AP Syllabus focus: ‘Interparticle attractions cause deviations most noticeably under conditions close to condensation, where attractions affect measured pressure and volume.’
Real gases do not always behave ideally. The most important non-ideal behavior appears when gases are near condensation, because attractive interparticle forces begin to influence both measured pressure and effective volume.
When attractions matter most
What “close to condensation” means
As a gas is cooled and/or compressed, it approaches the point where particles can remain close enough, long enough, for attractions to pull them into a liquid.
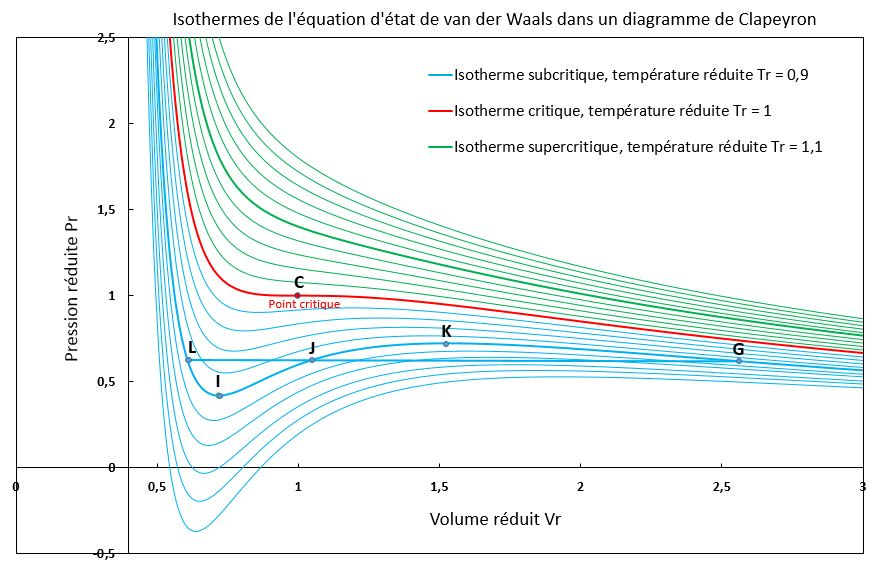
Van der Waals – isotherms showing how real-gas behavior changes as temperature approaches the critical temperature . Above the curves resemble ideal-gas hyperbolas, while near and below the isotherms signal the onset of the gas–liquid transition region associated with condensation. This diagram is a compact way to see why “close to condensation” is exactly where ideal assumptions fail most strongly. Source
Under these conditions, the assumptions behind “ideal” behavior (especially negligible attractions) become less valid.
Condensation — the phase change in which gas particles become a liquid as attractions overcome particle motion, allowing particles to stay in close contact.
Why attractions are usually “invisible” at other conditions
At typical laboratory temperatures and low pressures, gas particles are:
Far apart most of the time (large average separation)
Moving quickly (higher kinetic energy), so brief encounters do not allow attractions to noticeably redirect motion
Near condensation, both of these trends shift:
Particles are closer together, so attractions act more frequently
Particle speeds are lower at lower temperature, so attractions have more time to alter trajectories
How attractions change measured pressure
Pressure in the ideal model vs real measurements
In the ideal model, pressure comes from particle collisions with container walls. When attractions are significant, particles approaching a wall can be pulled slightly back toward the bulk gas by neighboring particles. This reduces the momentum transferred to the wall.
The key consequence is:
Measured pressure is lower than ideal under condensation-favoring conditions (low , moderate-to-high )
This is exactly what the syllabus statement emphasizes: attractions affect measured pressure and volume most noticeably near condensation.
Particle-level interpretation
Attractions effectively:
Reduce the frequency of wall collisions (some particles turn back sooner)
Reduce the “strength” of wall collisions (lower impact speed at the wall)
How attractions change measured (effective) volume
Why volume can appear smaller than expected
Attractions encourage particles to stay closer together, increasing local clustering. Even if the container volume is fixed, attractive forces can make the gas behave as though it occupies less effective volume, because particles are not uniformly spreading out as the ideal model predicts.
In practice, this shows up as:
Greater compressibility than expected from ideal behavior (the sample can be compressed “more easily”) when attractions dominate
Linking back to the ideal gas law (as the reference point)
When describing deviations, the “ideal” baseline is still the ideal gas law.
= pressure of the gas (atm or kPa)
= volume of the gas (L)
= amount of gas (mol)
= ideal gas constant (e.g., )
= temperature (K)
A real gas near condensation can give a smaller measured than predicts at the same , , and , because attractive forces partially counteract the outward “push” associated with particle motion.
Conditions and gas identity that intensify attraction-driven deviation
Temperature and pressure trends near condensation
Attraction-driven deviations are most noticeable when:
Temperature is low (reduced particle kinetic energy)
Pressure is high enough to bring particles closer together (increased encounter frequency)
The system is approaching the boundary where a liquid becomes stable (condensation becomes favorable)
Which gases deviate more (at the same conditions)
The strength of attractions depends on the substance. Gases with stronger intermolecular attractions will show attraction-driven deviations farther from “ideal” behavior, especially as they near condensation. Qualitatively, larger and more easily polarised particles tend to attract more strongly, making non-ideal effects more pronounced under the same and .
FAQ
The critical temperature is the highest temperature at which a gas can be liquefied by pressure alone.
Above it, “close to condensation” is not attainable by compression, so attraction-driven deviations are typically less dramatic.
Attractions scale strongly with distance.
When compression or cooling reduces average separation past a threshold, many more encounters occur within the range where attractions are significant, causing a rapid increase in non-ideal behaviour.
Often, but not always.
Polarity increases attractions, yet size and polarisability also matter; some large nonpolar gases can deviate strongly because their electron clouds are easily distorted.
You would compare measured $P$ to the ideal prediction at the same $V$, $n$, and $T$.
If measured $P$ is systematically lower than ideal under cooling/compression trends toward liquefaction, that pattern is consistent with attraction-dominated deviation.
A particle heading toward the wall can be pulled back by neighbouring particles.
That reduces the particle’s speed at impact (or prevents impact), lowering momentum transfer per unit time and therefore lowering measured pressure.
Practice Questions
(2 marks) Explain why a real gas can exert a lower pressure than predicted by when the gas is close to condensation.
Mentions interparticle attractions become significant near condensation. (1)
Explains attractions reduce momentum transfer to container walls / reduce effective wall-collision impact, so measured is lower. (1)
(5 marks) A student states: “Real gases deviate from the ideal gas law most strongly near condensation because attractions affect both pressure and volume.” Assess this statement using particle-level reasoning.
Identifies near-condensation conditions involve lower and/or higher leading to closer particles and longer interaction times. (1)
Explains attractions pull particles together, promoting clustering/less uniform spreading (effective volume decreases / greater compressibility). (1)
Explains attractions reduce wall-collision momentum transfer, lowering measured pressure compared with ideal. (1)
Connects deviations explicitly to comparison with predictions (ideal baseline). (1)
Notes gases with stronger attractions show larger deviations under comparable conditions. (1)

