AP Syllabus focus: ‘The impact of a common ion on solubility can be calculated using the salt’s Ksp expression for the dissolution equilibrium.’
A common ion suppresses the dissolution of a slightly soluble ionic solid.
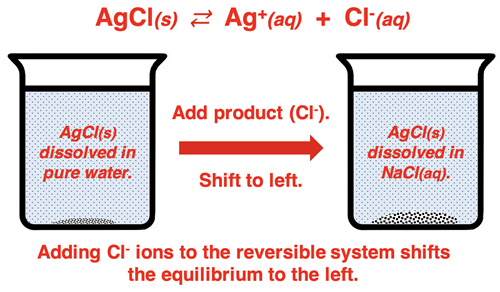
Beaker-level schematic of showing that adding the common ion drives the equilibrium to the left (toward the solid). This diagram connects Le Châtelier’s principle to the observed decrease in molar solubility when a common ion is present. Source
AP Chemistry expects you to quantify that suppression by combining the expression with equilibrium concentrations and careful stoichiometry.
Core idea: using with a common ion
When a sparingly soluble salt dissolves, the equilibrium is described by a solubility-product constant built from aqueous ion concentrations only (the solid is omitted). A common ion is an ion already present in solution that also appears among the dissolution products; it shifts the dissolution equilibrium toward the solid, decreasing solubility.
Molar solubility — the number of moles of an ionic solid that dissolve per litre of solution to form a saturated solution (mol L).
To calculate a common-ion effect, you treat the common-ion concentration as part of the initial equilibrium set-up, then solve for how much additional ion can be produced by dissolution while still satisfying .
Writing the correct expression
Start from a balanced dissolution equation. For a general salt :
= solubility-product constant (unitless in the thermodynamic sense; treated with concentration units in many AP problems)
= equilibrium molar concentration of the metal ion (mol L)
= equilibrium molar concentration of the anion (mol L)
Between setting up the expression and solving, keep track of stoichiometric powers (coefficients become exponents) and ensure you are using equilibrium ion concentrations, not initial values.
Setting up equilibrium concentrations (common-ion present)
A standard workflow is an ICE-style set-up (Initial, Change, Equilibrium), even if you do not draw a full chart.
Step 1: identify the common ion source
Common ions often come from:
a soluble salt (e.g., NaCl provides )
a strong acid/base that supplies an ion (e.g., HCl provides )
a previously dissolved portion of the same salt (less common in AP prompts)
Step 2: define the solubility variable
Let represent the additional amount of solid that dissolves (in mol L). Then write equilibrium ion concentrations as:
(initial common ion) (stoichiometric amount produced from )
For example, if is the common ion and its initial concentration is , then at equilibrium:
(if no initial is present)
Step 3: substitute into the expression
Insert those equilibrium expressions into and solve for . The value of is the molar solubility in the presence of the common ion.
Solving the algebra: approximation vs quadratic
Common-ion problems often produce an equation where is added to a much larger initial ion concentration, such as .
When an approximation is reasonable
If the common ion is present at a relatively high concentration, may be negligible compared with .

ICE-style concentration setup highlighting the common ion as a nonzero initial concentration and the typical simplifying assumption that the added amount from dissolution () is negligible compared with the initial common-ion concentration. This is the standard justification for approximations like when solving problems. Source
Then:
the algebra simplifies substantially, typically giving a direct expression for
After solving, you should validate the approximation by checking whether (for example, whether it is only a few percent of the initial concentration).
When you must solve a quadratic
If the common ion concentration is not overwhelmingly large (or is zero), you may need to keep the term and solve the resulting polynomial. In AP contexts, you are generally expected to choose the physically meaningful root (positive concentration) and reject negative values.
Interpreting the result correctly
A larger initial common-ion concentration leads to a smaller calculated , meaning lower solubility.
The effect is stronger when the common ion appears with a larger exponent in (for instance, ions with coefficient 2 contribute squared terms).
Ensure your final reported quantity matches what is asked:
molar solubility (mol L) vs
an individual ion’s equilibrium concentration (which may be or )
FAQ
Compare your final $ks$ to $c$.
A common rule is that if $\dfrac{ks}{c}\le 0.05$ (5%), the approximation is usually acceptable for typical AP-style problems.
In real solutions, yes: higher ionic strength can change ion activities, so the measured solubility may shift.
In ideal AP calculations, you typically ignore activity effects and treat $K_{sp}$ as depending only on concentrations of the ions in the expression.
You include both in the initial conditions: $[M^{n+}]=[M]_0+as$ and $[X^{m-}]=[X]0+bs$, then substitute into $K{sp}$.
This often forces a quadratic (or higher-order) equation.
Account for stoichiometry in the initial common-ion concentration.
For $CaCl_2$, the initial $[Cl^-]_0$ contributed is $2\times[CaCl_2]$, assuming complete dissociation.
Use the root that keeps equilibrium concentrations physically consistent and aligned with the “small dissolution” assumption for a sparingly soluble solid.
Often, one root would imply unrealistically large ion concentrations that contradict the premise of limited solubility.
Practice Questions
(1–3 marks) For , . Calculate the molar solubility of in .
Writes (1)
Uses and (1)
(1)
(4–6 marks) , . Calculate the molar solubility of in a solution initially containing . State any approximation used.
Writes (1)
Sets and (1)
Substitutes: (1)
Uses approximation and states it (1)
Solves (1)
Checks (e.g. ) (1)

