AP Syllabus focus: ‘Energy absorbed in a phase change equals energy released in the reverse change; for example, ΔHcondensation = −ΔHvaporization.’
Phase changes have consistent energy-sign patterns that help you reason about heat flow without memorising separate values. This page focuses on complementary phase changes and the sign conventions used for their enthalpy changes.
Complementary phase changes: the paired processes
A phase change can occur in two opposite directions. These paired, opposite directions are called complementary phase changes.
Complementary phase changes: A pair of opposite phase transitions between the same two phases (e.g., melting and freezing) whose enthalpy changes have equal magnitude and opposite sign.
Common complementary pairs include:
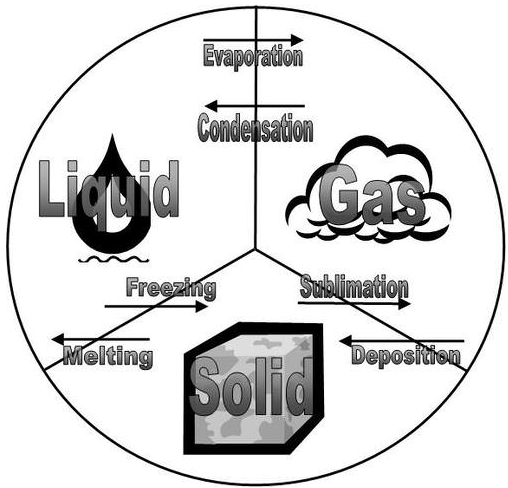
A states-of-matter transition diagram that links solid, liquid, and gas with the six common phase changes. Because each arrow represents a reversible path between the same two phases, it supports the idea that reversing a phase change flips the sign of while preserving its magnitude. This is a compact visual organizer for the complementary pairs you list. Source
Melting (solid to liquid) and freezing (liquid to solid)
Vaporisation (liquid to gas) and condensation (gas to liquid)
Sublimation (solid to gas) and deposition (gas to solid)
Each pair connects the same two phases, so the only difference is direction of energy transfer.
Sign conventions for enthalpy change (Delta H) in phase changes
For AP Chemistry, the key sign convention is defined from the system’s perspective:
Positive Delta H: the system absorbs thermal energy from the surroundings (endothermic).
Negative Delta H: the system releases thermal energy to the surroundings (exothermic).
Enthalpy change of a phase change (Delta H): The heat transferred at constant pressure when a substance changes phase, reported with a sign that indicates whether the system absorbs (positive) or releases (negative) energy.
This immediately classifies directions:
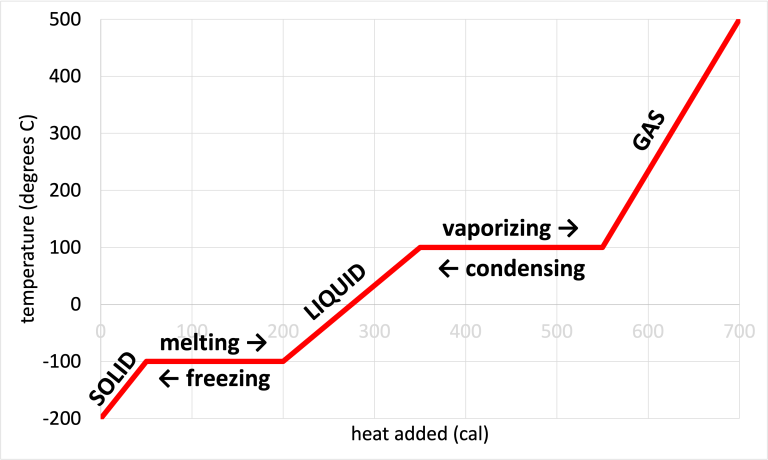
A labeled phase-change (heating/cooling) curve showing temperature vs. heat added, with plateaus at melting/freezing and vaporizing/condensing. The plateaus emphasize that energy transfer during a phase change changes intermolecular arrangements rather than temperature. The arrows also make the endothermic vs. exothermic directionality visually explicit. Source
Endothermic directions (system gains energy): melting, vaporisation, sublimation
Exothermic directions (system loses energy): freezing, condensation, deposition
Why complementary changes have equal magnitude and opposite sign
When a substance goes from phase A to phase B, the energy change depends on the difference in enthalpy between those two phases at the stated conditions. Reversing the direction swaps initial and final states, so the enthalpy change must change sign while keeping the same size.
This is the syllabus idea stated directly: energy absorbed in one direction equals energy released in the reverse direction (for example, condensation versus vaporisation).
= enthalpy change for the complementary (reverse) phase change, in kJ mol
= enthalpy change for the phase change written in the forward direction, in kJ mol
A practical way to say this (in words) is: “flip the process, flip the sign.”
Applying the convention to named phase-change enthalpies
AP Chemistry often uses “molar enthalpy of” language for phase changes. You do not need a new number for the reverse process; you only need the sign change.
Vaporisation and condensation
Delta H of vaporisation is positive because converting liquid to gas requires energy input to separate particles into the gas phase.
Delta H of condensation is negative because converting gas to liquid releases that same energy as particles come closer together.
So, by sign convention, the statement “Delta H condensation equals negative Delta H vaporisation” must be true when both refer to the same substance and conditions.
Fusion (melting) and freezing
Delta H of fusion is positive for melting.
Delta H of freezing is negative with the same magnitude.
Sublimation and deposition
Delta H of sublimation is positive.
Delta H of deposition is negative with the same magnitude.
Common pitfalls with signs and wording
Confusing “heat released” with “temperature decreases”: a phase change can release energy even when temperature does not change during the transition.
Forgetting the system viewpoint: Delta H describes the system’s energy change, not the surroundings’.
Mixing up the pair: condensation is the reverse of vaporisation (not the reverse of melting).
Dropping conditions: the “equal magnitude, opposite sign” relationship assumes the same substance undergoing the reverse process under the same stated conditions.
FAQ
Not exactly.
The relationship is exact for a forward process and its reverse at the same stated conditions. If pressure (or temperature) changes, the measured values can differ, so you cannot assume equal magnitudes across different conditions.
They refer to the same transition: solid to liquid.
“Fusion” is the traditional thermochemistry term for melting. The reverse is “freezing” (sometimes “solidification”), which is the complementary change with opposite sign.
Yes, if the forward direction is defined as an exothermic phase change.
For example, if you define the process as “condensation,” its Delta H is negative by convention because the system releases energy.
In AP-style usage, the sign belongs to the process as written.
So “enthalpy of vapourisation” is reported as positive because vapourisation is defined as liquid to gas. If you write the reverse process, you must change the sign.
They can.
Impurities can change the energetic and physical details of the transition, so the measured enthalpy for a real sample may differ from that of the pure substance. The reverse-process sign flip still applies for the same sample and conditions, but the numerical value may not match a pure-substance table value.
Practice Questions
The molar enthalpy of vapourisation of a substance is at its boiling point. State the molar enthalpy of condensation and explain your sign.
States (1)
Identifies condensation as the reverse of vapourisation (1)
Explains that reversing a phase change reverses the sign because energy released equals energy absorbed in the forward direction (1)
Explain, using sign conventions and the idea of complementary phase changes, why the enthalpy change for freezing has the same magnitude as the enthalpy change for melting but an opposite sign. Your answer should refer to energy absorbed/released by the system and the direction of the phase change.
States melting is endothermic: system absorbs energy, Delta H positive (1)
States freezing is exothermic: system releases energy, Delta H negative (1)
Identifies freezing as the complementary (reverse) process to melting (1)
Explains that the two processes connect the same phases and differ only in direction (1)
Explains that reversing initial and final states changes the sign of Delta H (1)
States magnitude is equal because the energy absorbed in melting equals the energy released in freezing under the same conditions (1)

