AP Syllabus focus: ‘Graphical representations of relationships among P, V, T, and n are useful for describing and predicting ideal gas behavior under changing conditions.’
Graphs make the ideal gas law visual. By holding two variables constant and plotting the other two, you can recognise curve shapes, interpret slopes, and predict how changing conditions shifts a gas’s measured pressure or volume.
Core idea: graphs come from the ideal gas law
Most AP Chemistry gas graphs are “slices” of ideal-gas behaviour where two state variables are held constant. The graph’s shape communicates whether the relationship is direct (linear) or inverse (hyperbolic), and whether a straight-line replot is possible (e.g., using ).
Isotherm: a curve or line showing how a gas variable changes at constant temperature.
On any gas graph, read the prompt carefully for what is constant: typical constraints are “at constant and ” or “at constant .”
= pressure (atm or kPa)
= volume (L)
= amount of gas (mol)
= temperature (K)
= ideal gas constant (value depends on units)
This equation is the source of the proportionalities used to interpret each graph.
P–V graphs (constant T and n)
Shape and interpretation
With constant and , rearrange to , so pressure is inversely proportional to volume.
A plot of vs. is a downward-curving hyperbola.
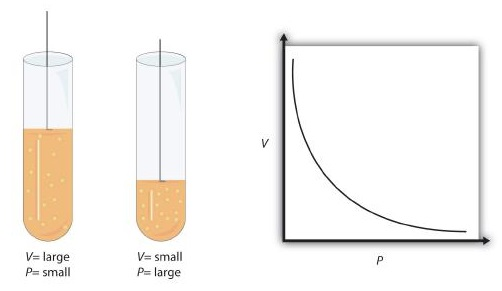
Boyle’s law graphical representation: pressure decreases nonlinearly as volume increases for a fixed amount of gas at constant temperature. The curve visually encodes the inverse proportionality , which is why equal fractional changes in do not produce equal absolute changes in . Source
The curve gets steeper at small because changing strongly affects collision frequency with container walls.
Linearising the relationship
AP questions often use a straight-line version to test proportional reasoning.
Plotting vs. gives a straight line through the origin for ideal behaviour.
The line’s steepness increases when or is larger (because is larger).
Families of curves
Multiple isotherms on the same axes show:
Higher isotherm: lies above a lower curve (at the same , higher gives higher ).
V–T graphs (constant P and n)
Why Kelvin matters
With constant and , , so volume is directly proportional to absolute temperature.
A plot of vs. (in K) is a straight line.
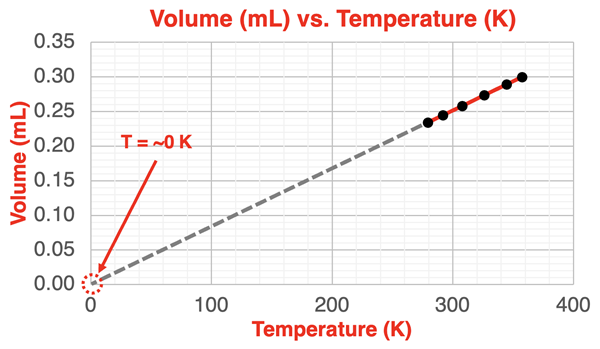
A volume–temperature plot illustrating Charles’s law at constant pressure and moles: the data fall on a straight line when temperature is measured in kelvins. The line extrapolates to zero volume at 0 K, highlighting why absolute temperature is required for a direct proportionality . Source
Using Celsius would shift the intercept and hide the proportionality; AP expects Kelvin for direct proportional relationships.
Slope meaning
For vs. at constant :
Larger makes the slope larger (more moles expand more per kelvin).
Larger makes the slope smaller (pressure resists expansion).
P–T graphs (constant V and n)
With constant and , , so pressure is directly proportional to absolute temperature.
A plot of vs. (K) is linear.
Increasing increases the slope; increasing decreases the slope.
Graphs involving n (amount of gas)
When and are constant, so:
vs. is linear (doubling moles doubles volume).
When and are constant, so:
vs. is linear (more moles means more wall collisions and higher pressure).
How to use these graphs to predict changes
Graphical representations help you connect a change in conditions to a shift in a line or curve:
Direct relationships (–, –, –, –): look for straight lines and compare slopes.
Inverse relationships (–): look for curvature or use a axis to compare lines.
FAQ
Because it converts the inverse relationship $P \propto 1/V$ (at constant $n,T$) into a straight line, making trend comparison and extrapolation visually simpler.
Look for “families” of lines/curves labelled by a fixed variable (e.g., isotherms), or infer from the shape: hyperbola suggests $P$–$V$ at constant $T,n$.
It represents a constant factor such as $\dfrac{nR}{P}$ (for $V$ vs $T$) or $\dfrac{nR}{V}$ (for $P$ vs $T$), linking particle amount and constraint conditions.
Kelvin is an absolute scale. Converting to °C adds an offset of 273.15, shifting the intercept and breaking the visual cue of direct proportionality.
At fixed $n$, a larger slope indicates larger $T$. At fixed $T$, a larger slope indicates larger $n$.
Practice Questions
A sample of ideal gas is kept at constant and . Describe the expected shape of a graph of against and state how the graph changes when is increased.
1 mark: vs is an inverse relationship (downward-curving hyperbola).
1 mark: Higher shifts the curve upwards (greater at the same ).
Two separate ideal gas samples A and B are plotted on a graph of vs (in K) at the same constant pressure. Line A is steeper than line B.
(i) State what this implies about compared with . (1 mark)
(ii) Explain, using the ideal gas law, why the lines are straight and pass through the origin. (3 marks)
(iii) State one consequence of plotting in °C instead of K for interpreting proportionality. (1 mark)
(i) 1 mark: .
(ii) 1 mark: From , at constant , .
(ii) 1 mark: Therefore giving a straight line.
(ii) 1 mark: Intercept at origin when is in K (zero K gives zero volume by ideal model).
(iii) 1 mark: °C shifts the intercept (not through origin), obscuring direct proportionality.

