AP Syllabus focus: ‘When a weak acid and a weak base are mixed, they react to an equilibrium state: HA + B ⇌ A− + HB+.’
Mixing a weak acid with a weak base creates a proton-transfer system that does not go to completion. Instead, the composition is set by an equilibrium that depends on the relative acid and base strengths.
Core reaction idea
A weak acid HA can donate a proton to a weak base B, producing the corresponding conjugates A− and HB+.
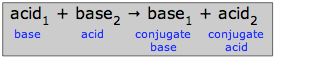
Generalized proton-transfer schematic: an acid and base react to form a conjugate base and conjugate acid, emphasizing the paired nature of products and reactants. This diagram helps you map any specific reaction onto the generic pattern (acid → conjugate base; base → conjugate acid). Source
Because both reactants are weak, appreciable amounts of multiple species often remain present at equilibrium.
Species and conjugate pairs
Conjugate acid–base pair: Two species that differ by one proton, such as HA/A− or HB+/B.
In the mixture, there are two conjugate pairs simultaneously:
HA (acid) and A− (its conjugate base)
HB+ (conjugate acid of the base) and B (base)
A key skill is recognising which species can act as acids or bases in water:
HA and HB+ can donate protons (acids)
A− and B can accept protons (bases)
Equilibrium description of weak acid + weak base mixtures
The reaction is best treated as an equilibrium rather than a “neutralisation” that always finishes.
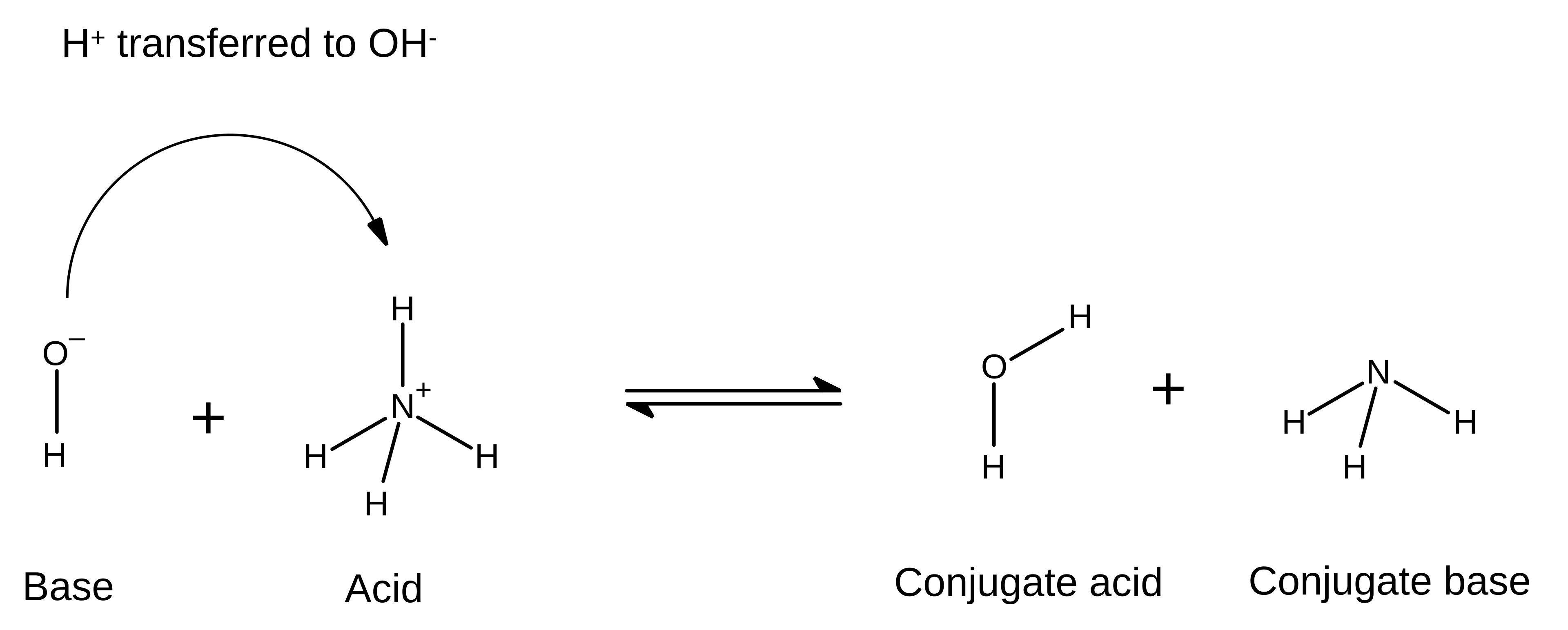
Example of a reversible Brønsted–Lowry proton-transfer equilibrium (ammonium/hydroxide system), drawn as a clean reaction diagram with a double arrow. The figure reinforces that weak acid–base mixtures are governed by equilibrium, so both sides’ species can remain present rather than reacting to completion. Source
This is the central distinction from mixtures involving a strong acid or strong base.
Equilibrium expression
For the reaction HA + B ⇌ A− + HB+, the equilibrium constant is written directly from the balanced equation.
= equilibrium constant (unitless)
= equilibrium concentrations (mol L)
Interpreting this expression:
If K is large, products A− and HB+ are favoured at equilibrium.
If K is small, reactants HA and B remain favoured.
What determines the direction (qualitative)
The equilibrium position depends on relative tendencies to donate/accept protons:
The reaction tends to form the weaker acid and weaker base (the more stable pair).
If HA is the stronger acid compared with HB+, proton transfer to make HB+ is more favourable.
If HB+ is the stronger acid compared with HA, equilibrium favours the left side (less proton transfer).
This comparison can be made using provided acid strength data (for example, relative Ka values or relative pKa values), without assuming complete reaction.
What you can and cannot assume in problem setups
Because both reactants are weak, you generally cannot assume a single “dominant” acid–base reaction that consumes the limiting reagent entirely. Instead:
Treat the initial mixing as setting up the equilibrium HA + B ⇌ A− + HB+.
Expect multiple major species to coexist, especially when the weak acid and weak base have comparable strengths.
The final pH is not automatically 7; it depends on the balance between acid-forming species (HA, HB+) and base-forming species (A−, B).
Practical reasoning cues (without calculation)
If the weak acid is much stronger than the conjugate acid HB+, the mixture shifts toward A− and HB+.
If the weak base is much stronger than A− (as a base), the mixture shifts toward products as well.
If strengths are similar, the equilibrium may lie closer to the middle, leaving significant amounts of all four species.
FAQ
Because both reactants are weak, neither strongly drives proton transfer to completion.
The forward and reverse reactions are both feasible, so equilibrium is established with multiple species present.
Compare $pK_a(HA)$ to $pK_a(HB^+)$.
Higher $pK_a$ = weaker acid
Equilibrium tends to favour the side with the weaker acid.
Not necessarily.
Whether the solution is acidic, basic, or near-neutral depends on how much $HB^+$ and $A^-$ form and which of the remaining species dominate proton donation/acceptance.
Sometimes.
If equilibrium leaves appreciable amounts of a conjugate pair (e.g., both $HA$ and $A^-$, or both $HB^+$ and $B$), the mixture can resist pH changes, but the effectiveness depends on the actual equilibrium composition.
A strong shift in measured pH compared with what you’d expect from simply diluting the original acid or base.
Large pH changes upon mixing imply substantial formation of either $HB^+$ (more acidic outcome) or $A^-$/$B$ (more basic outcome), consistent with an equilibrium strongly favouring one side.
Practice Questions
For the mixture described by , write the equilibrium expression for in terms of equilibrium concentrations.
Correct products over reactants form: 1 mark
Correct species included: in numerator and in denominator: 1 mark
Correct overall expression: : 1 mark
Two separate mixtures are prepared:
Mixture I: weak acid with mixed with weak base whose conjugate acid has
Mixture II: weak acid with mixed with weak base (same as above)
For each mixture, state whether equilibrium lies to the left or right for , and justify using relative acid strengths.
Identifies that equilibrium favours formation of the weaker acid (higher ): 1 mark
Mixture I: compares vs , concludes is weaker acid: 1 mark
Mixture I: states equilibrium lies to the right (products favoured): 1 mark
Mixture II: compares vs , concludes is slightly stronger acid than : 1 mark
Mixture II: states equilibrium lies slightly to the right (or near centre but product-favoured): 1 mark
Clear justification tied to acid strength comparison for both mixtures: 1 mark

