AP Syllabus focus: ‘Strong acids (e.g., HCl, HBr, HI, HClO4, H2SO4, HNO3) ionize completely in water, so [H3O+] equals the initial acid concentration for pH calculations.’
Strong acids are a cornerstone of AP Chemistry pH work because they simplify equilibrium reasoning: they react essentially to completion in water. This lets you connect concentration and pH directly using stoichiometry and dilution ideas.
What “strong acid” means in water
When an acid is described as strong, it undergoes complete ionisation in aqueous solution under typical AP conditions (dilute solutions, room temperature). That means the limiting reactant is not the ionisation reaction; it is the amount of acid you added and the solution volume.
Strong acid: an acid that ionises essentially 100% in water to produce hydronium ions, , so the equilibrium lies far to the products side.
Because of this, strong-acid pH problems are primarily stoichiometry (how many are produced) plus dilution (what is the final volume).
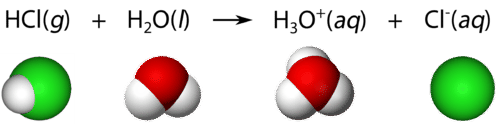
Particle-level diagram of hydrochloric acid dissolving in water, showing proton transfer to form hydronium, , and chloride, . This visual emphasizes why strong acids are treated as fully ionized under typical AP conditions: essentially every acid molecule contributes to . Source
Common strong acids you must recognise
The AP list you should treat as strong in water includes:
Hydrohalic acids (except HF): HCl, HBr, HI
Strong oxoacids: HNO, HClO
Sulfuric acid: HSO (see note below)
Memorising these avoids accidentally setting up a weak-acid equilibrium (ICE table) when it is not needed.
A note on HSO in pH calculations
is diprotic, so it can, in principle, produce two per formula unit. On many AP-style calculations, is treated as a strong acid source of ; however, unless a problem explicitly signals otherwise, be attentive to whether it expects:
1 mol per mol (only the first ionisation considered), or
2 mol per mol (both protons treated as fully released)
The key skill remains the same: translate the formula into moles of produced.
Linking strong-acid concentration to pH
Strong acids simplify to the idea that, after dissolving and reacting with water, the hydronium concentration is determined by the amount of acid present (after any mixing or dilution).
= hydronium ion concentration in
= initial (formal) molar concentration of the acid in
= number of produced per acid formula unit (often 1; can be 2 for diprotic acids if assumed)
The approximation becomes your working equality in typical AP contexts because “ionise completely” implies the remaining un-ionised acid is negligible compared with the initial amount.
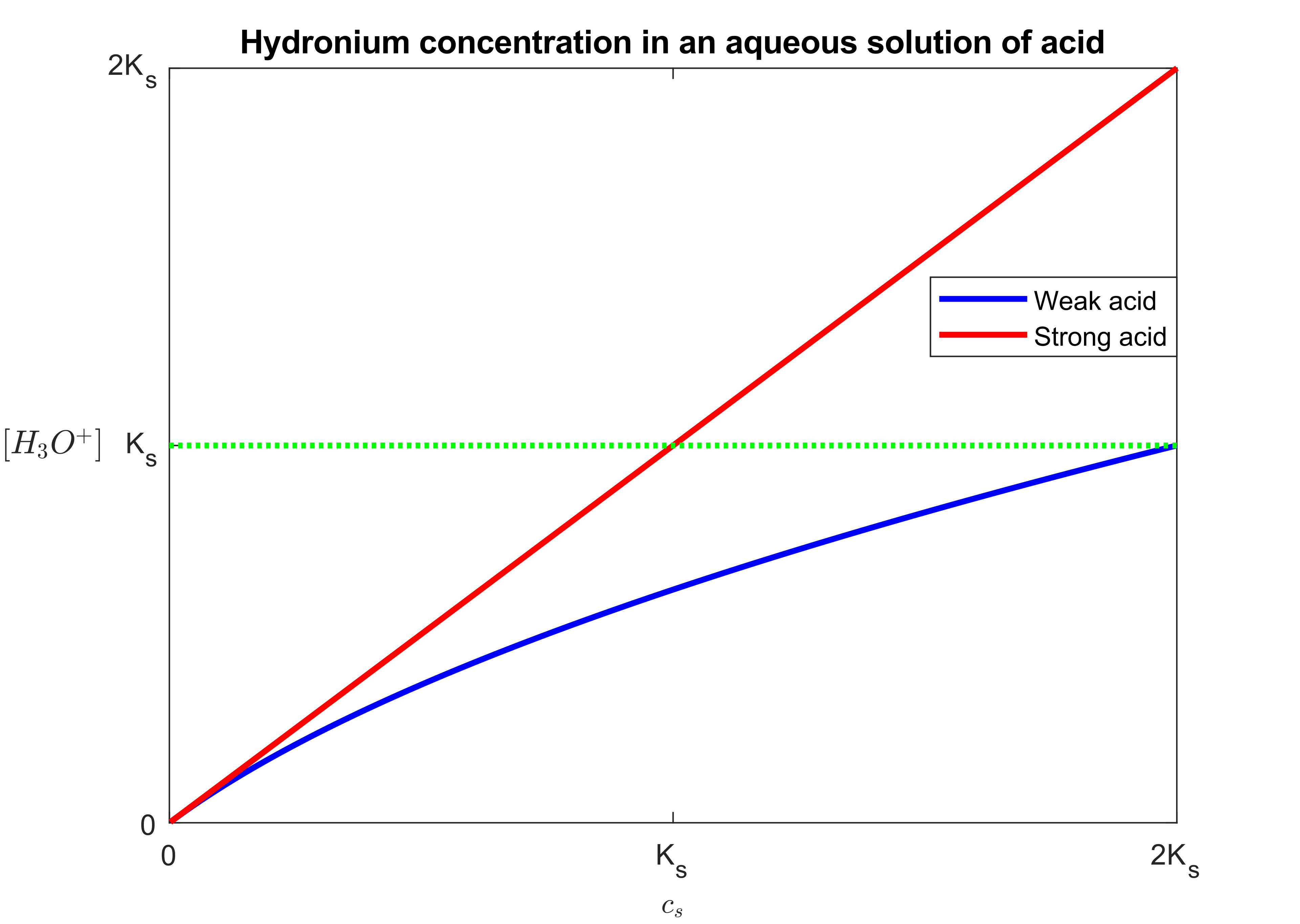
Graph illustrating hydronium concentration for a strong acid (treated as fully dissociated) versus a weak acid (partial dissociation). The strong-acid curve visually supports the AP shortcut that is essentially set by the formal acid concentration (after any dilution), while weak acids require equilibrium reasoning. Source
Practical procedure (no equilibrium setup)
To determine pH for a strong acid solution (including after mixing), follow this logic:
Write the ionisation reaction as producing (for example, ).
Use stoichiometry to determine moles of produced.
For monoprotic strong acids (HCl, HBr, HI, HNO, HClO): moles = moles acid.
For acids with more than one acidic proton (notably HSO): multiply by the expected number of released protons, if instructed or implied.
If solutions are mixed, compute the total volume and find .
Convert to pH using the definition.
Common pitfalls to avoid
Do not set up an ICE table for a strong acid’s ionisation; the equilibrium lies overwhelmingly to products.
Do not confuse initial concentration in the container with the final concentration after dilution/mixing; pH depends on the final .
Do not forget that the chemistry is about in water; strong-acid pH is fundamentally a hydronium concentration problem.
FAQ
Because their $K_a$ values are extremely large, the equilibrium lies overwhelmingly toward products.
In dilute aqueous solutions, the fraction un-ionised is negligible relative to the stated concentration, so it does not meaningfully affect pH.
Look for cues in the question wording and data provided.
If it says “strong acid” without qualification, some questions expect $2\mathrm{H^+}$ per $\mathrm{H_2SO_4}$.
If it emphasises stepwise behaviour or provides $K_a$ information, it may not.
Not necessarily. When the acid is extremely dilute, water’s own $\mathrm{H_3O^+}$ contribution can become comparable.
In that edge case, $[\mathrm{H_3O^+}]$ is not dominated by the acid alone.
Ionisation extent (percent ionised) stays effectively 100%, but concentration changes.
Dilution increases volume, lowering $[\mathrm{H_3O^+}]$, which increases pH via $\mathrm{pH}=-\log[\mathrm{H_3O^+}]$.
Yes, both notations are commonly used to represent the same aqueous species relationship.
However, $\mathrm{H_3O^+}$ is chemically more accurate in water because free $\mathrm{H^+}$ does not exist independently.
Practice Questions
(2 marks) State why can be taken as equal to the initial concentration of when calculating pH.
HCl is a strong acid / ionises completely in water (1)
Therefore essentially all acid molecules form so (1)
(5 marks) A student prepares a solution by adding of to of water. Assuming complete ionisation, determine the pH of the final solution.
States/uses that is strong so moles = moles (1)
Calculates moles: with in (1)
Determines total volume (or ) (1)
Finds (1)
Uses to obtain pH (1)

