AP Syllabus focus: ‘For weak acid/base titrations, at the half-equivalence point conjugate pair concentrations are equal (e.g., [HA] = [A−]); therefore pH = pKa and pKa can be found from the curve.’
The half-equivalence point is a key feature of weak acid–strong base and weak base–strong acid titrations. It links the titration curve directly to (or ) with minimal computation.
Understanding the half-equivalence point
What “half-equivalence” means chemically
Half-equivalence point: the point in a titration where the amount (in moles) of titrant added is exactly half the amount needed to reach the equivalence point.
For a weak acid titrated with a strong base, the reaction converts HA to A−. At the half-equivalence point:
Exactly half of the initial HA has been converted to A−
The solution contains a buffer made of the conjugate pair HA/A−
The conjugate pair concentrations are equal: [HA] = [A−] (as stated in the syllabus focus)
For a weak base titrated with a strong acid, the analogous conjugate pair is B/HB+, and at half-equivalence [B] = [HB+].
Practical identification on a titration curve
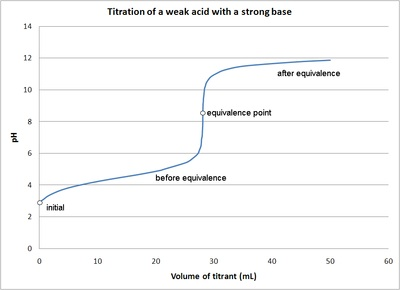
This titration curve shows pH versus volume of titrant for a weak acid–strong base titration, highlighting the buffer region before equivalence and the rapid pH change near the equivalence point. On this kind of graph, the half-equivalence point occurs at half the equivalence-point volume, and the pH read there equals for the weak acid. Source
To locate half-equivalence on a graph of pH vs volume of titrant:
Find the equivalence-point volume (the inflection/steepest-rise centre for a monoprotic titration curve)
Compute (conceptually) half that volume:
Read the pH at ; this pH equals (weak acid case)
This method is primarily graphical/interpretive: you are extracting from the curve rather than from tabulated equilibrium data.
Why pH equals pKa at half-equivalence
The key relationship is the Henderson–Hasselbalch equation, which applies to a buffer containing a conjugate acid–base pair.
= acidity of the solution (unitless)
= for the weak acid (unitless)
= concentration of conjugate base (mol L)
= concentration of weak acid (mol L)
At the half-equivalence point, the syllabus condition [HA] = [A−] makes the ratio , so:
Therefore
This is why the half-equivalence point is so valuable: the curve gives directly.
Weak base version (how to extract pKb)
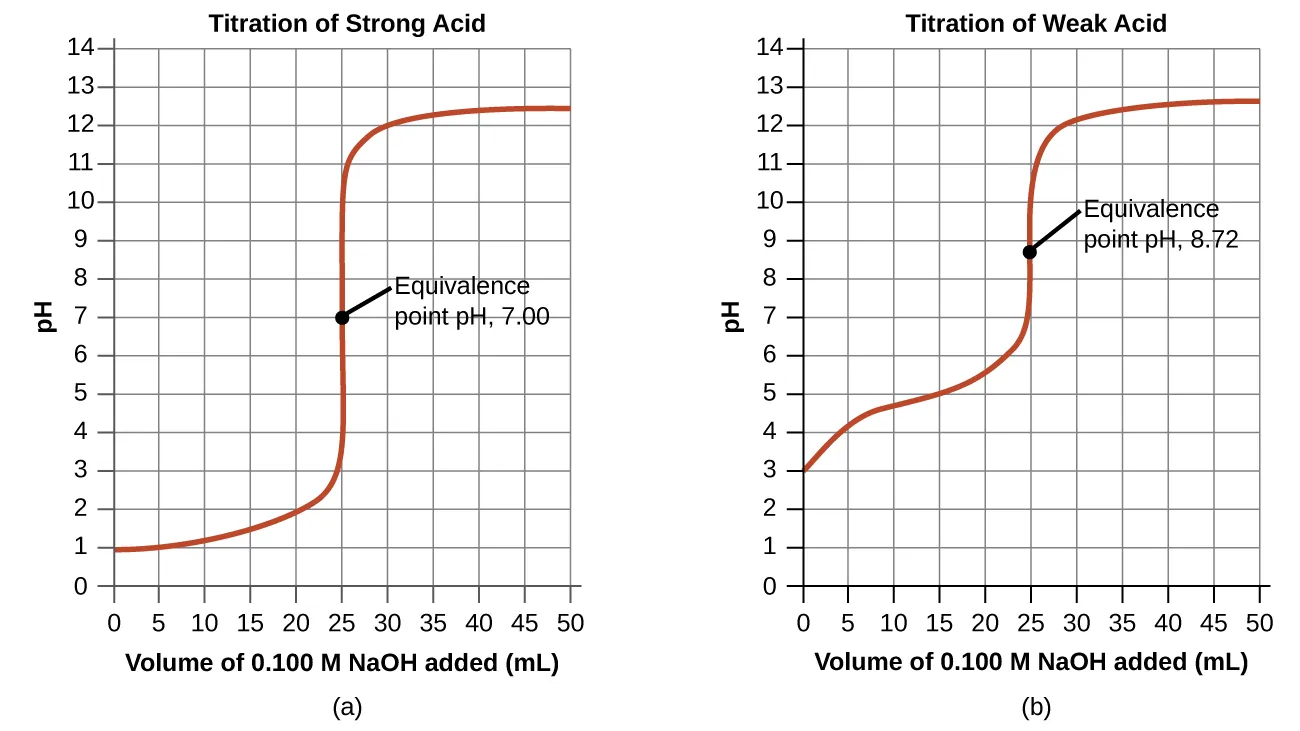
These paired titration curves compare (a) a weak acid titrated with a strong base and (b) a weak base titrated with a strong acid, emphasizing how curve shape and equivalence-point pH differ from strong/strong cases. In both cases, the half-equivalence (midpoint) occurs halfway to the equivalence volume, where the conjugate pair concentrations are equal and the buffer equation simplifies to (or ). Source
For a weak base titrated with strong acid, the buffer pair is B/HB+ and:
At half-equivalence, [B] = [HB+]
The analogous buffer form gives
You can then convert to pH using at 25 °C (if needed)
Using half-equivalence to determine pKa from data
What you must read or infer
To determine from a weak acid titration curve:
Identify that the titration is weak acid + strong base (buffer region present; equivalence pH > 7, but the half-equivalence method itself relies only on the buffer logic)
Locate and then
Read the pH at ; report that value as
Common pitfalls to avoid
Half-equivalence logic requires a conjugate pair buffer; it does not apply to strong acid–strong base titrations (no meaningful to extract).
Ensure you use the volume corresponding to half of the equivalence volume, not “halfway up the steep rise.”
For systems with more than one protonatable site, multiple buffer regions can appear; each half-equivalence corresponds to a different acid step (qualitatively).
FAQ
Because it lies in the strongest buffer region, where both conjugate partners are substantial and the pH changes slowly with added titrant.
No. Both $[A^-]$ and $[HA]$ are diluted by the same total volume, so their ratio remains 1 at half-equivalence.
Small pH probe offsets and sparse data near $V_\text{half}$ can shift the apparent midpoint pH. Denser sampling around half-equivalence reduces uncertainty.
You can misplace $V_\text{half}$ if $V_\text{eq}$ is uncertain. Methods like estimating the inflection point from the steepest slope improve $V_\text{eq}$.
Yes, provided a measurable buffer region exists and equilibrium is established. Extremely weak acids can give less distinct curves, making $V_\text{half}$ harder to read accurately.
Practice Questions
(2 marks) In a titration of a weak monoprotic acid HA with NaOH, the equivalence point occurs at of NaOH added. State the pH– relationship at the half-equivalence point and identify the NaOH volume at that point.
Half-equivalence volume is (1)
At half-equivalence, (1)
(5 marks) A student titrates weak acid HA with strong base and records a titration curve. Explain, using a suitable equation, why the pH at the half-equivalence point equals . Your answer should refer to the relative amounts or concentrations of HA and A at that point and the value of the logarithmic term.
States that at half-equivalence, conjugate pair concentrations are equal: (1)
Writes Henderson–Hasselbalch: (1)
Substitutes ratio or explains (1)
States (1)
Concludes (1)

