AP Syllabus focus: ‘Covalent network solids form 3D networks or 2D layers from nonmetals/metalloids; strong covalent bonding leads to high melting points, with hardness or softness linked to structural rigidity or layer slippage.’
Covalent network solids are a major solid type in AP Chemistry because their atoms are connected by extensive covalent bonding. Their structures explain unusually high melting points and distinctive mechanical properties such as extreme hardness or easy cleavage.
What a covalent network solid is
Covalent network solids are solids in which atoms are linked by covalent bonds into a continuous framework rather than existing as discrete molecules.
Covalent network solid: A solid made of atoms covalently bonded in an extended lattice (2D or 3D), so breaking the solid requires breaking many covalent bonds.
These solids typically involve nonmetals or metalloids (for example, C, Si, and O in various compounds), consistent with covalent bonding being most common among such elements.
Key structural idea: 3D networks vs 2D layers
The syllabus emphasises that covalent network solids can be:
3D networks: covalent bonds extend in all directions through the crystal.
2D layered networks: strong covalent bonds exist within sheets, but sheets are held together by much weaker attractions.
This structural difference is central for explaining why some network solids are extremely hard while others are soft and slippery.
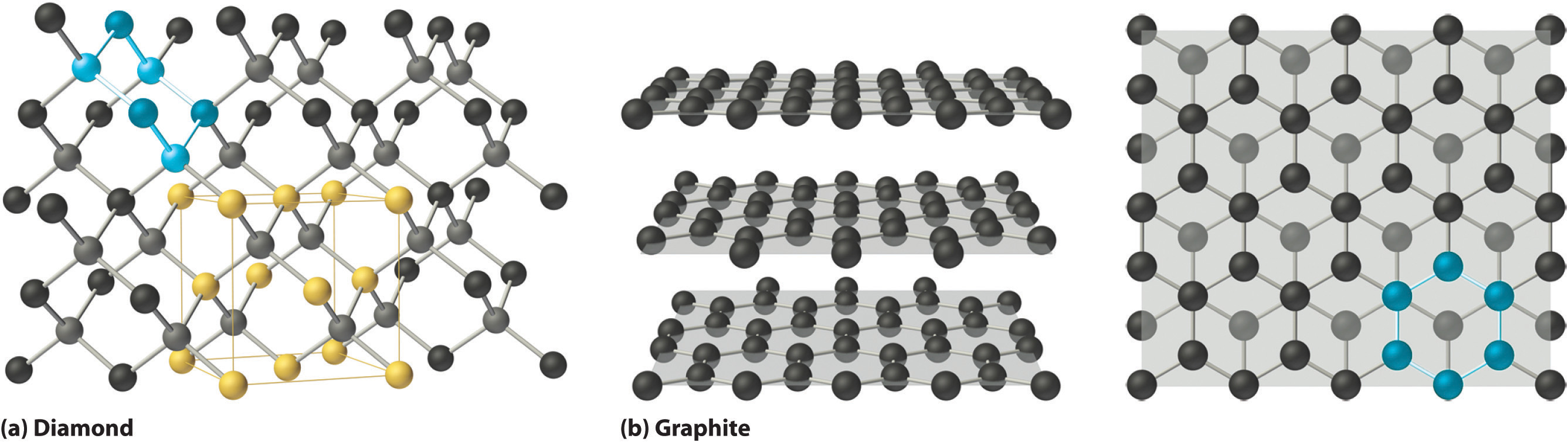
Side-by-side structural models of diamond and graphite. Diamond is shown as a 3D tetrahedral network (strong covalent bonds in all directions), while graphite is shown as stacked 2D sheets of fused hexagons with weaker interlayer attractions that allow layers to slide. The visual contrast supports structure→property reasoning for hardness versus lubricating softness. Source
Why melting points are so high
Strong covalent bonding leads to high melting points because melting requires disrupting the solid’s ordered structure. For a covalent network solid, that means breaking a large number of strong covalent bonds throughout the lattice, not just overcoming weak intermolecular forces.
Macroscopic implications students should connect to bonding:
Very high melting point (and typically high boiling point) compared with molecular solids.
Low volatility, because particles cannot easily escape the solid without extensive bond breaking.
Hardness often correlates with bonding rigidity, but depends on whether the solid is a 3D network or layered.
Diamond: a rigid 3D carbon network
Diamond is an allotrope of carbon where each carbon atom is covalently bonded to four others in a tetrahedral arrangement, creating a rigid 3D network.
Allotrope: One of two or more different structural forms of the same element in the same physical state.
This structure explains key properties that AP Chemistry expects you to reason from bonding:
Very high melting point: melting requires breaking covalent bonds throughout the 3D lattice.
Extreme hardness: the 3D framework resists deformation because there are no “planes of weakness” for atoms to slide past each other.
Electrical insulation (typical): all valence electrons are localised in bonds, so there are no mobile charge carriers in the ideal lattice.
Structural rigidity and hardness
The specification links hardness to structural rigidity. In diamond, rigidity comes from:
strong covalent bonds in all directions
a geometry that locks atoms into place across the entire crystal
Graphite: 2D carbon layers with easy slippage
Graphite is another carbon allotrope, but its atoms form 2D layers (sheets) of hexagonally arranged carbon atoms. Covalent bonds are strong within each layer, but layers are held together by relatively weak attractions, allowing them to slide.

Diagram of graphite showing extended hexagonal carbon sheets stacked into layers with larger spacing between sheets. It highlights that strong covalent bonding is confined within each layer, whereas the interlayer region is held together by much weaker forces. This structural feature explains graphite’s easy cleavage and lubricating behavior under shear. Source
This directly matches the syllabus statement that softness can be linked to layer slippage:
High melting point: within-layer covalent bonding is extensive, so large energy input is needed to disrupt the network.
Softness and lubricating behaviour: layers can slip past each other under stress because interlayer attractions are much weaker than covalent bonds.
Electrical conductivity along layers (common): each carbon forms three covalent bonds, leaving electrons that are delocalised within the sheet and can move under an applied potential.
Comparing diamond and graphite (structure → property)
Both are covalent network solids of carbon, so both have strong covalent bonding and high melting points, but:
Diamond: 3D covalent network → maximal rigidity → very hard
Graphite: 2D covalent layers + weak interlayer forces → layer slippage → soft
What to look for in exam prompts
When asked to justify a property, focus on the minimum structural claim needed:
If the prompt is about melting point, emphasise many strong covalent bonds must be broken.
If the prompt is about hardness vs softness, emphasise 3D rigidity (diamond) versus 2D layers that slide (graphite).
If the prompt mentions composition, connect “nonmetals/metalloids” to the likelihood of extensive covalent bonding in a lattice.
FAQ
Delocalised electrons are largely confined to the carbon sheets, so charge moves readily along layers but far less effectively between layers due to weak interlayer interactions.
No. Conductivity depends on electron availability. Diamond lacks mobile electrons, while graphite (and some other networks) can have delocalised electrons that act as charge carriers.
Imperfections disrupt the regular lattice. In diamond, defects can reduce ideal hardness by providing points where fracture initiates; in graphite, disorder can hinder smooth layer sliding.
The strong bonds are within layers, but the layers are separated by much weaker attractions. Under shear stress, layers move relative to each other, producing a lubricating effect.
Graphene is essentially a single graphite layer: a 2D covalently bonded network. Many of graphite’s properties arise from how these layers stack and interact.
Practice Questions
(2 marks) Explain why diamond has a much higher melting point than a typical molecular solid such as iodine.
Mentions diamond is a covalent network solid with an extended lattice (1)
States melting requires breaking many strong covalent bonds (not just weak intermolecular forces) (1)
(6 marks) Graphite is soft and conducts electricity, whereas diamond is extremely hard and does not conduct. Using bonding and structure, explain both differences.
Graphite consists of 2D layers with strong covalent bonding within each layer (1)
Layers are held together by weak forces so they can slide, making graphite soft (1)
Diamond has a 3D covalent network (1)
3D rigid lattice resists deformation, making diamond hard (1)
Graphite has delocalised/mobile electrons within layers that carry charge (1)
Diamond has no mobile electrons because electrons are localised in covalent bonds (1)

