AP Syllabus focus: ‘Energy diagrams can represent physical or chemical processes and show whether the process is endothermic or exothermic.’
Energy diagrams visually connect energy changes to what happens during a process. By reading relative energy levels for reactants and products, you can determine whether energy is absorbed or released and interpret what the curve implies physically.
What an energy diagram shows
An energy diagram plots energy (often potential energy/enthalpy) versus progress of the process (sometimes called reaction coordinate).
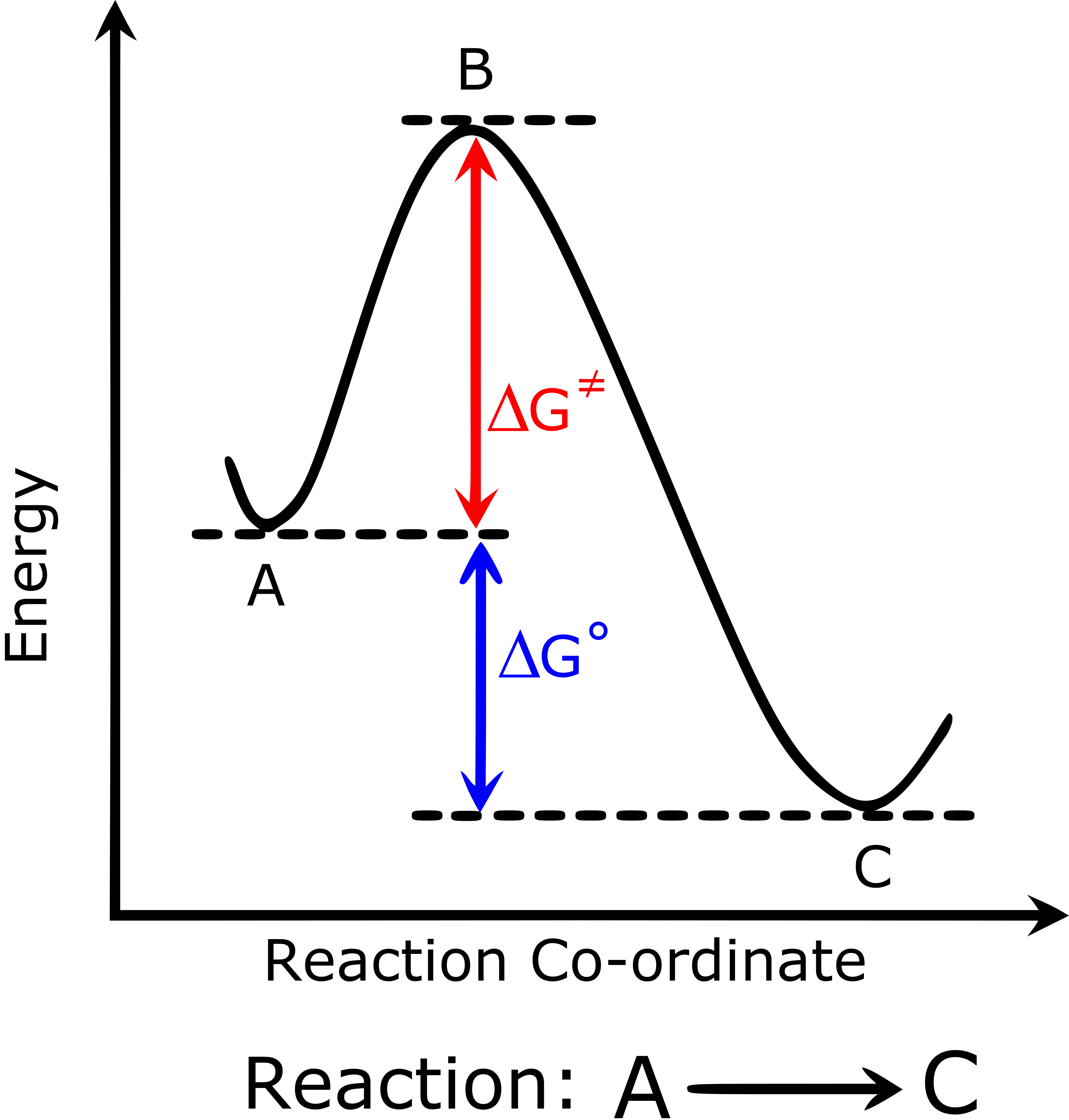
Reaction-coordinate (potential energy) diagram showing reactants and products connected through a higher-energy transition state (the peak). The peak height corresponds to the activation energy barrier, while the vertical difference between reactant and product levels corresponds to the overall energy change () for the process. Source
The key skill is comparing the energy level at the start to the energy level at the end.
Energy diagram: A graph that shows how the energy of a system changes as a physical or chemical process proceeds, allowing comparison of reactant and product energy levels.
Core features to locate
Reactants (initial state): the starting energy level on the left.
Products (final state): the ending energy level on the right.
Energy change (vertical difference): how much higher or lower products are than reactants.
Peak/“hump” (if shown): an energy barrier along the pathway; it does not determine endo/exo classification.
Energy diagrams can describe physical processes (like a phase change) or chemical processes (a reaction). In both cases, the endo/exo decision is based on the initial-versus-final energy comparison.
Endothermic vs. exothermic from the diagram
The diagram indicates whether the system’s energy increases or decreases during the process.
Enthalpy change (ΔH): The energy difference between products and reactants at constant pressure; it is negative for exothermic processes and positive for endothermic processes.
If the products are higher on the energy axis than the reactants, the system ended with more energy than it started with:
Endothermic: energy is absorbed by the system from the surroundings
Sign: ΔH is positive
Diagram cue: products drawn above reactants
If the products are lower than the reactants, the system ended with less energy:
Exothermic: energy is released from the system to the surroundings
Sign: ΔH is negative
Diagram cue: products drawn below reactants
The size of the vertical gap corresponds to the magnitude of the energy change; bigger gap means a larger energy absorbed or released.
= enthalpy change of the process (typically kJ or kJ·mol)
= enthalpy (energy level) of the products on the diagram (same units as )
= enthalpy (energy level) of the reactants on the diagram (same units as )
When reading, focus on the relative heights, not the absolute numbers: the “zero” on an energy axis is often an arbitrary reference.
Interpreting labels and common diagram styles
Physical vs. chemical processes
Energy diagrams for physical changes and chemical reactions are read the same way:
If the final state is higher: endothermic
If the final state is lower: exothermic
The main difference is what the endpoints represent (phases/arrangements for physical changes versus substances for reactions), not how you determine the sign.
Multi-step curves
Some diagrams show multiple peaks and valleys (several steps).
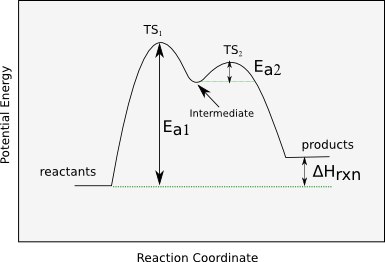
Multi-step energy diagram with more than one transition state (multiple peaks) and an intermediate (valley) between steps. Despite pathway complexity, the overall sign of is determined only by comparing the initial reactant energy level to the final product energy level. Source
To decide endo/exo:
Compare the very first energy level (initial state) to the very last energy level (final state).
Ignore intermediate bumps for classification; they reflect pathway complexity, not the overall energy sign.
Frequent reading mistakes to avoid
Confusing temperature with the energy axis: energy diagrams show energy levels, not a thermometer reading.
Using the peak height to decide endo/exo: the peak is a barrier along the path; only the start-to-finish difference determines endothermic vs exothermic.
Reversing the sign: always compute “final minus initial” (products minus reactants) when using ΔH.
FAQ
An energy diagram classifies the system’s energy change. If the surroundings warm up, it may be due to a separate process (e.g., heat from the container or environment) occurring alongside the system change.
Not always. Some use “potential energy” generically. For AP-level reading, treat the vertical axis as the system’s energy and use relative heights to decide endothermic vs exothermic.
You generally cannot determine a numerical value without a scale. You can still compare relative magnitudes: a larger vertical gap between reactants and products implies a larger $|\Delta H|$.
Only differences in energy are physically measurable for these diagrams. The baseline can be chosen for convenience, so absolute values depend on the reference level.
If reactants and products are at the same energy level, then $\Delta H = 0$. The process is neither endothermic nor exothermic overall, even if the curve shows peaks along the way.
Practice Questions
An energy diagram shows products at a higher energy level than reactants. Is the process endothermic or exothermic, and what is the sign of ? (2 marks)
Identifies process as endothermic (1)
States is positive, (1)
A process has an energy diagram where reactants are at 120 kJ, products are at 45 kJ, and there is a peak at 200 kJ. (a) Determine the sign of . (b) Calculate for the process using . (c) State whether the peak affects whether the process is endothermic or exothermic. (5 marks)
(a) Correct sign: exothermic / (1)
(b) Uses (1)
(b) Calculates kJ (1)
(c) States peak does not determine endo/exo classification; start vs end does (1)
(c) Identifies peak as an energy barrier along the pathway (1)

