AP Syllabus focus: ‘The ideal gas law does not fully describe real gases; deviations occur because real particles experience attractions and have nonzero volume.’
Real gases only approximate ideal behavior. Understanding why fails in some situations requires linking the ideal gas model’s assumptions to what actual particles do: attract one another and take up space.
The Ideal Gas Law as a Model
The ideal gas law treats gases as perfectly simple particles, so measured pressure and volume are predicted solely from amount and temperature.
Ideal gas: A hypothetical gas whose particles have negligible volume and no intermolecular attractions, so it obeys under all conditions.
In reality, all gases are made of particles with electron clouds, so they can attract and they occupy finite space. Those two facts create systematic departures from the ideal relationship.
Why Real Gases Deviate from
Two idealising assumptions break down for real gases.
Real gas: An actual gas whose particles have nonzero volume and experience intermolecular attractions, causing measured behavior to deviate from ideal-gas predictions.
These effects do not “turn on” suddenly; they become more influential as particle–particle interactions become harder to ignore.
Cause 1: Intermolecular attractions lower the measured pressure
Gas pressure arises from particle collisions with container walls. The ideal model assumes collisions are unaffected by neighboring particles. Real particles experience attractive intermolecular forces (especially at shorter separations), which changes the momentum delivered to the wall.
If a particle is near the wall, attractions to other particles in the gas can pull it slightly away from the wall just before impact.
As a result, the wall experiences slightly fewer effective impacts and/or slightly less force per impact.
Therefore, the measured pressure of a real gas can be lower than the ideal-gas prediction at the same , , and .
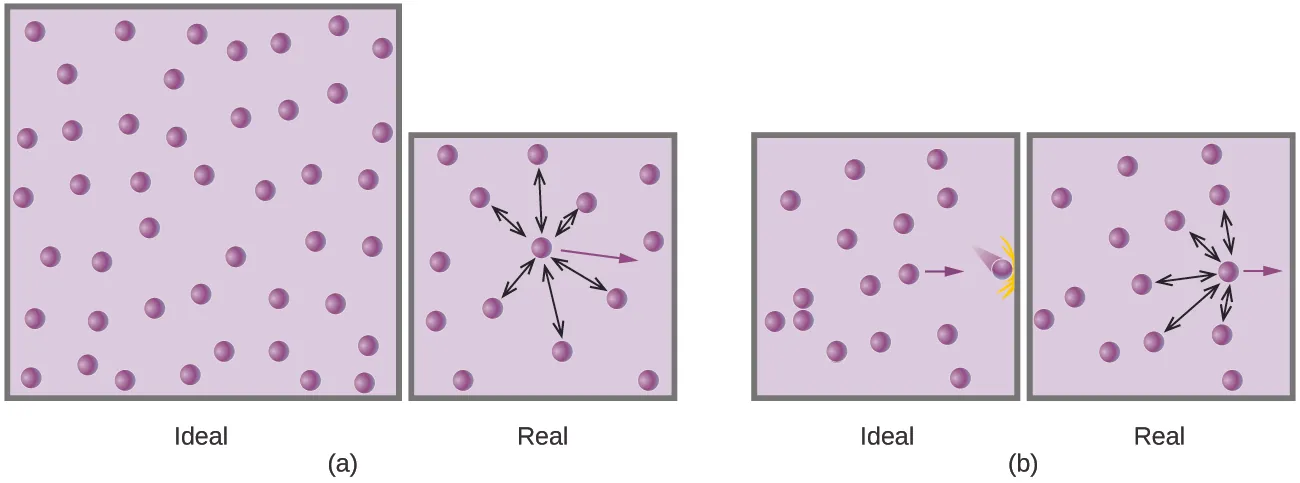
Schematic comparison of ideal vs. real gases showing how intermolecular attractions alter particle trajectories and collision forces. In the real-gas panels, attractive forces between neighboring molecules reduce the effective momentum transfer to container walls, which corresponds to a lower measured pressure under the same macroscopic conditions. Source
Attractions are a direct violation of the “no forces between particles” assumption. They matter most when particles are close enough together and slow enough that attractions noticeably influence trajectories.
Cause 2: Finite particle volume reduces the free space available
The ideal model treats particles as points, so the full container volume is available for particle motion. Real particles occupy space, so some fraction of the container is excluded volume (space that other particles cannot occupy).
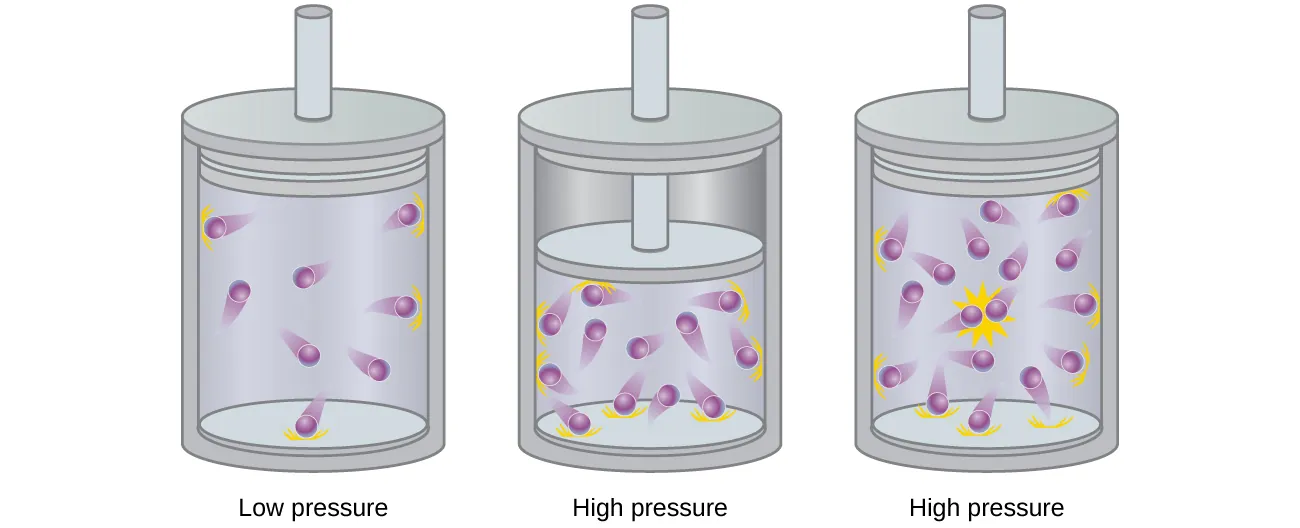
Piston-and-cylinder diagram illustrating that as pressure increases, gas particles are forced closer together so their finite size becomes non-negligible. The visual emphasizes that the free volume available for molecular motion is smaller than the container volume, which helps explain behavior at sufficiently high pressures. Source
The volume in is the container volume, but real particles move within a slightly smaller effective free volume.
With less free volume, particles have less distance to travel between collisions, altering collision frequency and the relationship among , , , and .
This violates the “negligible particle size” assumption.
Both deviations come from particle-level realities: electron clouds produce both size and attractive interactions.
Quantifying deviation with a compressibility factor
A convenient way to describe non-ideal behavior is to compare a gas’s actual value to the ideal expectation.
= compressibility factor (unitless)
= pressure (atm, bar, or Pa)
= volume (L or m)
= amount of gas (mol)
= gas constant (units consistent with and )
= temperature (K)
If , the gas behaves ideally under those conditions.
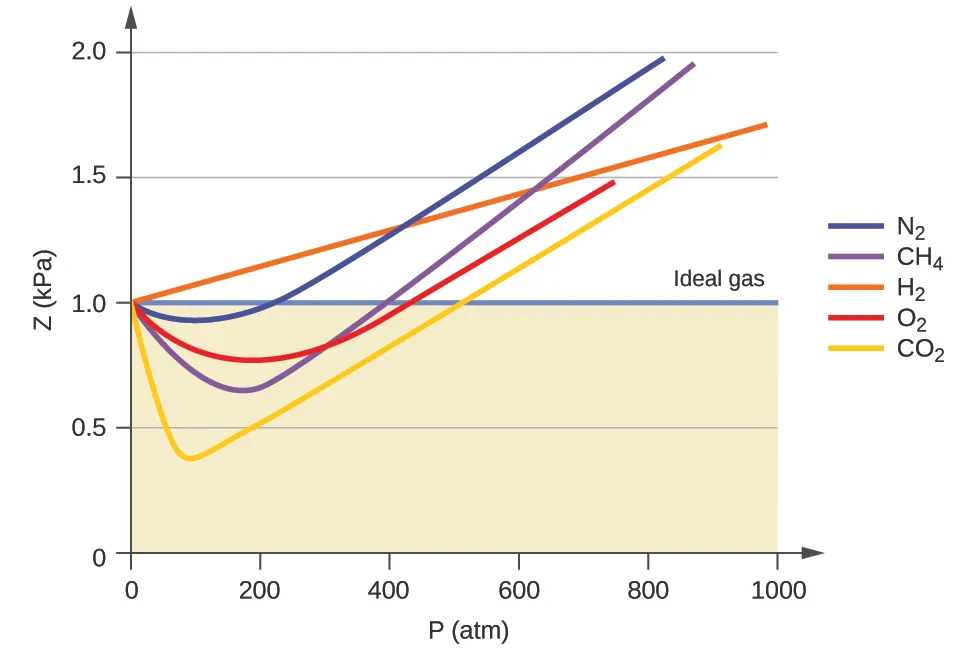
Compressibility-factor plot ( vs. pressure) for several gases, with the ideal-gas baseline shown as . The initial dip below 1 reflects attractive intermolecular forces lowering the measured pressure relative to the ideal prediction, while the rise above 1 at higher pressures reflects finite particle volume and short-range repulsions making the gas less compressible. Source
If , one or both non-ideal effects (attractions and particle volume) are influencing the measured relationship.
Interpreting qualitatively
suggests the gas is “more compressible” than ideal because attractions reduce measured pressure relative to the ideal prediction.
suggests the gas is “less compressible” than ideal because finite particle volume and short-range repulsions make the gas resist compression relative to the ideal prediction.
When the ideal model is most accurate (and why)
Although all gases are real, many conditions make the ideal assumptions approximately valid.
When particles are far apart, the impact of finite volume becomes negligible relative to the container volume.
When attractions are weak compared with particle kinetic energy, the “no attractions” assumption becomes a good approximation.
Under such conditions, measured behavior tends to approach closely, and approaches 1.
Common student pitfalls
Assuming “real gas” means “doesn’t follow gas laws.” Real gases still follow patterns; they simply deviate from the ideal model because the model neglects attractions and particle volume.
Blaming deviations on “chemical reactions.” Deviations discussed here occur even for chemically inert gases; they are physical interaction effects.
Treating as wrong. It is a powerful limiting model; the key is knowing which assumptions fail and what direction the deviations take.
FAQ
Even monatomic noble gases have polarisable electron clouds.
Short-lived induced dipoles create weak attractions, and atoms still occupy finite space, so both ideal assumptions are imperfect.
They change both attraction strength and “effective size”.
More polar molecules: stronger attractions
Larger/less spherical molecules: larger excluded volume effects
No. $Z$ depends on conditions (especially $T$ and $P$) and on the identity of the gas, because attractions and particle size matter differently as spacing changes.
It is the portion of container space that is inaccessible to the centres of other particles due to the finite size of particles and short-range electron-cloud repulsion.
Because their intermolecular potentials differ.
Larger/more polarisable particles: stronger attractions
Larger particles: greater finite-volume impact
Practice Questions
Q1 (2 marks) State two reasons why real gases deviate from the ideal gas law, .
1 mark: Mentions intermolecular attractions/forces between particles.
1 mark: Mentions particles have nonzero (finite) volume/are not point particles.
Q2 (5 marks) A sample of gas has a compressibility factor at a particular temperature and pressure. (a) Define using an equation. (2 marks) (b) Explain what indicates about the dominant non-ideal effect and how it changes the measured pressure compared with the ideal prediction. (3 marks)
(a)
1 mark:
1 mark: Identifies as unitless ratio comparing real behaviour with ideal. (b)
1 mark: States attractions/intermolecular forces are significant/dominant.
1 mark: Links attractions to reduced effective wall collisions (or reduced force per collision).
1 mark: Concludes measured is lower than ideal at same (or equivalently smaller than ).

