AP Syllabus focus: ‘Strong acids and strong bases react quantitatively (H+ + OH− → H2O); the resulting pH is determined by the concentration of any excess reactant.’
Mixing a strong acid with a strong base produces a fast, essentially complete neutralization. AP Chemistry focuses on using stoichiometry to find which reactant remains in excess and how that leftover sets pH.
Core idea: quantitative neutralization
Strong acid–strong base reactions go to completion because the reacting species are very strong: (or ) and . As a result, equilibrium reasoning is not needed; treat the reaction as quantitative.
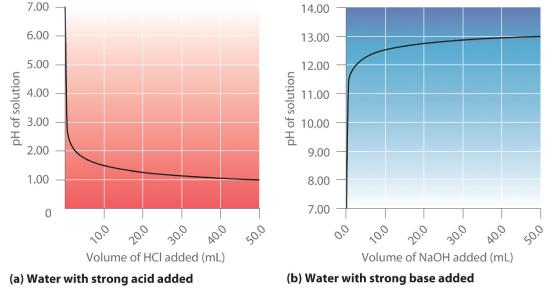
Plots of solution pH versus volume added for strong acid added to water and strong base added to water. Because strong acids/bases dissociate essentially completely, pH changes track the amount of or introduced (a stoichiometric effect). The curves illustrate the rapid pH change at low added volumes and the approach to limiting pH at large added volumes. Source
Neutralization (strong acid–strong base): A reaction in which and combine to form liquid water, consuming the limiting reagent essentially completely.
This means the final pH depends only on which species is left over after reaction: excess (acidic) or excess (basic). If neither is in excess, the mixture is neutral at 25 °C.
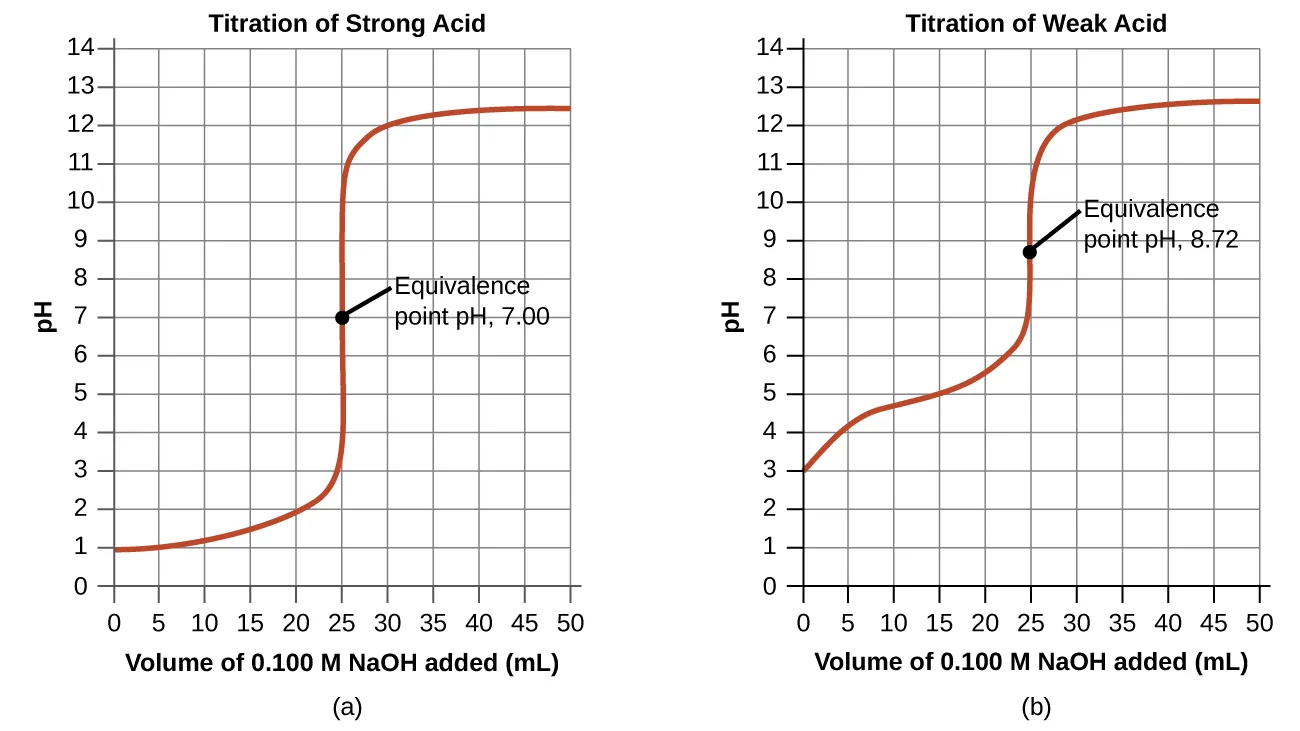
Titration curve for a strong acid (e.g., HCl) titrated by a strong base (e.g., NaOH), highlighting the steep pH jump near the equivalence point. At the equivalence point, moles of acid and base are equal, so the solution is neutral (pH ≈ 7.00 at 25 °C). Past equivalence, the pH is controlled by excess . Source
Essential chemical equation
Use the net ionic equation to represent what actually changes in solution.
= acidic species from the strong acid (mol)
= basic species from the strong base (mol)
When writing full molecular equations, strong electrolytes are typically written as ions in solution; however, for mixture problems the net ionic form is the most direct.
Workflow for strong acid–strong base mixtures (no equilibrium)
1) Convert each reactant to moles of reacting ions
Track moles, not molarity, because mixing changes volume.
Determine moles of provided by the strong acid.
Determine moles of provided by the strong base.
For typical monoprotic strong acids (e.g., HCl, HNO), moles acid = moles . For hydroxide bases, moles base = moles . (If a base formula contains more than one per formula unit, account for that stoichiometry when finding moles .)
2) Identify the limiting reagent and excess reagent
Because the stoichiometry is 1:1 between and :
If , then acid is in excess and leftover remains.
If , then base is in excess and leftover remains.
If , neither remains in excess.
3) Compute the concentration of the excess ion after mixing
First find leftover moles:
Then convert to concentration using the total volume after mixing (sum of solution volumes, assuming additivity):
if acid is in excess
if base is in excess
4) Translate the excess concentration to pH
If acid is in excess, pH is controlled by .
If base is in excess, pH is controlled by (then relate to pH using pH and pOH relationships you have learned).
If neither is in excess, the mixture is neutral (at 25 °C), so pH is 7.00.
Common AP Chemistry checkpoints
Use stoichiometry first, then pH; do not start with pH formulas before determining what remains after reaction.
Do not confuse concentration before mixing with concentration after mixing; dilution by added volume is often the dominant effect.
The spectator ions (e.g., , ) do not affect the neutralization stoichiometry for this topic.
FAQ
When the leftover strong acid/base concentration is much larger than $1.0\times10^{-7}$ M.
If the excess is near $10^{-7}$ M, water autoionization can noticeably affect pH and a more careful approach may be needed.
Strong acids and strong bases are essentially fully dissociated, and $H_3O^+$ and $OH^-$ react to form $H_2O$ with a very large effective equilibrium constant.
This drives the reaction to completion for typical AP conditions.
In the AP context, no: ions such as $Na^+$, $K^+$, $Cl^-$, and $NO_3^-$ are spectators and do not hydrolyse.
They can affect activity in advanced treatments, but that is beyond scope here.
Using initial molarities after mixing (forgetting dilution)
Forgetting the factor of 2 for bases like $Ca(OH)_2$ or $Ba(OH)_2$
Taking $\log$ of moles instead of concentration
At 25 °C with typical strong acid/strong base pairs that produce spectator ions only, it is treated as neutral (pH 7).
At other temperatures, “neutral” pH differs, and very concentrated solutions can deviate slightly from ideal behaviour.
Practice Questions
Q1 (2 marks): 25.0 mL of 0.100 M HCl(aq) is mixed with 10.0 mL of 0.200 M NaOH(aq). State whether the final solution is acidic, basic, or neutral, and identify the species in excess.
1 mark: Calculates/compares moles to show mol and mol (or equivalent comparison).
1 mark: Concludes acidic with excess (or ).
Q2 (5 marks): 30.0 mL of 0.150 M HNO(aq) is mixed with 20.0 mL of 0.100 M Ba(OH)(aq) at 25 °C. Determine the final pH. (Assume volumes are additive.)
1 mark: Finds moles : mol.
1 mark: Finds moles from Ba(OH): mol.
1 mark: Determines excess : mol.
1 mark: Calculates using total volume L: M.
1 mark: pH .

