AP Syllabus focus: ‘Metallic solids conduct heat and electricity due to mobile valence electrons and are malleable and ductile; interstitial alloys become more rigid but typically remain conducting.’
Metallic solids have distinctive properties—conductivity, malleability, and ductility—that come from a unique bonding model. Understanding the “sea of electrons” explains how metallic structure produces these macroscopic behaviours and how alloying modifies them.
Metallic bonding and structure
The “sea of electrons” model
In a metallic solid, atoms pack into a crystal lattice and their valence electrons are not localised to individual atoms. Instead, those electrons are shared across the entire solid, creating mobile charge carriers.
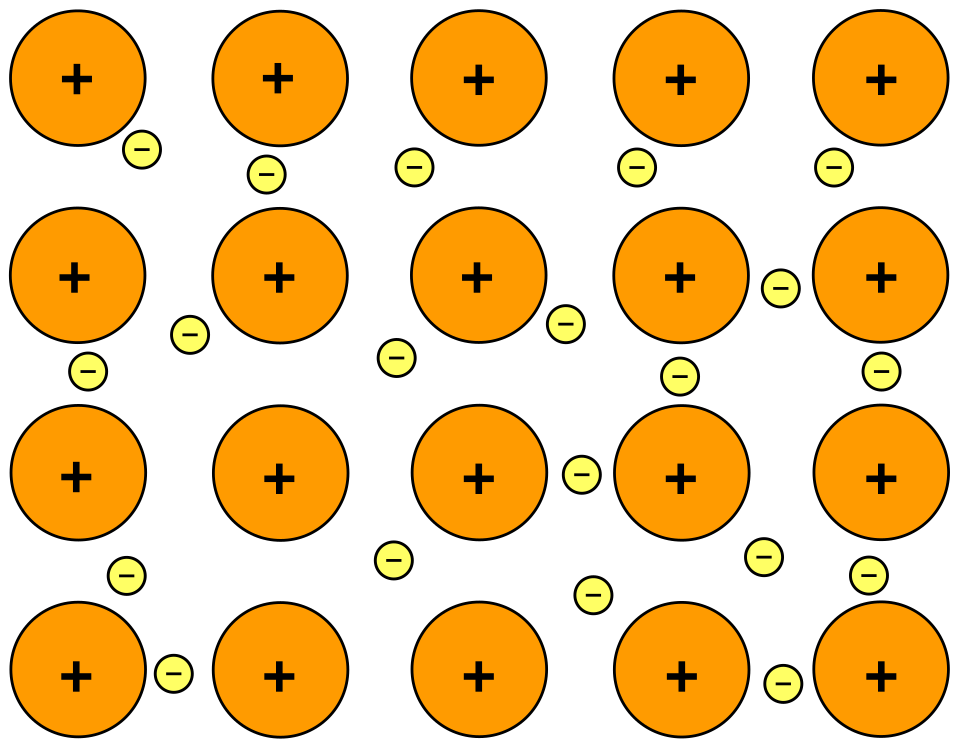
Schematic of metallic bonding showing fixed metal cations in a crystal lattice immersed in a delocalized electron “sea.” The diagram visually connects electron delocalization to both cohesion (electrostatic attraction) and the presence of mobile charge carriers. Source
Metallic bonding: electrostatic attraction between a lattice of metal cations and a delocalised “sea” of mobile valence electrons.
This model accounts for metals being solids with strong cohesion while still allowing internal movement of particles under stress.
What is actually in the lattice?
Positive metal ions (cations): often described as fixed at lattice points (they vibrate, but do not translate through the solid).
Delocalised electrons: move throughout the solid, balancing charge and holding the lattice together via Coulombic attraction.
Close packing: many metals adopt efficient packing arrangements, increasing the number of cation–electron attractions per unit volume.
Key properties explained by delocalised electrons
Electrical conductivity
Metals conduct electricity because electrons are mobile and can respond to an applied electric field.
When a potential difference is applied, electrons drift through the lattice.
Conductivity is retained because the lattice does not need to move for charge to flow; only the electron sea reorganises.
Thermal conductivity
Metals conduct heat well because energy is transferred efficiently by:
Mobile electrons carrying kinetic energy through the solid
Lattice vibrations (vibrational energy can be passed from ion to ion)
In many metals, electron mobility is the dominant reason heat spreads quickly.
Malleability and ductility
Metals are malleable (hammered into sheets) and ductile (drawn into wires) because metallic bonding is non-directional.
Under stress, layers of cations can slide past one another.
The electron sea continues to attract cations after shifting, so the structure does not shatter the way an ionic crystal might.
Other common metallic traits (linked to the same model)
Lustre: free electrons interact with light, producing reflective surfaces.
Variable melting points: depend on how strongly cations and electrons attract (influenced by charge density and number of delocalised electrons).
Alloys and why they change properties
What an alloy does to a metal
Pure metals often have regular layers that slide easily. Adding other atoms disrupts that regularity and changes mechanical properties.
Alloy: a solid containing a mixture of elements (at least one metal) whose particles share a metallic lattice, producing modified properties compared with the pure metal.
A key AP focus is the effect of interstitial alloys on rigidity while maintaining conductivity.
Interstitial alloys: increased rigidity, still conducting
Interstitial alloys form when small atoms fit into the gaps (interstices) between larger metal atoms in the lattice (for example, C in Fe).
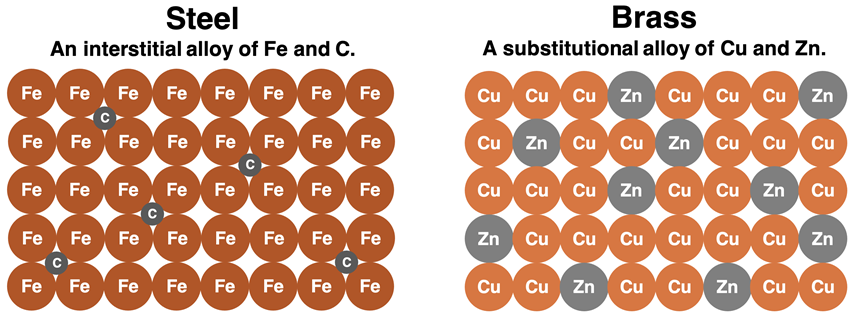
Particle-level comparison of an interstitial alloy (steel: small C atoms in the gaps of an Fe lattice) versus a substitutional alloy (brass: Zn atoms replacing some Cu atoms). The interstitial case emphasizes how occupying interstices distorts the lattice and inhibits layer motion, helping explain increased hardness while maintaining a metallic structure. Source
Small atoms in holes distort the lattice and hinder the movement of metal layers.
This reduces the ability of layers to slide, so the alloy becomes harder and more rigid.
Despite added atoms, these solids typically remain conducting because the metallic electron sea persists and electrons can still move through the structure.
Key idea to state clearly: alloying changes how easily the lattice can deform, but does not usually eliminate delocalised electrons.
FAQ
Corrosion resistance can improve if alloying promotes formation of a thin, adherent oxide layer that blocks further attack.
Examples include passivation behaviour where the surface film is stable and self-repairing.
As temperature rises, lattice vibrations increase, scattering electrons more frequently.
This usually increases resistance and decreases conductivity, even though electrons remain delocalised.
Reduced ductility can result from microstructural features that block layer motion, such as:
impurities
grain boundaries
work hardening (deformation increasing defect density)
These factors impede slip without changing the basic metallic bonding model.
Interstitial: small atoms occupy holes between metal atoms.
Substitutional: atoms of similar size replace host metal atoms on lattice sites.
Both disrupt regular packing, but the size-compatibility requirement differs.
Mercury’s bonding is weakened by its electronic structure and relativistic effects that stabilise certain orbitals, reducing cohesive strength.
This lowers its melting point enough that it is liquid near room temperature.
Practice Questions
(2 marks) Explain, using the “sea of electrons” model, why solid metals conduct electricity.
Mentions delocalised/mobile electrons in the metal (1)
Links electron mobility to charge flow under an applied potential difference/electric field (1)
(5 marks) A metal is converted into an interstitial alloy by adding a small non-metal atom. Explain how this change affects (i) rigidity and (ii) electrical conductivity, in terms of structure and bonding.
States small atoms occupy interstitial sites/gaps in the metal lattice (1)
Explains lattice distortion/hindered movement of layers or dislocations (1)
Links hindered movement to increased hardness/rigidity (1)
States alloy still has delocalised electrons/metallic bonding (1)
Links delocalised electrons to electrical conductivity being retained (typically) (1)

