AP Syllabus focus: ‘At extremely high pressures, the volume occupied by gas particles becomes significant, contributing to non-ideal behavior compared with the ideal gas model.’
Ideal gases assume particles are point-like and occupy no space. Under extremely high pressure, that assumption fails: the particles’ own volume reduces the space available for motion and alters measured relationships among , , , and .
Why Particle Volume Matters at High Pressure
In the ideal gas model, gas particles are treated as having negligible volume, so the container volume is fully available for particle motion. When pressure becomes very large, particles are forced close together, and their finite size becomes a significant fraction of the container volume.
High pressure decreases the average distance between particles.
As spacing shrinks, the “empty” space a particle can move into is less than the container volume.
Collisions occur at shorter separations, and the assumption of perfectly free translational motion in volume becomes inaccurate.
This effect is most noticeable when gases are compressed to small volumes (for example, in high-pressure cylinders), where the particle volume is no longer negligible compared with the container volume.
Connecting to Non-Ideal Behaviour
Non-ideal behaviour means is not exactly proportional to under real conditions.
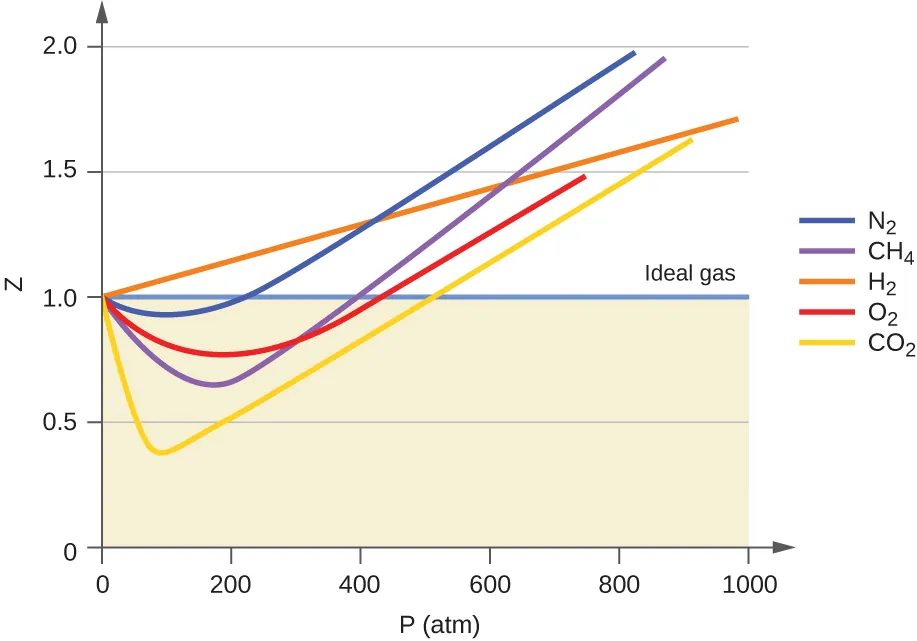
This figure plots the compressibility factor versus pressure for several real gases, with the ideal-gas reference line at . The upward trend at very high pressure illustrates increasing positive deviation, consistent with repulsive/finite-size effects becoming dominant. Source
Finite particle volume changes the relationship because the gas does not have access to the full measured container volume.
Non-ideal behaviour: measured gas properties that do not follow the ideal gas law due to real-particle effects such as finite particle volume (especially at high pressure).
A useful way to describe the direction of the deviation is qualitative: if particles take up space, the effective free volume is smaller than , so the same number of particles at the same temperature will produce a higher pressure than predicted by the ideal model (because they are confined to less usable space).
“Free Volume” and the Excluded-Volume Idea
Real particles behave more like tiny, impenetrable objects than points. Two particles cannot overlap, so each particle effectively blocks a region of space from the centres of other particles. This is often described as excluded volume.
Excluded volume: the portion of volume unavailable for particle centres because real particles have finite size and cannot occupy the same space.
Even without using advanced mathematics, the AP-level takeaway is that at extremely high pressures the measured container volume overstates the volume that actually governs particle motion and collision frequency.
A Common Correction Model (Volume Only)
Chemists sometimes express the finite-volume effect by replacing the container volume with an adjusted volume that accounts for space “taken up” by particles. One widely used form is the van der Waals volume correction (shown here focusing only on finite volume, not attractions).
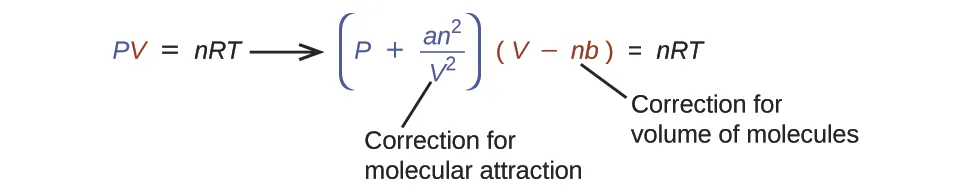
This diagram shows how the van der Waals equation modifies the ideal gas law by correcting both pressure and volume. The volume correction highlights that the usable space for motion is smaller than the measured container volume when particle volume (excluded volume) matters. Source
= effective (free) volume available for particle motion, in L
= container (measured) volume, in L
= amount of gas, in mol
= excluded-volume parameter (volume per mole of particles), in L mol
This equation captures the core syllabus point: at high pressure, the finite particle volume forces you to treat the usable volume as less than . In this model, increasing at fixed and reduces substantially, intensifying non-ideal behaviour.
When the Finite-Volume Effect Becomes Important
Finite particle volume is negligible when the container volume is huge compared with the combined molecular volume—typically at low pressure and moderate temperature. It becomes significant when compression makes small.
Key conditions that amplify the effect:
Extremely high pressures (dominant factor in this subtopic)
Large in a fixed container (many moles crowded into the same volume)
Larger particles (greater molecular size increases the excluded-volume contribution)
As pressure rises, the fraction of the container occupied or blocked by particles increases, so the ideal assumption “particles take up no space” becomes progressively worse.
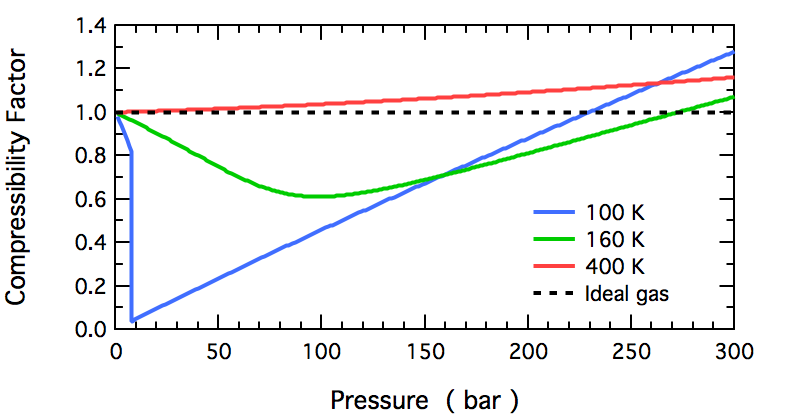
These plots show how the compressibility factor for varies with pressure across multiple temperatures. The approach to at low pressure and the rise to at very high pressure illustrate how real gases become increasingly non-ideal as they are strongly compressed. Source
What You Should Be Able to Explain Conceptually
For AP Chemistry, focus on qualitative reasoning tied to the ideal model’s assumptions.
You should be able to:
State that the ideal gas law assumes zero particle volume, and that this assumption fails at extremely high pressure.
Explain that finite particle volume reduces the available space for motion, so real gases deviate from ideal predictions.
Predict that, due to reduced free volume, the real gas can exert greater pressure than ideal at the same , , and when the finite-volume effect dominates.
Link the strength of this deviation to how tightly the gas is compressed (how “crowded” the particles are), not merely to the identity of the gas.
FAQ
By fitting real-gas measurements (typically high-pressure $P$–$V$ data at fixed $T$) to an equation of state that includes $b$.
Common approaches include regression to a van der Waals-type form or using published tabulated constants derived from such fits.
Not exactly. Excluded volume refers to the space unavailable to molecular centres due to repulsion and finite size.
It is typically larger than the literal sum of molecular volumes because it accounts for the fact that particles cannot approach closer than a certain distance.
Larger or less compressible particles generally have larger effective excluded volumes.
Molecular size and shape influence how closely particles can pack and therefore how quickly $V_{\text{free}}$ shrinks as pressure rises.
$Z=\dfrac{PV}{nRT}$ quantifies deviation from ideality.
When finite particle volume dominates (very high pressure), $Z$ often becomes greater than 1 because the effective free volume is smaller than $V$.
At very high pressures, many gases show pressures higher than ideal predictions at the same $T$, $V$, and $n$.
A practical indicator is that deviations grow strongly with compression even at temperatures where condensation is not imminent, consistent with crowding/finite-size effects.
Practice Questions
Question 1 (2 marks) A sample of gas shows increasing deviation from as pressure is raised from 1 bar to 400 bar at constant . Using the particle model, explain why finite particle volume causes this deviation.
1 mark: States that real gas particles have non-zero/finite volume (not point particles).
1 mark: Explains that at very high pressure particles are close together so the free/available volume is less than the container volume, causing to differ from (e.g., pressure higher than ideal prediction).
Question 2 (5 marks) A chemist compresses 2.00 mol of a gas into a small rigid vessel at constant temperature. The measured pressure is higher than the ideal gas law prediction.
(a) Explain, in terms of finite particle volume, why the ideal model underestimates the pressure. (3 marks)
(b) A simple correction uses . State what physical idea represents and how increasing affects . (2 marks)
1 mark: Identifies that gas particles occupy space/are not point-like.
1 mark: States that at high pressure particles are forced close together so their volume is a significant fraction of .
1 mark: Explains that less free volume means more frequent collisions with the container per unit time (or confinement in smaller effective volume), so actual is higher than ideal prediction. (b)
1 mark: represents excluded volume/volume per mole unavailable due to particle size.
1 mark: Increasing increases , so decreases.

