AP Syllabus focus: ‘A salt is less soluble in a solution that already contains one of its ions; this common-ion effect can be explained qualitatively using Le Châtelier’s principle.’
A slightly soluble ionic solid exists in a balance between undissolved solid and dissolved ions. Adding an ion already present disturbs this balance, reducing additional dissolution and often promoting precipitation.
Core idea: the common-ion effect
What it is and when it matters
Common-ion effect: The decrease in solubility of an ionic compound when a solution already contains one of the ions produced by the compound’s dissolution.
This effect is most noticeable for slightly soluble salts (those that establish a dissolution equilibrium rather than dissolving completely). The key is that the solution already has a common ion (an ion that appears on the product side of the dissolution equilibrium).
Dissolution is a reversible equilibrium
For a salt , dissolution and precipitation occur simultaneously. At equilibrium, the rate of dissolution equals the rate of precipitation, and ion concentrations stay constant.
= Undissolved ionic solid (activity effectively constant)
= Dissolved ions in solution
= Solubility-product constant at a given temperature
The equilibrium constant expression is useful here because it shows that the ion concentrations are linked at equilibrium; if one ion concentration is forced higher, the other cannot remain as high without violating the equilibrium condition.
Qualitative explanation using Le Châtelier’s principle
Applying Le Châtelier correctly
Le Châtelier’s principle states that a system at equilibrium responds to a stress by shifting in the direction that reduces that stress.
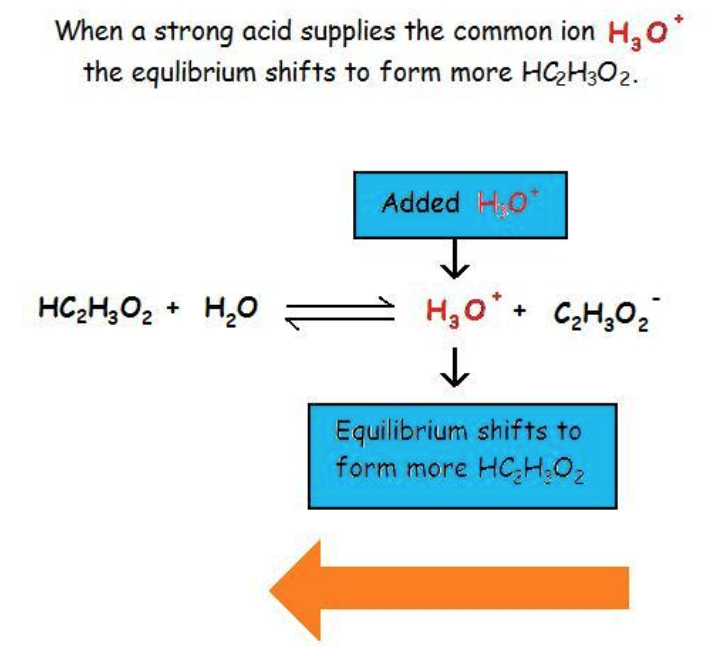
This diagram shows how adding a strong acid increases , a product in the weak-acid equilibrium, causing the equilibrium to shift left. It visually reinforces the Le Châtelier idea that increasing a product concentration drives the reaction toward reactants, reducing ion formation at the new equilibrium. Source
For the dissolution equilibrium, a “stress” can be the addition of an ion already present among the dissolved products.
What happens when you add a common ion
If you add a soluble compound that provides (or ), you immediately increase the concentration of a product of the dissolution reaction.
The system responds by shifting left, toward the solid, which reduces the added ion’s effect by removing ions from solution.
Qualitatively, this means:
More solid remains undissolved (or forms if ions were already present at high enough levels).
The salt’s molar solubility decreases in the presence of the common ion.
The solution contains fewer ions from further dissolution of that salt than it would in pure water.
Why the shift reduces solubility (molecular-level picture)
Adding a common ion increases the frequency of ion-ion encounters near the solid surface. This makes it more likely for ions to recombine into the ionic lattice (precipitation) rather than escape into solution (dissolution). The dynamic equilibrium still exists, but the new balance favours the solid phase.
“Less soluble” does not mean “insoluble”
The salt does not necessarily stop dissolving entirely; rather, the equilibrium is re-established at a lower concentration of the ions that come from the slightly soluble salt. Some dissolution still occurs, but to a smaller extent.
Recognising common-ion situations (no calculations)
Typical ways a common ion is introduced
Adding a soluble salt that contains one of the ions of the slightly soluble salt
Mixing two solutions where one already contains a relevant ion
Using an electrolyte “background” solution that supplies a shared ion
Observable consequences (qualitative)
Depending on concentrations, you may observe:
Suppressed dissolving of a solid added to a solution containing a common ion
Increased cloudiness or formation of a precipitate if the ion product becomes large enough that precipitation is favoured
A mixture that looks unchanged but has a different equilibrium composition (equilibrium changes can be invisible)
Common pitfalls in reasoning
Do not say equilibrium “stops”; it shifts to a new position while remaining dynamic.
The stress is adding a product ion, so the shift is toward reactants (solid).
The qualitative prediction assumes temperature constant, since equilibrium constants depend on temperature.
FAQ
No. A common ion always favours the solid side for a slightly soluble salt, but you only see a precipitate if conditions allow enough solid to form to be noticeable.
Even without visible change, the equilibrium ion concentrations can still shift.
Adding more solid to a saturated equilibrium mixture does not change equilibrium ion concentrations (as long as some solid is already present).
Adding a common ion changes the dissolved-ion conditions, so the equilibrium position shifts.
Then both product concentrations increase, strongly favouring the solid side.
Qualitatively, you expect even lower solubility and a higher tendency to precipitate.
Yes. High ionic strength can change effective concentrations (activities), so the simple concentration picture becomes less exact.
Qualitatively, the direction of shift remains the same, but the magnitude can differ.
Because the ion is shared between two dissolved sources:
the slightly soluble salt’s dissolution
an added soluble compound providing the same ion
Practice Questions
(2 marks) Explain, using Le Châtelier’s principle, why adding decreases the solubility of in water.
Identifies that is a common ion/product of dissolution (1)
States equilibrium shifts left/towards solid to reduce added , so solubility decreases (1)
(5 marks) A student compares the solubility of in pure water versus in a solution containing . Without using numbers, describe how the concentrations of ions and the amount of solid present differ at equilibrium, and justify your statements.
Writes/describes dissolution equilibrium: (1)
States from is a common ion/product (1)
Predicts shift left/towards solid upon adding (Le Châtelier) (1)
Concludes molar solubility decreases; less from dissolution at equilibrium than in pure water (1)
States more undissolved remains (or precipitation more favoured) at equilibrium (1)

