AP Syllabus focus: ‘At equivalence, pH depends on the major species: strong acid–strong base titrations are neutral, while weak systems have conjugate species that react with water to give basic or acidic solutions.’
Equivalence points are not all “pH 7.” The pH at equivalence is controlled by what solutes remain after stoichiometric neutralisation and whether those species subsequently react with water.
Core idea: pH at equivalence is set by the major species present
In an acid–base titration, reactants undergo a fast, essentially complete stoichiometric reaction (neutralisation). At the equivalence point, neither acid nor base is left in excess; the solution contains products and any spectator ions. The pH is then determined by whether the remaining solute(s) are neutral in water or undergo hydrolysis to form or .
Equivalence point: the point in a titration at which moles of acid and base have reacted in the stoichiometric ratio (for monoprotic systems, moles added equals moles base originally present, or vice versa).
A practical way to decide the pH at equivalence is to identify the major species (highest-concentration solutes) immediately after the neutralisation step, then ask: do any of them react with water to make or ?
Strong acid–strong base titrations: neutral at equivalence
What remains at equivalence
Mixing a strong acid with a strong base leads to complete neutralisation:
Strong acids produce quantitatively in water.
Strong bases produce quantitatively in water.
At equivalence, and have been consumed to form water.
The major dissolved species are typically spectator ions (e.g., , ) plus water. Spectator ions from strong acids and strong bases are negligibly basic/acidic, so they do not hydrolyse to a meaningful extent.
= ion-product constant for water (at 25 °C, )
= hydronium concentration,
= hydroxide concentration,
Because no acidic or basic solute remains (beyond water itself), the pH is governed by water autoionisation. At 25 °C, this gives pH ≈ 7 at equivalence for strong–strong titrations.
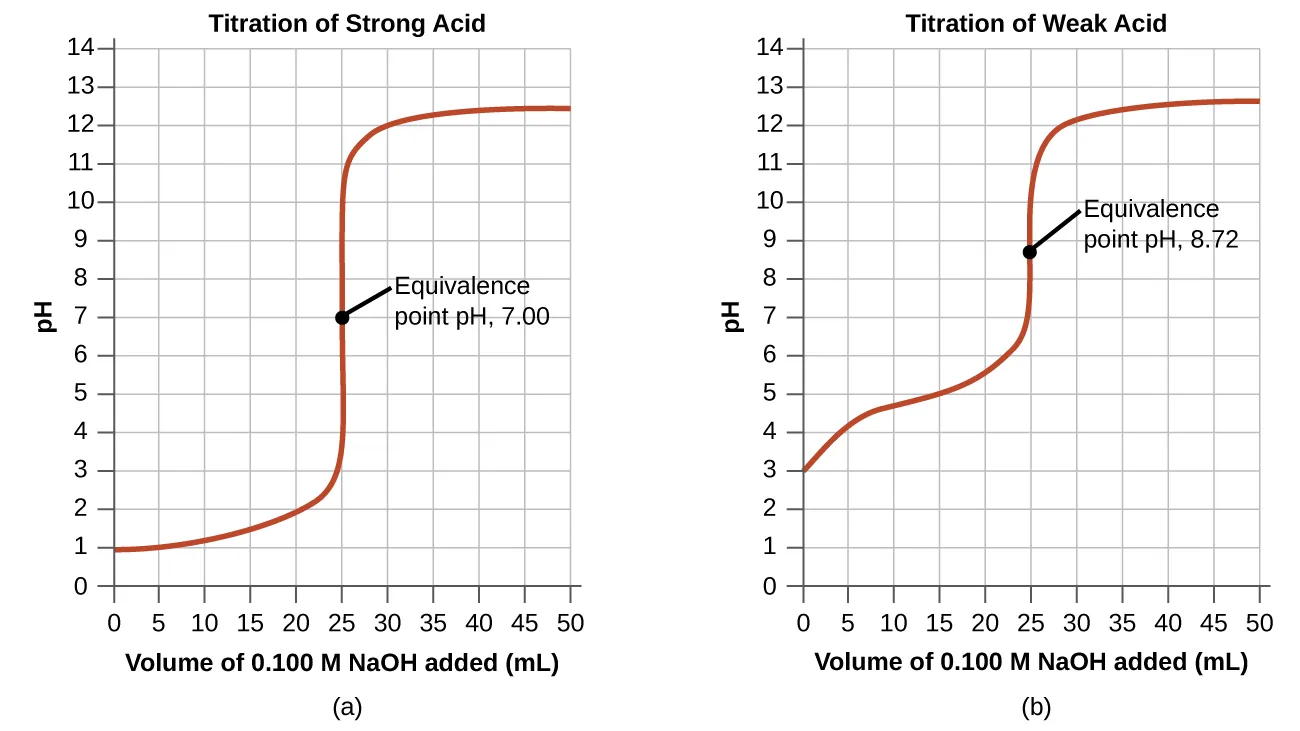
Side-by-side titration curves comparing a strong acid–strong base titration (equivalence at pH 7.00) with a weak acid–strong base titration (equivalence above 7). The labeled equivalence points make the key AP Chemistry idea visually concrete: the equivalence-point pH depends on the acid/base strengths (and thus the species present after neutralization), not on a universal “pH 7” rule. Source
Key takeaway for “what sets pH”
The pH is set by water, since the salt formed is effectively neutral.
Any deviation from pH 7 is usually due to non-ideal effects (outside typical AP assumptions), not acid–base chemistry of the major species.
Weak acid–strong base titrations: basic at equivalence
What remains at equivalence
A weak acid neutralised by a strong base converts mostly into its conjugate base at equivalence. The major species become:
(often at moderate concentration)
spectator cations from the strong base (e.g., )
water
Why pH is greater than 7
The conjugate base is a weak base and hydrolyses water, producing :
This hydrolysis is what sets the pH at equivalence: the solution is basic because is generated. The stronger the original weak acid (larger ), the weaker its conjugate base (smaller basicity), and the closer the equivalence pH is to 7.
Hydrolysis: a reaction in which an ion reacts with water to produce or , thereby affecting pH.
Important reasoning cues:
If the equivalence-point solute is the conjugate base of a weak acid, expect pH > 7.
The pH is set by base hydrolysis, not by leftover strong base (there is none at equivalence).
Weak base–strong acid titrations: acidic at equivalence
What remains at equivalence
A weak base neutralised by a strong acid is converted at equivalence mostly into its conjugate acid . The major species become:
(often at moderate concentration)
spectator anions from the strong acid (e.g., )
water
Why pH is less than 7
The conjugate acid is a weak acid and hydrolyses water, producing :
Thus, the pH at equivalence is acidic because is generated by the conjugate acid.
The stronger the original weak base (larger ), the weaker its conjugate acid, and the closer the equivalence pH is to 7.
Reasoning cues:
If the equivalence-point solute is the conjugate acid of a weak base, expect pH < 7.
The pH is set by acid hydrolysis, not by leftover strong acid (there is none at equivalence).
Practical decision workflow at equivalence (strong vs weak systems)
Use this chemistry-first checklist:
Confirm you are at equivalence (stoichiometric neutralisation complete; no excess strong reactant).
Identify the dominant solute produced:
strong acid + strong base → neutral salt (spectator ions only) → neutral
weak acid + strong base → conjugate base present → basic
weak base + strong acid → conjugate acid present → acidic
State what reaction with water sets the pH:
none (water only) for strong–strong
hydrolysis for weak acid–strong base
hydrolysis for weak base–strong acid
FAQ
Only salts made from a strong acid and a strong base are effectively neutral.
If either ion is the conjugate of a weak species, it can hydrolyse:
conjugate base of a weak acid $\rightarrow$ produces $OH^-$
conjugate acid of a weak base $\rightarrow$ produces $H_3O^+$
Ask whether the ion is a conjugate of a weak species:
$A^-$ hydrolyses if it comes from a weak acid $HA$
$BH^+$ hydrolyses if it comes from a weak base $B$
Ions like $Na^+$, $K^+$, $Cl^-$, and $NO_3^-$ are typically spectators (negligible hydrolysis).
Hydrolysis depends on how much conjugate ion is present.
More concentrated $A^-$ or $BH^+$ at equivalence generally leads to a larger shift from pH 7 because more hydrolysing solute is available per unit volume.
The conjugate base $A^-$ is usually a weak base, so its hydrolysis produces limited $OH^-$.
Also, the solution at equivalence is diluted by the titrant volume, which reduces the concentration of $A^-$ and limits the pH increase.
Neutrality corresponds to $[H_3O^+] = [OH^-]$, but the value of $K_w$ changes with temperature.
So “neutral pH” is not always exactly 7; it equals $\tfrac{1}{2}pK_w$ at that temperature, even though the solution is still neutral in the sense of equal $H_3O^+$ and $OH^-$.
Practice Questions
(2 marks) In separate titrations at 25 °C, (i) is titrated with , and (ii) is titrated with . For each titration, state whether the pH at the equivalence point is less than 7, equal to 7, or greater than 7, and give one reason.
(i) pH equal to 7 (1 mark); reason: salt from strong acid/strong base is neutral so pH set by water/spectator ions do not hydrolyse (1 mark).
(ii) pH greater than 7 (1 mark); reason: (conjugate base) hydrolyses water to form (1 mark).
(6 marks) A student compares equivalence points for two titrations: (A) a weak base titrated with strong acid , and (B) a strong base titrated with strong acid . Explain what major species are present at equivalence in each case and which reaction(s) set the pH. Predict whether each equivalence point is acidic, neutral, or basic.
Titration A: identifies major species include (or in solution) plus spectator and water (1 mark).
States is a conjugate acid that hydrolyses water (1 mark).
Gives hydrolysis reaction (1 mark).
Correctly predicts equivalence pH is acidic, pH < 7 (1 mark).
Titration B: identifies major species are spectator ions (, ) and water (1 mark).
Correctly predicts equivalence pH is neutral (≈7 at 25 °C) because no conjugate species hydrolyses; pH set by water autoionisation (1 mark).

