AP Syllabus focus: ‘Combine stoichiometry with the ideal gas law and with molarity-based solution calculations to study reactions quantitatively.’
Stoichiometry with gases and solutions extends mole-ratio reasoning to real laboratory measurements: pressures, volumes, temperatures, and solution concentrations. You will convert measured gas or solution data to moles, apply reaction coefficients, then convert to the requested quantity.
Core idea: convert to moles, use coefficients, convert back
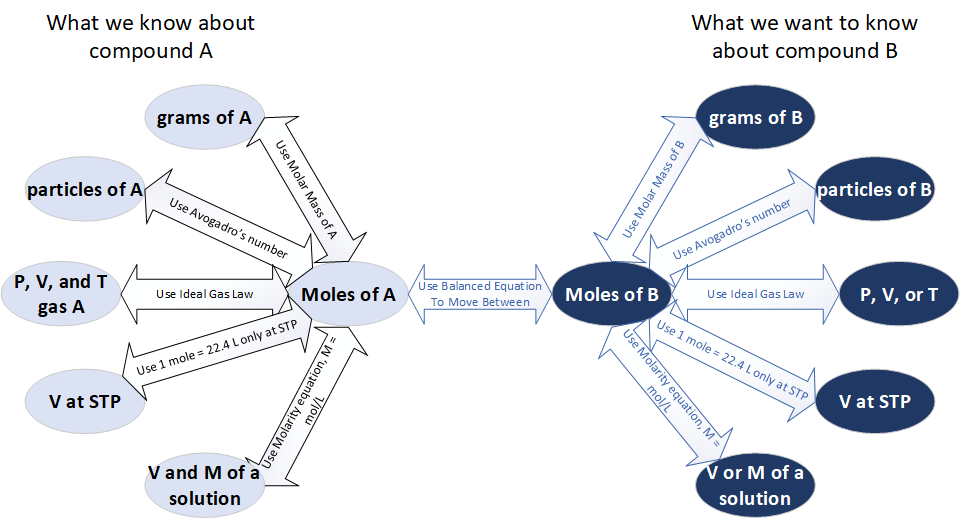
A stoichiometry “roadmap” showing that virtually all quantitative reaction problems pass through moles: convert the given quantity to moles, apply the balanced-equation mole ratio, then convert moles to the desired unit. This is the same logic used for gas data (via ) and solution data (via ), but presented as a single, unified process. Source
Chemical equations relate substances by mole ratios (coefficients). For gas and solution data, the central skill is choosing the correct “bridge” to moles.
A reliable workflow is:
Write a balanced chemical equation.
Convert given quantities to moles (gas: ideal gas law; solution: molarity).
Apply stoichiometric mole ratios from the balanced equation.
Convert moles of the target substance into the requested unit (gas volume/pressure, solution concentration, mass, etc.).
Stoichiometry with gases (ideal gas law)
Most AP problems treat reaction gases as behaving ideally under the stated conditions.
Ideal gas: A model gas whose particles have negligible volume and no intermolecular forces, so its behaviour is described by the ideal gas law.
Use consistent units (especially for pressure and volume) with the gas constant selected. Unit consistency is often the deciding factor in getting the correct magnitude.
= amount of gas, in
= pressure, commonly in (or with a matching )
= volume, commonly in
= gas constant, unit-set dependent (e.g., )
= temperature, in
In reaction contexts, gas stoichiometry commonly involves:
A gaseous reactant consumed (use initial to find initial moles available).
A gaseous product formed (use moles predicted by coefficients, then infer gas volume or pressure at stated and ).
Changing conditions between “before” and “after” states; treat each state with its own and relate through moles.
Stoichiometry with solutions (molarity)
Solution stoichiometry typically starts with concentration and volume and converts to moles of solute.

A labeled relationship diagram for solution calculations that connects moles of solute (), concentration ( or ), and solution volume (). It reinforces that molarity problems are fundamentally “moles per volume,” so converting between measured volume and chemical amount is a straightforward algebraic rearrangement. Source
Molarity (M): The concentration of a solution expressed as moles of solute per litre of solution.
Molarity applies to the dissolved species as written in the reaction. If the reacting species is an ion from a strong electrolyte, problems may implicitly treat it as fully dissociated; the mole accounting must match the reaction equation you are using.
= moles of solute, in
= molarity, in
= solution volume, in
Common uses include:
Finding moles of reactant delivered from a measured solution volume.
Determining an unknown concentration from known moles reacted (often at completion).
Predicting moles of product formed in solution-phase reactions, then converting to mass or a gas quantity if a gas is produced.
Combining gas law and molarity in one reaction
Many reactions link an aqueous reactant amount to a gaseous product (or vice versa). The key is to perform two conversions with moles in the middle:
Solution measurement moles (via )
Mole ratio (from coefficients)
Moles gas measurement (via ), or the reverse direction
When combining these tools, be especially careful to:
Use Kelvin for temperature.
Use litres for solution volumes in molarity calculations.
Ensure the balanced equation matches the chemical species you are counting (molecules vs ions) so the coefficient-based mole ratio is valid.
FAQ
Use $PV=nRT$ whenever $P$ and $T$ are not explicitly at a stated reference condition, or when the problem gives non-standard pressure/temperature.
It is also safer when unit consistency and significant figures matter.
Subtract water vapour pressure from the total pressure: $P_{\text{gas}}=P_{\text{total}}-P_{\mathrm{H_2O}}$.
Then use $P_{\text{gas}}$ in $PV=nRT$. Water vapour pressure depends on temperature.
Molarity can change because solution volume changes with temperature.
Unless data are provided, AP-style questions typically treat stated molarity as valid at the conditions used.
Using $\mathrm{mL}$ instead of $\mathrm{L}$ in $n=MV$, and using Celsius instead of Kelvin in $PV=nRT$.
Also watch pressure units: $\mathrm{atm}$ vs $\mathrm{kPa}$ must match the chosen $R$.
Only if the problem explicitly indicates high pressure/low temperature or provides a correction method.
In typical AP Chemistry contexts, gases are assumed ideal unless told otherwise.
Practice Questions
(2 marks) A reaction produces . The gas collected has , , and . Calculate the moles of produced.
Uses with in K and consistent units (1)
Correct numerical value for with units (1)
(5 marks)
of reacts completely with excess . Calculate the volume of produced at and .
Calculates moles of using with in L (1)
Uses stoichiometric ratio to find moles (2)
Uses (or equivalent) (1)
Correct volume with appropriate units and significant figures (1)

