AP Syllabus focus: ‘In large biomolecules or polymers, noncovalent interactions within or between molecules strongly influence shape, which in turn determines functionality and macroscopic properties.’
Large biomolecules and polymers do not rely only on covalent bonds for structure. Their 3D shapes arise from many weak, reversible attractions whose combined effect controls flexibility, strength, and how the material behaves.
Core idea: shape from many weak forces
Noncovalent interactions are individually weaker than covalent bonds, but they are numerous and can act cooperatively across a large structure. Because these interactions are reversible, shapes can be stable yet responsive to changes in environment.
Noncovalent interactions: Attractive forces that do not involve sharing electron pairs (non-bonding attractions) and can act within a molecule or between molecules.
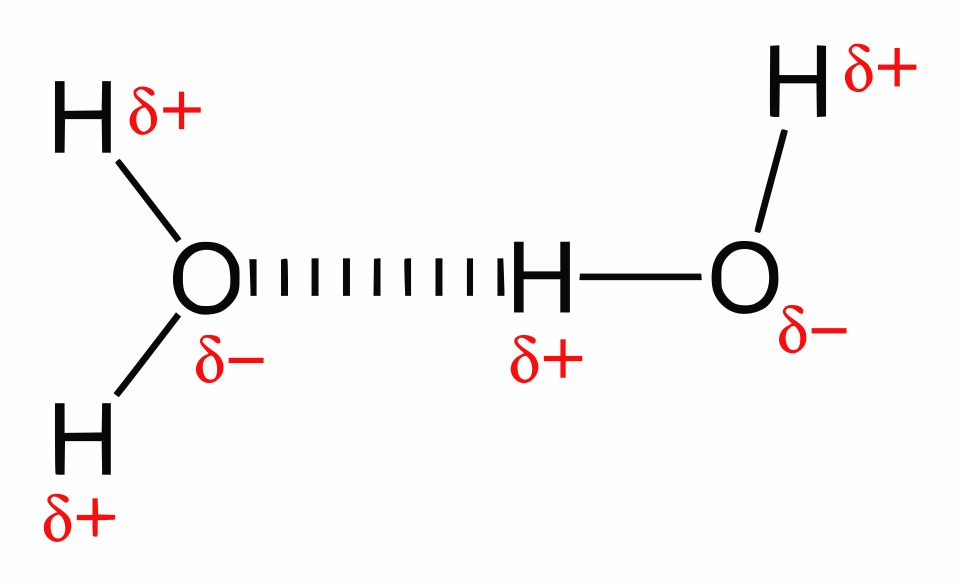
This diagram shows water molecules engaging in hydrogen bonding, typically depicted as dotted lines between an hydrogen and a lone-pair-bearing oxygen. It reinforces that hydrogen bonds are noncovalent (no electron-pair sharing) yet highly directional, which is why many of them together can strongly influence 3D structure. Source
In a polymer strand or a biomolecule, different regions can attract, repel, or pack together, giving a preferred conformation (shape) that minimises potential energy.
Types of noncovalent interactions that control shape
Within one large molecule (intramolecular)
Intramolecular attractions fold or coil a long chain into a compact, organised structure.
Hydrogen-bonding contacts between distant segments can “zip” parts of a chain together.
Dipole-based attractions align polar groups so opposite partial charges approach.
London dispersion attractions help closely packed nonpolar regions stabilise.
Electrostatic attractions/repulsions between charged sites can tighten folds (attraction) or force expansion (repulsion).
Steric effects (crowding) limit how closely segments can approach, shaping possible conformations.
Between molecules (intermolecular)
Intermolecular attractions link separate chains or molecules into larger assemblies.
Cross-linking by attractions increases rigidity and mechanical strength.
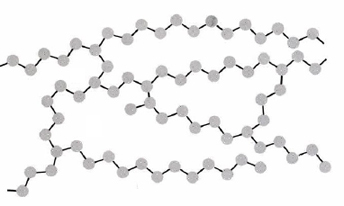
This schematic depicts polymer chains connected by cross-links, forming a network that restricts chain motion. The key structural idea (more connections between chains → less ability to slide → higher rigidity/strength) also applies to physical cross-links formed by strong noncovalent associations, even though this particular figure shows chemical (covalent) cross-linking. Source
Interchain alignment and packing can create ordered regions that resist deformation.
Aggregation can occur when many molecules present compatible interacting surfaces.
Cross-linking (physical): Association between polymer chains due to noncovalent attractions that connect chains without forming new covalent bonds.
Because cross-links are not covalent, temperature and solvent conditions can weaken them, changing macroscopic properties.
How shape connects to function and macroscopic properties
Biomolecules: function depends on precise shape
A biomolecule’s function often depends on having the right binding surfaces and flexibility.
Specific recognition: complementary shapes and charge distributions enable selective binding.
Stability vs flexibility: many weak interactions allow motion while maintaining overall structure.
Misfolding risk: if key noncovalent interactions are disrupted, the molecule may adopt a different shape with different (often reduced) function.
Polymers: bulk properties emerge from chain interactions
Polymer properties reflect how chains move, pack, and stick to one another.
Elasticity increases when chains can stretch and return due to reversible attractions.
Rigidity increases with stronger/more numerous interchain attractions and more physical cross-links.
Softening with heat: raising temperature increases molecular motion, weakening effective noncovalent attractions and allowing chains to slide more easily.
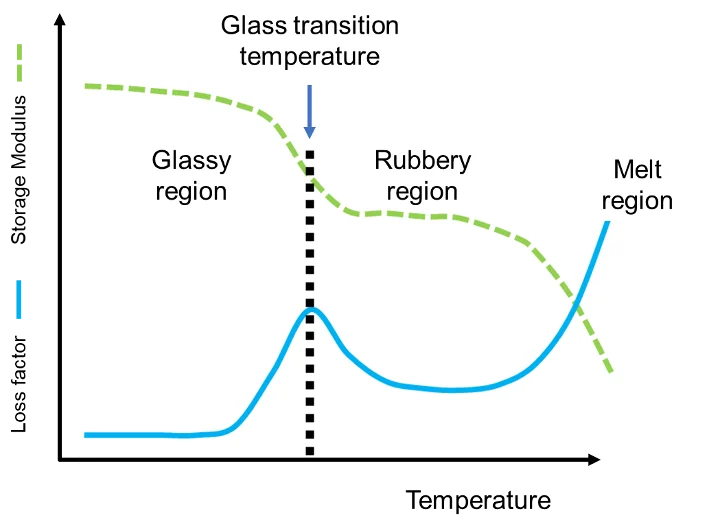
This graph illustrates how a polymer’s mechanical response changes with temperature: the storage modulus drops sharply through the glass-transition region while tan peaks, indicating increased energy dissipation. It connects microscopic mobility (chains moving more as rises) to macroscopic softening and viscoelastic behavior. Source
Solvent effects: if solvent–polymer attractions compete effectively with polymer–polymer attractions, chains separate and the material swells or dissolves; if not, the material remains intact.
Environmental factors that modulate noncovalent interactions
Small chemical or physical changes can shift the balance among many weak forces, changing shape and properties.
Temperature: higher increases motion and can reduce the net stabilising effect of weak attractions.
Solvent polarity: changes which groups are stabilised by interacting with solvent versus with each other.
pH (for ionisable groups): alters charge states, changing electrostatic attraction/repulsion patterns.
Concentration: higher concentration increases the likelihood of intermolecular association and aggregation.
FAQ
Specificity comes from many small matches acting together: shape complementarity plus aligned partial charges.
If even a few key contacts are missing, the total stabilisation drops enough that binding is much weaker.
Swelling: solvent penetrates and expands the polymer network, but enough interchain attraction remains to keep it as one piece.
Dissolving: solvent–polymer attractions dominate so chains separate and disperse as individual molecules.
Small differences that change packing or interchain attractions can matter greatly, such as:
more/less polar functional groups
ability to align closely (packing efficiency)
extent of physical cross-linking
pH can change whether certain groups are charged. That alters electrostatic attractions/repulsions within the molecule, shifting which conformations are most stable.
They continuously break and reform due to thermal motion.
This allows materials and biomolecules to be stable overall yet adaptable—responding to temperature, solvent, or concentration changes without permanent bond breaking.
Practice Questions
(2 marks) Explain why noncovalent interactions can strongly influence the shape of a large biomolecule even though each interaction is weak.
States that many noncovalent interactions act simultaneously across the molecule / cumulative effect (1)
Links cumulative stabilisation to a preferred 3D shape (lower energy conformation) and thus function (1)
(5 marks) A polymer material becomes softer when warmed and swells in a particular solvent. Using noncovalent interactions, explain both observations.
Warming increases particle motion, reducing the effective stabilising influence of noncovalent attractions (1)
Reduced interchain attractions allows chains to slide past one another more easily, lowering rigidity/softening (1)
Swelling explained by solvent molecules entering between chains (1)
Solvent–polymer attractions compete with and partially replace polymer–polymer attractions (1)
Resulting increased chain separation/volume increase described (1)

