AP Syllabus focus: ‘Melting or boiling requires energy transfer to the system, increasing its energy while temperature remains constant during the phase change.’
Melting and boiling are endothermic phase changes: energy flows into the substance, yet the thermometer can “stall.” Understanding why temperature stays constant requires connecting temperature to particle kinetic energy and phase changes to intermolecular attractions.
What “constant temperature” means during melting and boiling
Temperature reflects the average kinetic energy of particles. During a phase change at the melting point or boiling point, added energy does not increase average particle speed, so the measured temperature remains constant until the phase change is complete.
Phase change: A physical transition between states of matter (such as solid to liquid or liquid to gas) that occurs with energy transfer but without changing chemical identity.
A substance can absorb energy continuously while its temperature is constant because the energy is being redirected into changing how particles are arranged and how strongly they attract each other.
Melting: energy increases at the melting point
Particle-level explanation
In a solid, particles occupy relatively fixed positions and experience strong intermolecular forces (IMFs) that restrict motion. As heat is supplied:
At first, temperature rises (particles vibrate faster).
Once the melting point is reached, additional energy is used to partially overcome IMFs and loosen the ordered structure.
The system’s potential energy increases (particles become less tightly bound), while average kinetic energy stays about the same.
What happens macroscopically
During melting, both solid and liquid can coexist.
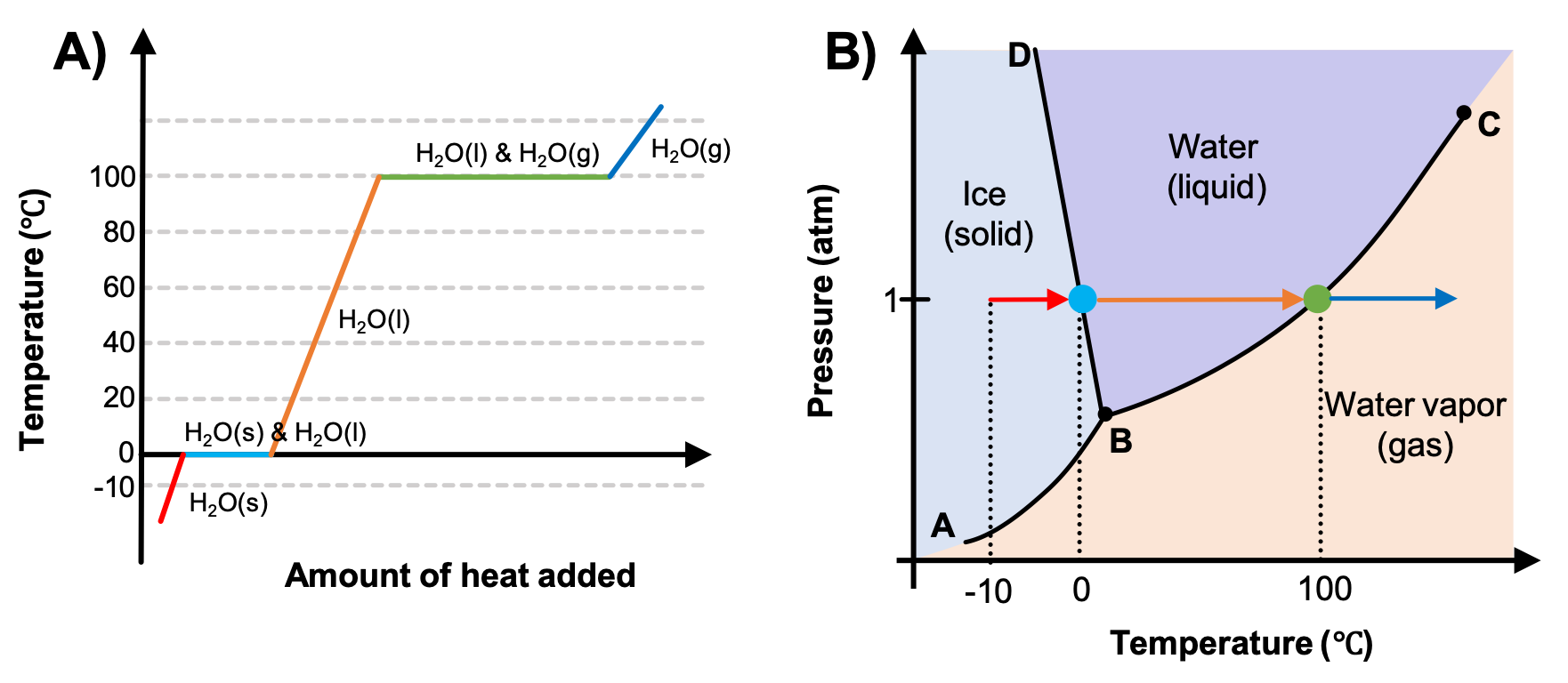
A heating curve for water (temperature vs. heat added) shows sloped regions where added heat increases temperature and flat plateaus where added energy is used for phase change at constant temperature. The accompanying phase diagram links each plateau to crossing a phase boundary at a fixed pressure, reinforcing that melting and boiling occur at specific conditions. Source
Heat input goes into converting more solid to liquid rather than raising temperature, producing a “plateau” on a heating curve.
Enthalpy of fusion (): The energy absorbed per mole to convert a substance from solid to liquid at its melting point (at a specified pressure, typically 1 atm).
Melting requires energy because separating particles enough to move past one another demands work against IMFs.
Boiling: energy increases at the boiling point
Particle-level explanation
In a liquid, particles are already mobile but still held together by IMFs. When the liquid reaches its boiling point:
Added energy enables particles to separate far enough to form the gas phase.
Energy input primarily increases potential energy by overcoming IMFs (and allowing expansion), not average kinetic energy.
Temperature remains constant until essentially all liquid has become gas.
What happens macroscopically
Boiling occurs throughout the liquid when vapor bubbles can form and persist. During the boiling plateau, the system absorbs energy continuously even though the temperature does not increase.
Enthalpy of vaporization (): The energy absorbed per mole to convert a substance from liquid to gas at its boiling point (at a specified pressure, typically 1 atm).
Because gas-phase particles are much more separated than liquid-phase particles, boiling typically requires substantially more energy than melting for the same amount of substance.
Quantifying energy absorbed during melting or boiling
Energy transferred during a phase change is proportional to how much substance changes phase, not to a temperature change.
= heat absorbed during melting (J or kJ)
= amount melted (mol)
= enthalpy of fusion (J/mol or kJ/mol)
= heat absorbed during boiling (J or kJ)
= amount boiled (mol)
= enthalpy of vaporization (J/mol or kJ/mol)
For both melting and boiling, is positive for the substance undergoing the phase change because energy is transferred into the system, increasing its energy while temperature remains constant during the phase change.
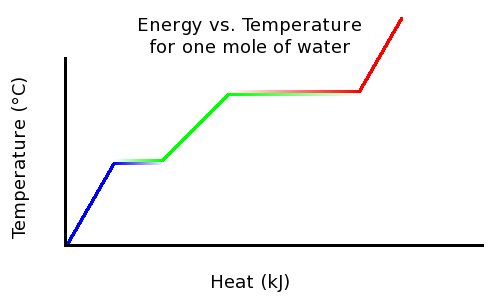
This temperature-vs.-heat heating curve highlights that energy input continues during the flat (constant-temperature) regions corresponding to melting and boiling. Those plateaus represent latent heat: the added energy increases potential energy by overcoming intermolecular attractions rather than increasing average kinetic energy. Source
Common pitfalls to avoid
Confusing the plateau with “no heat added”: energy is still being absorbed; it is just not raising temperature.
Saying particles “stop moving” during the plateau: particles continue moving; the key is that average kinetic energy stays roughly constant while potential energy increases.
Treating melting/boiling like chemical reactions: no new substances form; only particle arrangement and spacing change.
FAQ
Boiling separates particles much further than melting does, so far more intermolecular attractions must be overcome.
This larger change in particle spacing corresponds to a larger increase in potential energy.
Energy is being redistributed into separating particles and enabling escape into the new phase.
Average particle speed does not increase much, so the measured temperature stays the same.
Yes, if it is not pure. Mixtures can show a melting/boiling range because different components change phase at different temperatures.
Impurities also disrupt orderly melting behaviour.
Boiling occurs when vapour pressure matches external pressure; changing external pressure shifts the temperature at which this happens.
Lower pressure generally lowers the boiling point; higher pressure raises it.
Both are at the boiling point (for that pressure), so their temperatures match.
More vigorous boiling reflects a higher rate of energy transfer (more liquid converting to gas per unit time), not a higher temperature.
Practice Questions
(2 marks) A pure substance is heated at constant pressure. Its temperature remains constant for several minutes while heating continues. Identify the process occurring and state what happens to the substance’s internal energy during this time.
Identifies melting or boiling as the process occurring at constant temperature (1)
States internal energy increases / energy is absorbed by the system (1)
(5 marks) Explain, in terms of particles and energy, why the temperature of a pure liquid remains constant at its boiling point while heat is continually supplied. Your answer should refer to kinetic energy, intermolecular forces, and the energy change of the system.
States temperature is proportional to average kinetic energy; at the boiling point average kinetic energy stays constant (1)
Explains supplied energy is used to overcome intermolecular forces / increase particle separation (1)
Links this to an increase in potential energy rather than kinetic energy (1)
States the system absorbs heat / is endothermic / (1)
Notes temperature stays constant until the phase change is complete (1)

