AP Syllabus focus: ‘Relate wavelength and frequency using the wave equation c = λν, where c is the speed of light and λ and ν describe the photon’s electromagnetic wave.’
Electromagnetic radiation behaves as a wave characterised by its wavelength and frequency. AP Chemistry frequently asks you to connect these quantities quantitatively and qualitatively using the constant speed of light in vacuum.
The wave relationship for light
What the equation states
= speed of light in vacuum,
= wavelength, in (often reported in )
= frequency, in (Hz)
This relationship links two ways of describing the same electromagnetic wave: wavelength (a distance per cycle) and frequency (cycles per second).
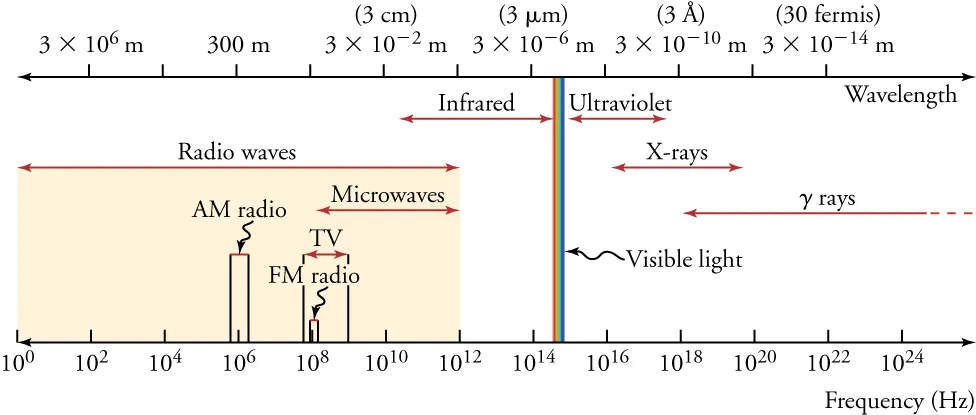
Electromagnetic spectrum chart showing the major EM categories (radio through gamma) alongside aligned wavelength and frequency scales. It visually emphasizes that moving toward shorter wavelength corresponds to higher frequency, consistent with . Source
Key terms you must use correctly
Wavelength (): The distance between identical points on successive wave cycles (e.g., crest to crest), measured in metres.
Wavelength is a spatial measure, so unit conversion is often the main challenge in AP Chemistry contexts.
Frequency (): The number of wave cycles that pass a point each second, measured in (hertz, Hz).
Frequency is a rate; in the wave equation it tells you how many cycles occur per second as the wave propagates.
How to rearrange and interpret
You should be able to isolate any variable without memorising separate formulas:
(wavelength from frequency)
(frequency from wavelength)
The proportionalities are essential:
At constant , frequency and wavelength are inversely proportional.
Increasing forces to decrease so that the product remains equal to .
Units and dimensional reasoning
Common units on the AP Chemistry exam
Although is given in , wavelengths are commonly stated using metric prefixes:
Frequencies may appear as:
Scientific notation for very large values (e.g., )
Dimensional check (a fast error-catcher)
Use units to verify your setup:
has units , matching the units of .
If your final unit is not metres for or for , a conversion step is missing.
Physical meaning in AP Chemistry contexts
Why “” is used
In AP Chemistry, the equation is typically applied to photons as electromagnetic waves in vacuum (or air approximated as vacuum), so the wave speed is taken as the constant . This lets you connect a radiation’s reported wavelength (often in nm) to its corresponding frequency (in Hz) using one direct relationship.
Language that earns credit
When describing the relationship (without doing a calculation), use precise statements:
“For electromagnetic radiation, shorter wavelength means higher frequency because is constant.”
“If the wavelength doubles, the frequency halves (inverse proportionality).”
“Wavelength is a distance between repeating points; frequency is cycles per second.”
Common pitfalls to avoid
Mixing up symbols: (nu) is frequency, not “v” for velocity.
Forgetting to convert nm or m to metres before using .
Treating as variable in typical AP problems involving light; unless told otherwise, use as a constant.
Rounding too early; keep significant figures consistent with given data.
FAQ
$c$ is the speed in vacuum. In a medium, the wave’s speed is $v=\dfrac{c}{n}$, where $n$ is the refractive index.
Frequency stays the same when entering a new medium; wavelength changes to match $v=\lambda \nu$.
Wavenumber is $\tilde{\nu}=\dfrac{1}{\lambda}$, commonly reported in $\text{cm}^{-1}$ in infrared spectroscopy.
It increases as wavelength decreases, so it tracks the “shortness” of the wave.
Yes. In some physics contexts, frequency is written as $f$.
On chemistry exams and data tables, $\nu$ is common, while $v$ is better reserved for speed.
Use the limiting measurement (typically the given $\lambda$). Treat $c=3.00\times10^{8}\ \text{m s}^{-1}$ as 3 significant figures unless the problem provides a different value.
Round at the end to avoid compounding rounding error.
Angular frequency is $\omega=2\pi \nu$, with units of $\text{rad s}^{-1}$.
It is mainly used in more advanced wave and quantum treatments and is not typically required for AP Chemistry calculations using $c=\lambda \nu$.
Practice Questions
A photon has wavelength . Calculate its frequency.
Uses and rearranges to (1)
Substitutes correctly to obtain (Hz) (1)
Radiation A has wavelength and radiation B has wavelength .
(a) State which radiation has the higher frequency, and justify your answer. (2 marks)
(b) Calculate the frequency of each radiation. (3 marks)
(a)
Identifies A has higher frequency (1)
Justifies using inverse relationship from (shorter gives larger ) (1)
(b)
Converts nm to m for at least one wavelength: and (1)
Calculates (1)
Calculates (1)

