AP Syllabus focus: ‘When a photon is absorbed or emitted by an atom or molecule, the species’ energy increases or decreases by an amount equal to the photon’s energy.’
Energy changes during light–matter interactions are quantized. In AP Chemistry, you must connect absorption and emission of a photon to discrete changes in an atom’s or molecule’s energy.
Core idea: photon exchange changes particle energy
When an atom or molecule interacts with electromagnetic radiation, it can exchange energy only in packets.
Photon: A discrete packet (quantum) of electromagnetic energy that can be absorbed or emitted by matter.
The syllabus statement means:
If a species absorbs a photon, the species gains exactly that amount of energy.
If a species emits a photon, the species loses exactly that amount of energy.
No “partial photons” are exchanged; the energy change is all-or-nothing for a single event.
Energy levels and allowed changes
Atoms and molecules have discrete energy states, so absorption or emission corresponds to a transition between two allowed states.
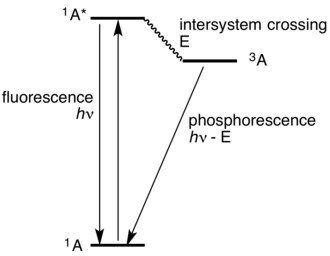
A Jablonski diagram organizes molecular energy states vertically (higher energy up) and uses arrows to show transitions. Upward arrows represent photon absorption (population of an excited state), while downward arrows represent radiative emission processes such as fluorescence or phosphorescence. The key AP takeaway is that each transition corresponds to a specific, quantized energy gap between levels. Source
Energy level (energy state): A specific, quantized energy value an atom or molecule can have, with transitions occurring only between allowed states.
A transition must satisfy energy conservation:
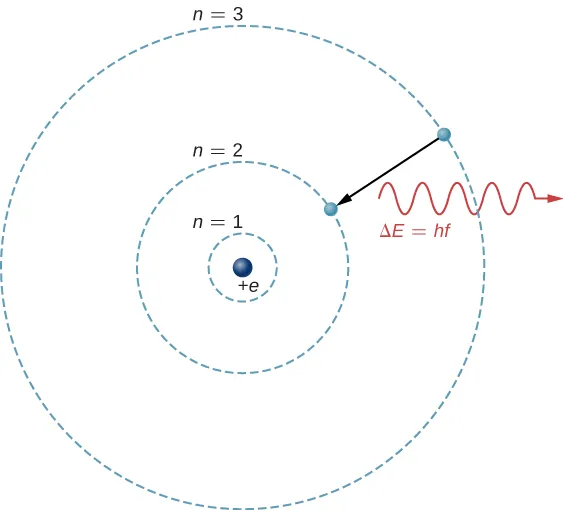
Bohr-style schematic showing an electron dropping from a higher shell to a lower shell while emitting a photon whose energy equals the level spacing. The wavy line labeled emphasizes that photon energy is quantized and exactly matches the energy difference between the two states. The same energy gap must be supplied (via absorption) to drive the reverse, upward transition. Source
The photon’s energy matches the difference between the two energy levels involved.
This is why spectra often show distinct lines or bands rather than a continuum (each line/band corresponds to a specific energy gap).

Simulated visible hydrogen emission spectrum showing discrete bright lines at specific wavelengths rather than a continuous band. Each line corresponds to a particular electronic transition whose photon energy equals the energy gap between two allowed states. This is a direct visual consequence of quantized energy levels producing only specific photon energies (and thus specific wavelengths). Source
Absorption: energy increases by the photon’s energy
During absorption, a photon is removed from the radiation field and its energy becomes internal energy of the species.
The species moves from a lower-energy state to a higher-energy state.
The energy increase of the species equals the photon energy.
Sign convention for absorption
Chemists often describe the species’ energy change as:
Positive for absorption (energy in)
Emission: energy decreases by the photon’s energy
During emission, the species releases energy as a photon.
The species moves from a higher-energy state to a lower-energy state.
The energy decrease of the species equals the photon energy.
Sign convention for emission
Chemists often describe the species’ energy change as:
Negative for emission (energy out)
Expressing the relationship mathematically
You should be able to write the relationship between a species’ energy change and the photon energy, including the correct sign.
= Change in energy of the atom or molecule (J per particle)
= Energy carried by the absorbed photon (J per photon)
= Change in energy of the atom or molecule (J per particle)
= Energy carried by the emitted photon (J per photon)
In words:
Absorption: species energy increases by the photon’s energy.
Emission: species energy decreases by the photon’s energy.
What you should be able to state from a transition description
Given a description of two energy states (initial and final), you should be able to:
Identify whether the process is absorption (upward transition) or emission (downward transition)
Relate the photon energy to the energy gap between the states
Use consistent language linking microscopic events to energy change:
“The atom/molecule gains energy equal to the photon energy” (absorption)
“The atom/molecule loses energy equal to the photon energy” (emission)
Common pitfalls to avoid
Confusing the sign: the photon’s energy is always positive, but the species’ can be positive (absorption) or negative (emission).
Saying “energy is created” during emission: emission is a conversion of the species’ internal energy into photon energy.
Treating energy levels as continuous: AP expects quantized energy changes tied to specific transitions.
FAQ
No. The excited state can persist for a finite lifetime.
Competing pathways include radiative emission, non-radiative relaxation (energy transferred to motion/heat), or transfer to another species.
Spontaneous emission occurs without an external trigger.
Stimulated emission occurs when incident radiation induces emission of a photon matching the triggering photon’s energy (and typically direction/phase).
Energy states are quantised, so only certain energies are allowed.
A photon exchange must exactly match an allowed energy gap; otherwise the interaction does not produce a lasting transition.
Molecules also have quantised states (electronic, vibrational, rotational).
Absorption/emission still changes molecular energy by exactly the photon energy, but there are more possible transitions than in many atoms.
Microscopic events use joules per photon (per particle event).
Laboratory-scale reporting often uses joules per mole of photons (or per mole of molecules), converting with Avogadro’s constant.
Practice Questions
(1–3 marks) An atom absorbs a photon and transitions from a lower to a higher energy level. State the sign of and its relationship to .
is positive for absorption. (1)
stated clearly. (1)
(4–6 marks) An atom has two energy levels, (lower) and (higher).
(a) Write an expression for the photon energy absorbed when the atom transitions from to .
(b) Write an expression for for this absorption.
(c) For the reverse transition from to , state the sign of and relate it to the emitted photon energy.
(a) . (1)
(b) or . (2)
(c) Emission: is negative. (1)
(c) with implied or stated. (1)

