AP Syllabus focus: ‘Strong bases (group I and II hydroxides) dissociate completely; [OH−] equals the initial group I hydroxide concentration and is twice the initial group II hydroxide concentration.’
Strong bases are treated as fully dissociated electrolytes in water, letting you determine hydroxide concentration directly from formula and concentration. From this, you can calculate pOH and describe solution basicity quantitatively.
What “strong base” means for pOH problems
In AP Chemistry, a strong base is assumed to dissociate completely in aqueous solution, so the equilibrium lies essentially entirely toward ions. This makes hydroxide concentration a stoichiometry result rather than an equilibrium result.
Strong base: A base that dissociates essentially 100% into ions in water, producing hydroxide ions, so the ion concentrations are determined directly from the initial amount dissolved.
Complete dissociation is a modelling assumption used for typical dilute aqueous solutions; you do not set up an ICE table or use a expression for strong bases.
Dissociation patterns you must know
Common strong bases in this subtopic are Group 1 hydroxides and many Group 2 hydroxides.
Group 1 hydroxides (e.g., LiOH, NaOH, KOH) dissociate as:
Group 2 hydroxides that are treated as strong bases when dissolved (e.g., Ca(OH), Sr(OH), Ba(OH)) dissociate as:
The key AP skill is converting a base’s formula and molar concentration into using the dissociation stoichiometry.
Determining from the initial base concentration
Because dissociation is complete, the hydroxide concentration equals the amount of hydroxide released per formula unit times the initial molarity of base that actually dissolves.
For a Group 1 hydroxide :
For a Group 2 hydroxide :
This matches the syllabus requirement: equals the initial Group 1 hydroxide concentration and is twice the initial Group 2 hydroxide concentration.
Writing the relationship in a general way
If a strong base provides hydroxide ions per formula unit, then:
Typical values:
for
for
Be careful to use molar concentration (mol L) for the initial base concentration so that is also in mol L.
Calculating pOH from
Once is known, pOH is found using the logarithmic definition.
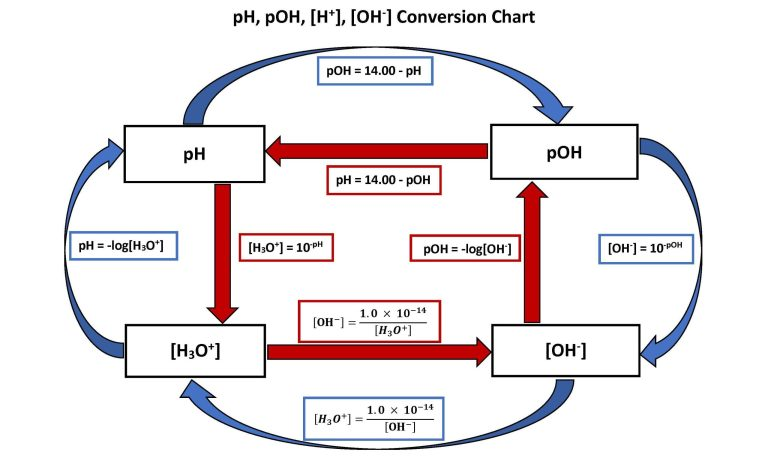
Flow chart linking , pOH, pH, and through the core equations used in AP Chemistry calculations. It emphasizes that pOH is computed directly from hydroxide concentration via , and shows the common follow-up conversions (e.g., and at 25 °C). Source
= measure of hydroxide ion concentration (unitless)
= hydroxide ion concentration in mol L
A lower pOH corresponds to a higher (a more basic solution).
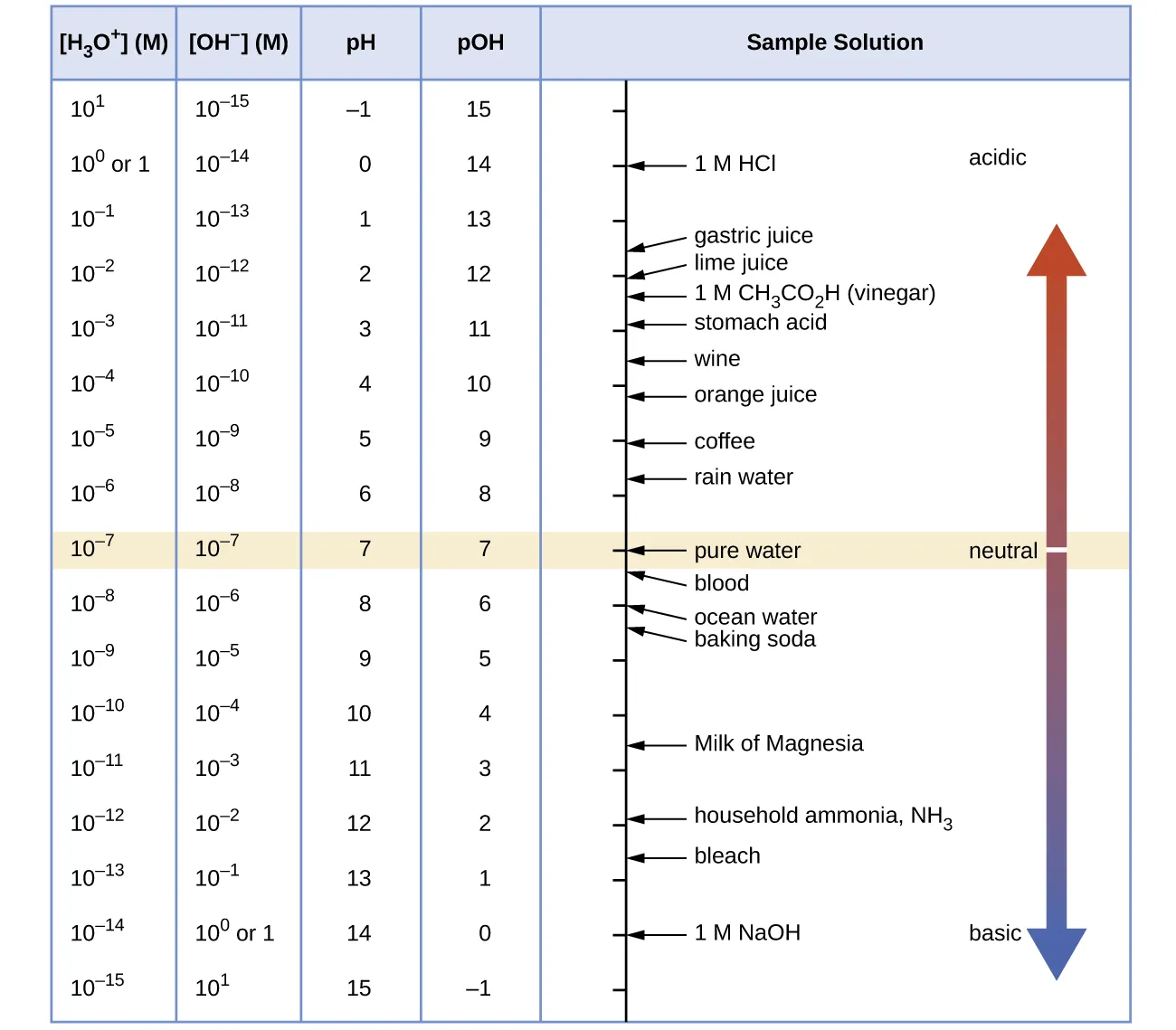
Chart of the pH and pOH scales that maps orders of magnitude in and to corresponding pH/pOH values and common reference solutions. Reading the scale makes the inverse relationship clear: increasing shifts solutions toward lower pOH and greater basicity. This is a conceptual check for whether calculated pOH values are reasonable. Source
When using logs, ensure is expressed in scientific notation with correct significant figures for final reporting.
Common pitfalls (stoichiometry-based)
Do not confuse the base concentration with for compounds; the factor of 2 must be applied.
The coefficient “2” comes from the balanced dissociation equation, not from itself.
If asked for pOH, you must compute first; going directly from base molarity to pOH only works when the hydroxide stoichiometry is handled correctly.
FAQ
Each formula unit contains two hydroxide groups.
When $M(OH)_2$ dissociates completely, it produces $2$ moles of $OH^-$ for every $1$ mole of dissolved base, so $[OH^-]=2[,M(OH)_2,]$.
Not always, because “strong” (dissociation) and “soluble” (amount that dissolves) are separate issues.
Some Group 2 hydroxides are only slightly soluble; if little dissolves, the resulting $[OH^-]$ may be small even though the dissolved portion dissociates fully.
At higher ionic strengths, ion–ion interactions can make concentration differ from activity.
In very concentrated solutions, using activities (rather than concentrations) improves accuracy, but AP problems generally treat concentration as sufficient.
Use the hydroxide stoichiometric factor $n$ from the formula.
For example, a hypothetical $M(OH)_3$ would give $[OH^-]=3[\text{base}]$ if it dissociated completely in water.
Entering the wrong sign or using natural log.
pOH uses base-10 logarithms: $pOH=-\log_{10}[OH^-]$. Ensure brackets are correct and that $[OH^-]$ is in $\text{mol dm}^{-3}$.
Practice Questions
Q1 (2 marks) A student prepares . Assuming complete dissociation, calculate .
States/determines , so (1)
(1)
Q2 (5 marks) Two separate solutions are prepared:
Solution A:
Solution B:
(a) For each solution, determine .
(b) Calculate the pOH of each solution.
(c) State which solution is more basic in terms of hydroxide concentration.
(a)
Solution A: recognises gives 1 , so (1)
Solution B: recognises gives 2 , so (1)
(b)
pOH(A) (1)
pOH(B) (1)
(c)
Concludes equal basicity by hydroxide concentration (both have same ) (1)

