AP Syllabus focus: ‘In mixtures of ideal gases, each gas exerts a partial pressure independent of others; partial pressure is proportional to mole fraction, and total pressure equals the sum of partial pressures.’
Gas mixtures are common in labs and the atmosphere. Dalton’s law provides a simple way to connect the total pressure of an ideal-gas mixture to each component’s contribution using mole fraction.
Dalton’s Law of Partial Pressures
In an ideal gas mixture, gas particles are assumed to have negligible volume and negligible intermolecular attractions, so each gas behaves as if it were alone in the container. As a result, each component contributes independently to the measured pressure.
Key terms
Partial pressure (): the pressure a single gas component in a mixture would exert if it alone occupied the entire container at the same temperature.
Partial pressure is a contribution to the total pressure, not a separate pressure in a separate space.
Mole fraction (): the fraction of total moles in a mixture contributed by component , (unitless).
Because mole fraction is a ratio of moles, it has no units, and the mole fractions of all components add to 1.
Dalton’s law relationships
Dalton’s law states that the total pressure is the sum of the partial pressures of each gas in the mixture, and each partial pressure is proportional to that gas’s mole fraction.
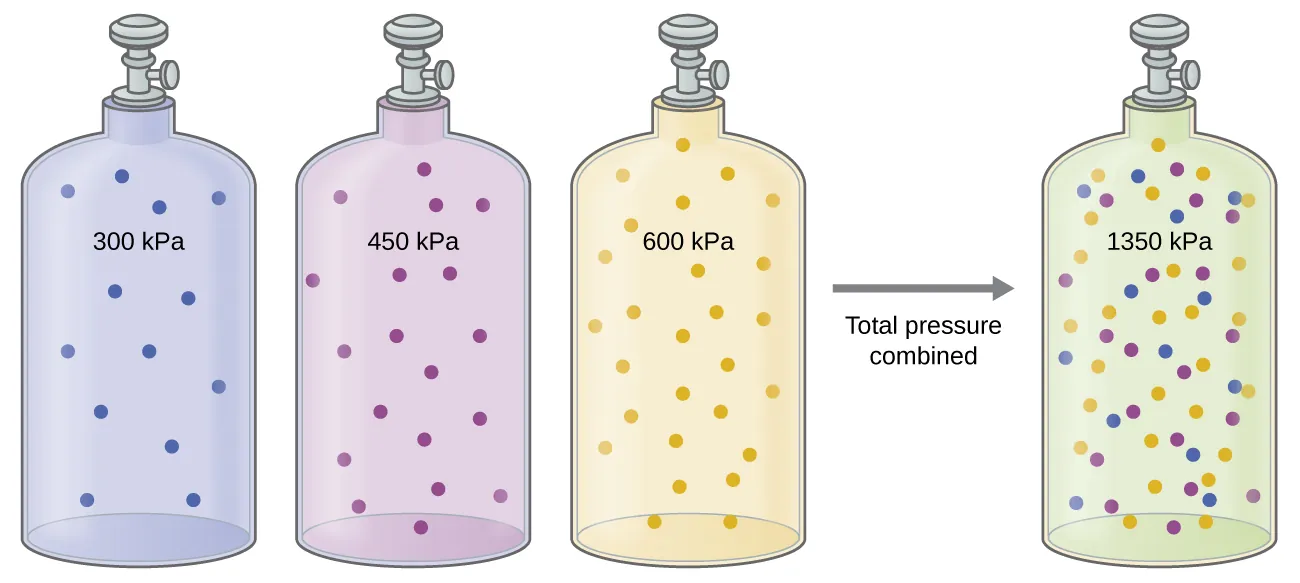
Separate equal-volume gas samples each exert their own pressure, and when combined into one container the measured pressure becomes the sum of the individual contributions. This visual reinforces the core statement of Dalton’s law: for an ideal-gas mixture. The particle-color coding helps connect “which particles are present” to “which part of the total pressure they contribute.” Source
= total pressure of the gas mixture (atm, kPa, mmHg, etc.)
= partial pressure of component (same units as )
= mole fraction of component (unitless)
These equations are most reliable when gases are close to ideal behavior (moderate pressures, sufficiently high temperatures).
Connecting Mole Fraction to Pressure
Why partial pressure is proportional to mole fraction (ideal gases)
At fixed temperature and volume, the ideal gas law implies .
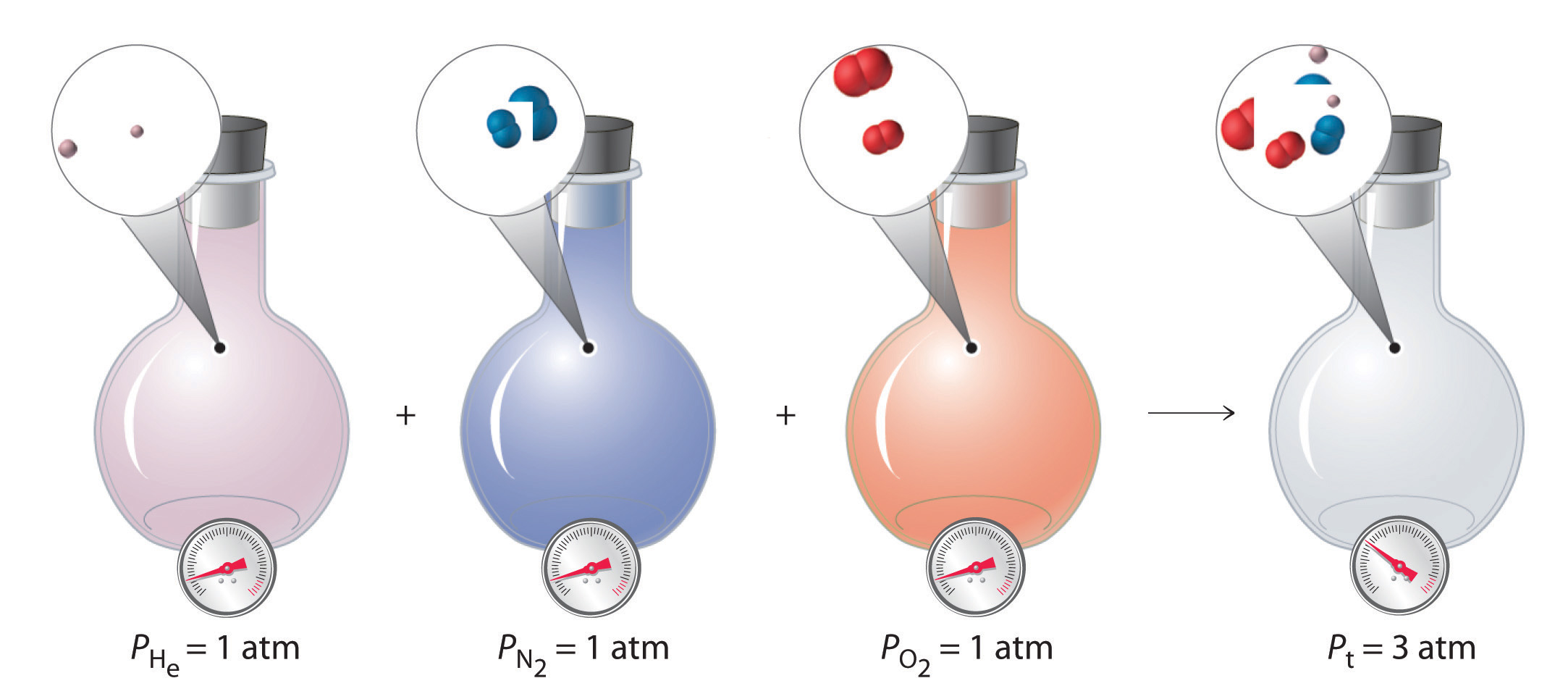
This resource connects the ideal gas law to mixture behavior by showing that, at constant and , each component’s pressure depends only on its amount in moles. It directly supports the mole-fraction form by relating ratios of partial pressure and total pressure to ratios of moles. Use it as the conceptual bridge between the definitions of mole fraction and partial pressure. Source
In a mixture:
All gases share the same container volume and temperature.
Each component’s contribution to pressure depends on how many moles of that component are present.
Therefore, a component with twice as many moles contributes twice the pressure, giving and thus .
Practical interpretation in mixtures
Dalton’s law is especially useful for reasoning about:
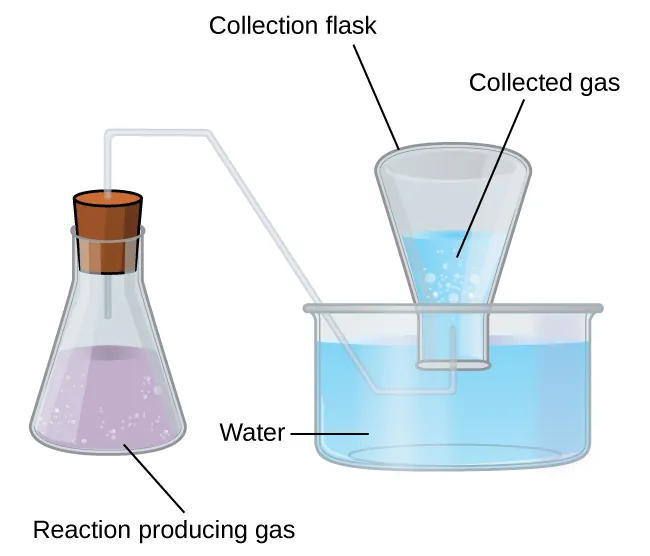
A gas collected over water is not pure: the trapped gas space contains both the produced gas and water vapor, so the measured pressure is a sum of partial pressures. The diagram highlights the experimental condition (equal water levels) used to ensure the trapped-gas pressure equals atmospheric pressure. This setup motivates the common correction in real lab calculations. Source
Gas collection over a mixture: the measured pressure reflects contributions from all gaseous species present.
Changing composition: if is fixed and one component’s mole fraction increases, its partial pressure increases proportionally while others decrease (since ).
Unit consistency: any pressure unit works as long as all pressures use the same unit.
Common pitfalls to avoid
Mole fraction vs percent: convert percent composition to a fraction before using .
Forgetting “total moles”: always uses for the entire mixture, not just selected components.
Assuming non-ideality doesn’t matter: Dalton’s law is an ideal model; large deviations can occur for strongly interacting gases or at high pressures.
FAQ
Only if the gases remain as the same chemical species while mixed.
If a reaction occurs, the composition (and therefore $X_i$ and $P_i$) changes with time, so you must account for reaction progress before applying $P_i=X_iP_\text{total}$.
For ideal gases at the same $T$ and $P$, volume fraction equals mole fraction, but mole fraction is more general.
It stays valid even when you compute composition from masses or chemical amounts, not measured volumes.
Convert each mass to moles using molar mass, then compute $X_i=\dfrac{n_i}{n_\text{total}}$.
$n_i=\dfrac{m_i}{M_i}$
Sum all $n_i$ to get $n_\text{total}$
Partial pressure ($P_i$) measures a gas’s contribution to total pressure; concentration measures amount per volume.
For ideal gases, they are proportional: $P_i=\left(\dfrac{n_i}{V}\right)RT$, so higher concentration implies higher partial pressure at fixed $T$.
At high pressures or low temperatures, gases deviate from ideality, so $P_i$ may not scale perfectly with $X_i$.
Strong intermolecular attractions or significant molecular volume can also cause systematic deviations from simple additivity.
Practice Questions
(2 marks) State Dalton’s law of partial pressures and write an expression relating to the partial pressures in a gas mixture.
States that the total pressure equals the sum of the partial pressures of the component gases (1)
Correct expression: or (1)
(5 marks) A mixture contains mol , mol , and mol at a total pressure of atm. Determine (i) the mole fraction of each gas and (ii) the partial pressure of each gas.
Calculates total moles: mol (1)
Correct mole fractions: , , (1)
Uses (1)
Correct partial pressure for : atm (1)
Correct partial pressures for and : atm each (1)

