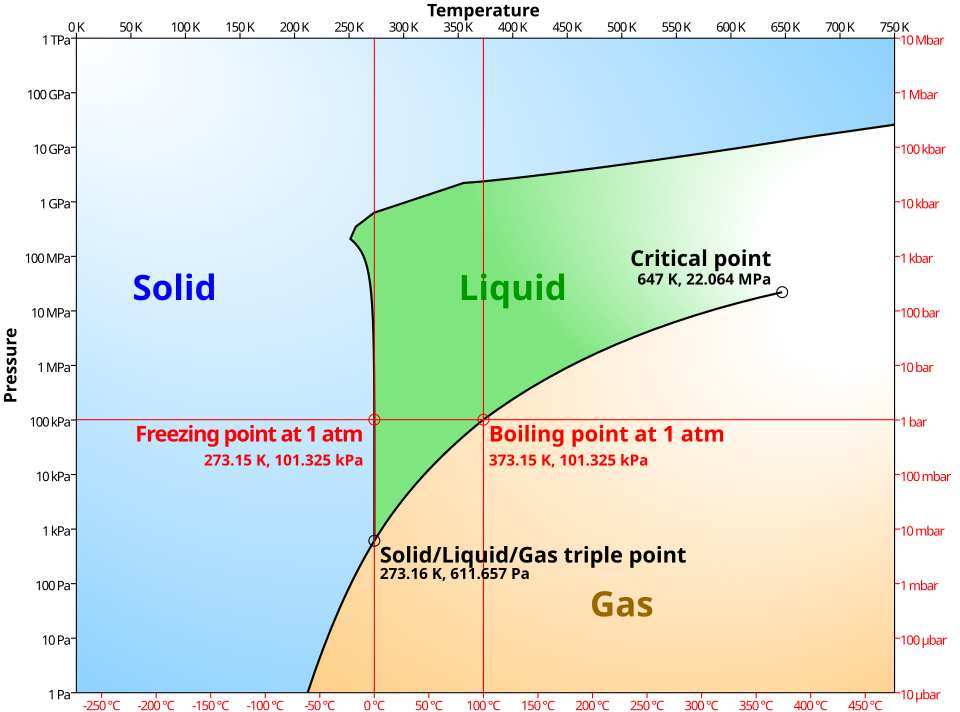
Simplified phase diagram of water showing the regions where solid, liquid, and vapor are stable, separated by equilibrium boundaries (fusion, vaporization, and sublimation curves). The labeled triple point and critical point provide anchor concepts for when phase boundaries (where and apply) are encountered. Source
In AP Chemistry, you quantify these transfers using molar enthalpy of fusion and molar enthalpy of vaporization, scaled by the amount of substance undergoing the phase change.
Molar enthalpies used for phase-change calculations
Molar enthalpy of fusion, ΔHfus
Molar enthalpy of fusion (): The energy absorbed to melt 1 mole of a substance (solid liquid) at its melting point, at constant pressure.
Key points for using :
Applies only to the solid–liquid phase change.
Reported in kJ/mol (or J/mol).
The tabulated magnitude corresponds to melting; the reverse direction uses the opposite sign.
Molar enthalpy of vaporization, ΔHvap
Molar enthalpy of vaporization (): The energy absorbed to vaporise 1 mole of a substance (liquid gas) at its boiling point, at constant pressure.
Key points for using :
Applies only to the liquid–gas phase change.
Reported in kJ/mol (or J/mol).
The tabulated magnitude corresponds to boiling/vaporising; the reverse direction uses the opposite sign.
Core relationship: scaling by moles
In these problems, the phase-change energy is proportional to the number of moles that change phase.
= heat absorbed by the system (positive) or released by the system (negative), in kJ or J
= amount of substance undergoing the phase change, in mol
= molar enthalpy for the relevant phase change (e.g., or ), in kJ/mol or J/mol
Use this relationship only for the phase-change step itself; temperature change steps are handled differently and should not be mixed into this equation.
Choosing the correct molar enthalpy and sign
To match the syllabus language (“absorbed during melting/boiling” vs “released during freezing/condensing”), decide the process direction first.
Melting (solid liquid): energy is absorbed by the system
Use in
Freezing (liquid solid): energy is released by the system
Use
Boiling/vaporising (liquid gas): energy is absorbed by the system
Use
Condensing (gas liquid): energy is released by the system
Use
Sign discipline is essential:
means the system absorbs heat (endothermic phase change).
means the system releases heat (exothermic phase change).
Converting the given sample information into moles
The equation requires in moles. Most AP questions provide a mass and a molar mass (or enough information to find it).
If given mass (in g), find moles using:
Use the moles of the substance actually undergoing the phase change (typically the same chemical formula given for or ).
Common unit checks:
If is in kJ/mol, keep in kJ.
If is in J/mol, keep in J.
Convert only once at the end if needed (1 kJ = 1000 J).
What the calculation represents physically
When you compute or , you are accounting for energy associated with overcoming or forming interparticle attractions during a phase change:
Melting/boiling: energy goes into separating particles enough to change phase.
Freezing/condensing: energy is released as particles come closer and form more stabilising interactions.
These molar enthalpies are used specifically because the temperature does not change during the phase change step; the energy transfer changes the phase, not the kinetic energy distribution.
High-frequency pitfalls to avoid
Do not use for the phase-change segment; use .
Match the correct to the phase boundary:
solid–liquid uses , liquid–gas uses .
Include the correct sign for freezing/condensing (released energy implies negative for the system).
Use the correct amount: the energy depends on how many moles actually change phase, not the total moles present if the process is partial.
Keep significant figures consistent with the given data and avoid mixing kJ and J within the same step.
FAQ
Vaporisation requires much greater separation of particles than melting.
Gas formation typically involves overcoming nearly all intermolecular attractions, whereas melting only partially disrupts them.
$\Delta H_{vap}$ depends on the boiling temperature, which changes with pressure.
At higher external pressure, the boiling point increases; the measured $\Delta H_{vap}$ at that new temperature can differ from the standard tabulated value.
“Per mole” refers only to the amount that actually undergoes the phase change.
If a process stops early, use moles transformed (not initial moles present) when applying $q=n\Delta H_{phase}$.
They are typically obtained by calorimetry at the phase-change temperature.
A known heat input (or output) is related to the measured amount of substance that melts or vaporises at constant pressure.
They are best used near the stated conditions (often 1 bar and the normal melting/boiling point).
Far from those conditions, phase-change enthalpies can vary with temperature and pressure, so additional data or assumptions may be required.
Practice Questions
(1–3 marks) A sample of 12.0 g of water freezes at C. Given , determine the heat change for the water (sign included).
Convert mass to moles: (1)
Freezing releases heat: use (1)
(1)
(4–6 marks) A 0.250 mol sample of a substance is first melted, then completely vaporised at its phase-change temperatures. Given and , calculate the total heat absorbed by the system for both phase changes.
Identify both steps are endothermic: and (1)
Calculate (1)
Calculate (1)
Add phase-change heats: (1)
State absorbed by system, so positive: (1)

