AP Syllabus focus: ‘If energy released in bond formation exceeds energy required to break bonds, the reaction is exothermic; otherwise it is endothermic.’
Bond energies let you predict whether a reaction tends to release or absorb heat by comparing energy needed to break reactant bonds with energy released when new product bonds form.
Core idea: compare energy in vs. energy out
Chemical reactions rearrange atoms by breaking bonds (requires energy input) and forming bonds (releases energy). Using bond energies, you estimate whether the net energy change is negative (exothermic) or positive (endothermic) by comparing these two totals.
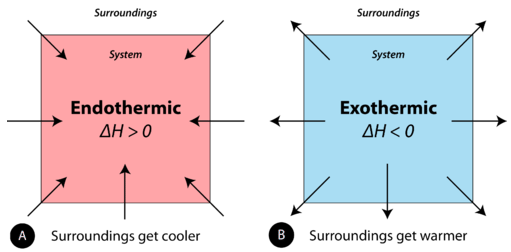
System–surroundings energy-flow schematic contrasting endothermic and exothermic processes. The arrows indicate the direction of heat transfer, and the sign of summarizes whether the reaction mixture gains or loses enthalpy at constant pressure. Source
Energy in: sum of bond energies for all bonds broken in the reactants
Energy out: sum of bond energies for all bonds formed in the products
Decide process type:
If energy out > energy in, more energy is released than absorbed → exothermic
If energy in > energy out, more energy is absorbed than released → endothermic
What bond energies represent (and what they don’t)
Average bond enthalpy (bond energy): the average energy required to break one mole of a specific covalent bond in gaseous molecules, forming separated gaseous atoms (units: kJ/mol).
Because these are averages, they give an estimate of reaction energy, not an exact experimental value. They are most appropriate when the reaction involves covalent bonds and can be reasonably approximated as gas-phase bond breaking/forming.
The sign of the energy change from bond energies
The comparison can be expressed as an estimated reaction enthalpy.
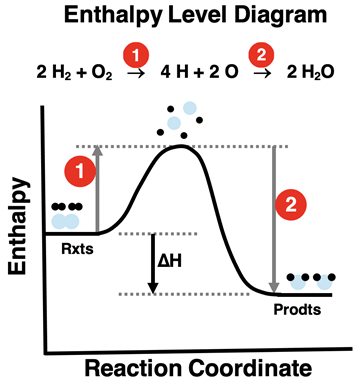
Enthalpy-level diagram modeling a reaction as two conceptual stages: bond breaking (energy input raises the system enthalpy) followed by bond formation (energy release lowers the enthalpy). The net vertical difference between reactants and products corresponds to the overall estimate from summed bond energies. Source
A negative result corresponds to an exothermic process, consistent with the syllabus statement that greater energy released in bond formation implies exothermicity.
= estimated enthalpy change of reaction, kJ/mol of reaction (as written)
= bond energy (average bond enthalpy) for a specific bond type, kJ/mol
Use the balanced chemical equation to ensure you count the correct number of each bond broken and formed. The stoichiometric coefficients determine how many molecules (and therefore how many bonds) are involved “per mole of reaction.”
How to decide exothermic vs. endothermic from the estimate
Once you compute (or conceptually compare) the two bond-energy sums:
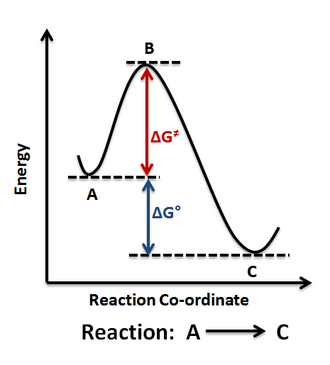
Reaction-coordinate (energy profile) diagram showing reactants converting to products through a transition state (activated complex). The vertical axis represents relative energy, so the products being lower or higher than the reactants corresponds to exothermic or endothermic overall energy change, respectively. Source
If :
the products’ bonds are, in total, stronger (more energy released upon forming them)
the reaction is exothermic (heat flows to surroundings at constant pressure)
If :
the reactant bonds are, in total, stronger (more energy required to break them)
the reaction is endothermic (heat must be absorbed from surroundings)
Practical bond-counting guidance (no calculation required)
Correct classification depends on correctly identifying which bonds change.
Bonds broken
Count bonds present in reactants that are not present in products (or are reduced in number). Tips:
Treat double and triple bonds as distinct bond types (e.g., C=C differs from C–C).
Only include bonds that are actually broken; many bonds are unchanged spectators.
Bonds formed
Count bonds present in products that were not present in reactants (or are increased in number). Tips:
If a bond type changes (single to double), model this as breaking the original bond type and forming the new bond type.
Use the balanced equation so totals reflect the full reaction.
Interpreting your decision scientifically
Bond-energy reasoning links macroscopic heat flow to microscopic structure:
Exothermic: forming new attractions lowers the system’s potential energy more than breaking old attractions raises it, so excess energy can appear as thermal energy in the surroundings.
Endothermic: the system must “store” energy to create products because breaking bonds dominates, so it draws in thermal energy from the surroundings.
FAQ
They are averaged over many compounds because the same bond (e.g., C–H) has slightly different strengths in different chemical environments.
Factors include neighbouring atoms, bond polarity, and molecular geometry.
Bond energies mainly model covalent bond breaking/forming in gases.
For strongly ionic processes, estimates based only on covalent bond enthalpies can be unreliable because lattice-level electrostatic interactions are not captured.
Use the bonding implied by the structural formula you are given, but recognise that delocalisation means “one bond type” may not match a single average bond enthalpy well.
This is a key source of estimation error.
Yes. Average bond enthalpies are defined for gaseous species.
If reactants/products are liquids or solids, the estimate omits additional energetic effects associated with intermolecular attractions.
It suggests bond-breaking and bond-forming energies nearly balance.
In practice, the true $ \Delta H_{\text{rxn}} $ may still be meaningfully positive or negative because the “average” values introduce uncertainty.
Practice Questions
(2 marks) Using bond energies, state the criterion for a reaction to be exothermic and relate it to the sign of .
States that if energy released forming bonds exceeds energy required to break bonds, reaction is exothermic. (1)
Links exothermic to (negative). (1)
(5 marks) A student estimates using average bond enthalpies and obtains a positive value. Explain, in terms of bonds broken and formed, what this implies about energy transfer and classify the reaction as endothermic or exothermic. Include one limitation of using average bond enthalpies.
Identifies that a positive estimate means energy to break bonds is greater than energy released forming bonds. (2)
Correctly classifies as endothermic. (1)
States that energy is absorbed from the surroundings (net heat input) at constant pressure. (1)
Gives one valid limitation (e.g., bond enthalpies are averages; depend on molecular environment; best for gas-phase covalent bonds). (1)

