AP Syllabus focus: ‘Use Planck’s equation E = hν to connect photon energy to frequency and perform calculations that relate photon properties to electronic transitions.’
Planck’s relationship links the microscopic energy of light to a measurable wave property. In AP Chemistry, it is used to quantify photon energies and connect them to specific electronic transitions in atoms and molecules.
Planck Relationship and Photon Energy
What the equation states
Light can be treated as packets of energy whose size depends on frequency, not on how bright the light is.
Photon: A discrete “packet” (quantum) of electromagnetic energy that can be absorbed or emitted by matter.
A higher-frequency photon carries more energy per photon.
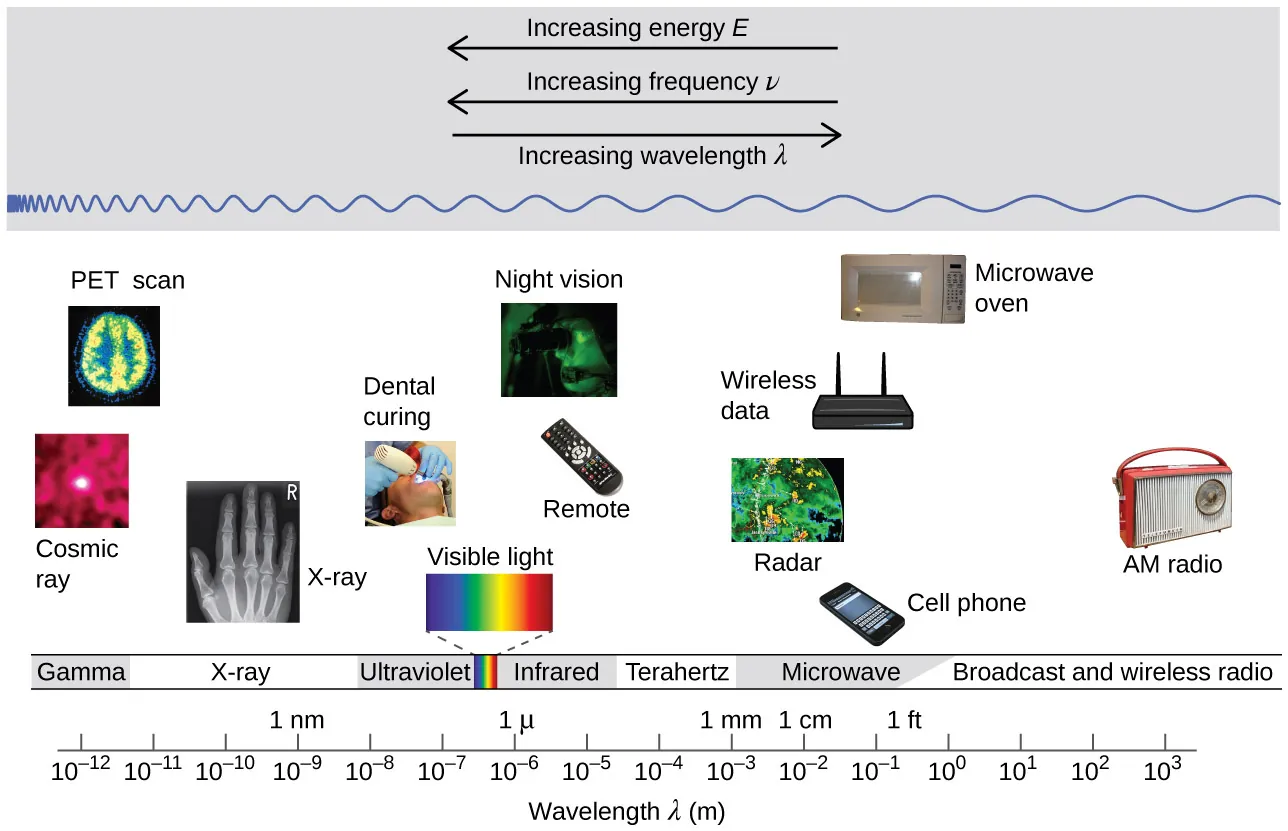
Electromagnetic spectrum diagram showing that frequency and photon energy increase together while wavelength decreases. The labeled arrows reinforce the proportionality in and the inverse relationship between and across radio → gamma radiation. Source
This is why higher-energy radiation can drive larger changes in electronic energy.
Planck constant (h): The proportionality constant that relates photon energy to frequency, setting the scale of quantized energy changes.
The Planck relationship is written as:
= energy of one photon (J per photon)
= Planck constant ()
= frequency (Hz, )
= energy per mole of photons (J·mol or kJ·mol)
= Avogadro constant ()
Interpreting the proportionality
Because is directly proportional to :
Doubling doubles .
If two photons have different frequencies, the one with the greater must correspond to the greater energy change in the absorbing/emitting species.
Using in AP Chemistry
Units to track (what AP expects you to manage)
Calculations are mainly about consistent units and clear interpretation of “per photon” vs “per mole.”
Frequency must be in Hz ().
comes out in joules per photon when is in J·s.
To report energies on a chemical (molar) scale, use and often convert J·mol to kJ·mol.
Connecting photon energy to electronic transitions
An electronic transition occurs when an electron moves between quantized energy levels.
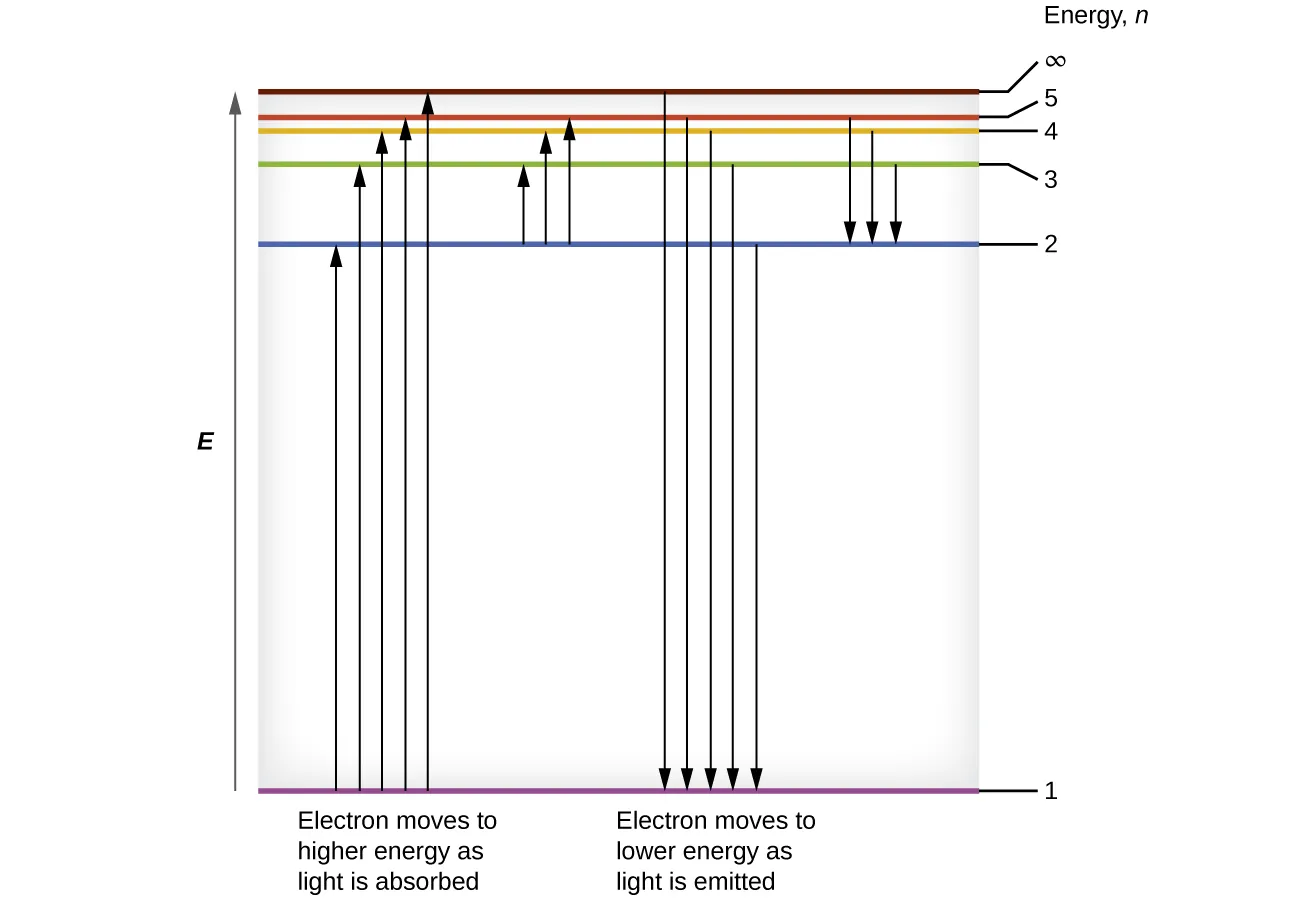
Bohr-model energy-level diagram for hydrogen with discrete levels labeled by and transition arrows. Upward arrows indicate absorption to higher-energy states, while downward arrows indicate emission as electrons drop to lower-energy levels, emphasizing that only specific values (and thus specific ) are allowed. Source
The photon involved must match the energy gap.
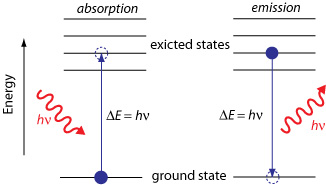
Energy-level schematic comparing absorption and emission, with the energy gap labeled as . The upward transition represents photon absorption into an excited state, while the downward transition represents photon emission as the system returns toward the ground state. Source
Absorption: an atom/molecule takes in a photon; the photon energy corresponds to the increase in electronic energy.
Emission: an excited species releases a photon; the photon energy corresponds to the decrease in electronic energy.
In both cases, provides the bridge between:
a measured or given frequency of radiation, and
the energy change associated with the transition.
Calculation workflow (without doing the arithmetic)
Use a consistent procedure to avoid common AP errors:
Identify what is requested: energy per photon () or energy per mole of photons ().
Convert frequency to Hz if needed (ensure ).
Apply to get joules per photon.
If a molar quantity is needed, multiply by to scale from photon-level to mole-level energy.
Apply unit conversions at the end (for example, J to kJ) while keeping significant figures appropriate to the given data.
FAQ
$E=h\nu$ gives the energy of each photon.
Intensity is mainly related to the number of photons arriving per second (photon flux), not the energy carried by each individual photon.
Only specific electron energies are allowed, so changes in energy happen in discrete jumps.
Therefore, only photons with energies matching allowed gaps can be absorbed or emitted.
Use per photon when discussing a single transition event.
Use per mole of photons when comparing to macroscopic chemical quantities (e.g., kJ·mol$^{-1}$) or when linking to amounts measured in moles.
Because $h$ is tiny, photon energies at everyday frequencies can be very small, making quantisation hard to notice macroscopically.
At high frequencies, the energies become large enough that quantum effects (like electronic transitions) are prominent.
Yes (for example, J·s is standard; other unit systems exist).
Be careful that changing energy units (J to eV, for instance) requires a consistent value of $h$ in matching units, or a conversion after calculating $E$ in joules.
Practice Questions
(2 marks) Calculate the energy of a single photon with frequency using .
Uses correctly. (1)
Correct substitution and value with unit J (allow 3 s.f.). (1)
(5 marks) A substance absorbs light of frequency , promoting an electron to a higher energy level. (a) Calculate the energy absorbed per photon. (2 marks) (b) Calculate the energy absorbed per mole of photons in kJ·mol. Use . (3 marks)
(a) States/uses . (1)
(a) Correct numerical result with unit J per photon. (1)
(b) Uses or . (1)
(b) Correct numerical result in J·mol. (1)
(b) Converts to kJ·mol correctly and includes unit. (1)

