AP Syllabus focus: ‘The kinetic molecular theory relates macroscopic gas properties to particle motion; the Maxwell–Boltzmann distribution describes particle kinetic energies at a given temperature.’
Gases appear simple at the macroscopic scale, yet their pressure, volume, and temperature emerge from countless microscopic particle collisions and motions. Kinetic molecular theory connects these levels and predicts characteristic energy distributions.
Kinetic Molecular Theory (KMT): Core Model
What KMT assumes about gas particles
Kinetic molecular theory (KMT): A model that explains macroscopic gas properties by treating a gas as a large number of tiny particles in constant random motion, with simplified rules for their motion and collisions.
In KMT, macroscopic measurements are understood as averages over many particles.
Key assumptions used in AP Chemistry reasoning:
Constant random motion: Gas particles move in straight lines between collisions and change direction when they collide.
Elastic collisions: Collisions between particles (and with container walls) conserve total kinetic energy of the colliding objects.
Negligible particle volume (idealised): Individual particles are treated as having no significant volume compared with the container.
No significant attractions/repulsions (idealised): Particle interactions are ignored except during collisions.
Large numbers: Because so many particles are present, gas properties are stable averages even though individual particles behave unpredictably.
How particle motion produces macroscopic gas behaviour
KMT directly links observable properties to particle-level events:
Pressure: Arises from particle collisions with container walls.
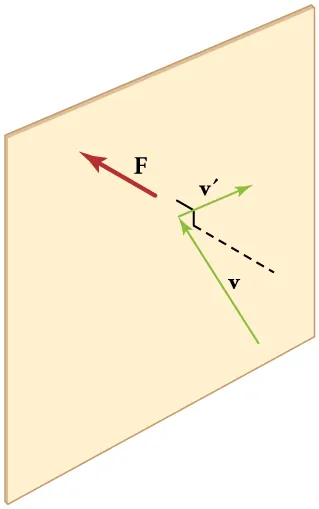
An elastic collision between a gas particle and a container wall reverses the particle’s velocity component perpendicular to the wall, implying a momentum change. By Newton’s laws, that momentum change corresponds to a force on the wall; many such impacts per unit area produce the macroscopic pressure you measure. The labeled and vectors emphasize that direction changes during the collision while (for an idealized elastic collision) the speed can remain the same. Source
More frequent or more forceful collisions produce higher pressure.
Temperature: Reflects how energetically particles are moving on average.
Heating a gas increases typical particle speeds, changing collision frequency and impact strength.
Volume (container size): Controls how often particles reach the walls.
In a larger volume (at fixed amount and temperature), collisions with walls are less frequent, lowering pressure.
Maxwell–Boltzmann Distribution: Energies in a Gas
What the distribution represents
Maxwell–Boltzmann distribution: A curve showing the spread of particle kinetic energies in a gas sample at a particular temperature, indicating how many particles have low, medium, or high kinetic energies.
A crucial idea is that not all particles have the same kinetic energy at a given temperature, even though the sample has one measured temperature.
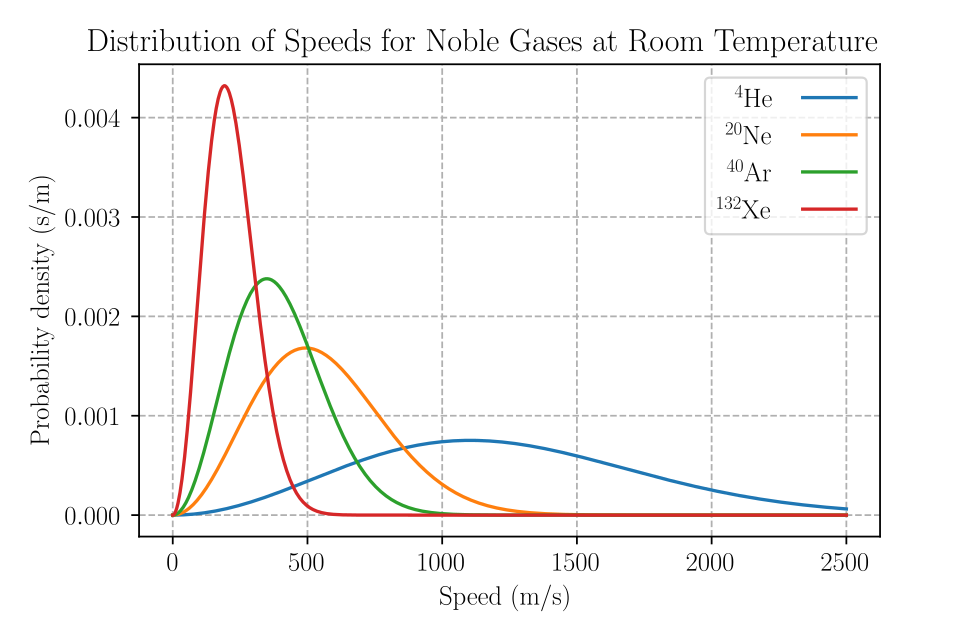
This plot compares Maxwell–Boltzmann speed distributions for different gases at the same temperature, showing distinct peaks and spreads for each substance. Lighter gases have distributions shifted to higher speeds, while heavier gases peak at lower speeds, even though all samples share the same temperature. The multiple curves reinforce that “temperature” summarizes an ensemble distribution rather than assigning the same kinetic energy to every particle. Source
Essential qualitative features (AP-level)
At a fixed temperature:
Most particles cluster near a most probable kinetic energy.
Some particles have very low kinetic energy.
A small fraction have very high kinetic energy (a “tail” to high energies).
KMT uses this distribution to connect microscopic variability to macroscopic consistency:
Macroscopic properties (like pressure) come from the combined effect of many particles with a range of energies.
Changes in conditions (especially temperature) change the overall distribution of kinetic energies in the sample.
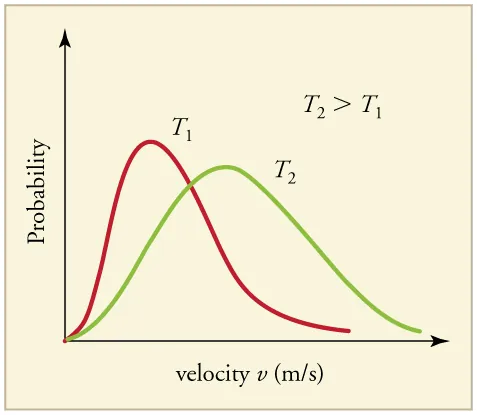
Two Maxwell–Boltzmann speed distributions are plotted for , showing how increasing temperature shifts the distribution to higher speeds and broadens it. The lower peak at higher temperature reflects that particles are spread over a wider range of speeds rather than clustered tightly near one most probable value. This visual also highlights the larger high-speed “tail” at higher temperature, which matters for collision frequency and energetic collisions. Source
Linking the Syllabus Statement to What You Observe
“The kinetic molecular theory relates macroscopic gas properties to particle motion” means you should be able to:
Explain pressure changes using collision frequency and collision strength.
Describe temperature changes as changes in the typical kinetic energies present in the Maxwell–Boltzmann distribution.
Justify why gas behaviour can be predicted statistically: individual particles vary, but the distribution and averages are reliable for large samples.
FAQ
Kinetic energy cannot be negative, so the distribution is bounded at zero but unbounded at high energy. That constraint naturally produces a long tail towards higher energies.
Total kinetic energy of the colliding particles is conserved overall, though it can be redistributed between them. Individual particles can speed up or slow down after colliding.
Ignoring intermolecular forces becomes problematic because attractions alter trajectories and reduce the momentum reaching the walls, so pressure is no longer predicted well by collision arguments alone.
With an enormous number of particles, random fluctuations average out over time and space. The wall experiences a steady average force even though individual impacts vary.
Mainly the particle’s speed and direction relative to the wall and the momentum change on impact. More perpendicular, faster impacts produce a larger momentum change and greater force.
Practice Questions
Q1 (2 marks) Using kinetic molecular theory, state how gas pressure arises and give one particle-level reason pressure increases when a gas is heated at constant volume.
Pressure is due to collisions of gas particles with the container walls. (1)
On heating, particles on average move faster so collisions are more frequent and/or more forceful (greater momentum change), increasing pressure. (1)
Q2 (5 marks) A sealed rigid container holds a gas. Describe, using KMT and the Maxwell–Boltzmann distribution, how particle motion and energy explain (i) why the gas has a single measured temperature, yet (ii) particles do not all have the same kinetic energy, and (iii) how this connects to the measured pressure.
Temperature reflects an average measure of particle kinetic energy in the sample. (1)
Maxwell–Boltzmann distribution shows a spread of kinetic energies at one temperature. (1)
Most particles have intermediate energies; fewer have very low or very high energies (distribution with a high-energy tail). (1)
Pressure is caused by particle collisions with the walls. (1)
The measured pressure is the combined effect of many collisions from particles across the energy distribution (higher-energy particles contribute more forceful collisions). (1)

