AP Syllabus focus: ‘During a chemical reaction, bonds are broken and/or formed, changing the system’s potential energy.’
Chemical reactions rearrange atoms by breaking some bonds and forming others. Because chemical bonds store energy as potential energy, these bond changes shift the system’s potential energy and control whether energy must be absorbed or can be released.
Core idea: bonds as stored potential energy
Atoms in molecules are held together by electrostatic attractions between nuclei and electrons. That attraction lowers the system’s potential energy compared with separated atoms.
Potential energy (in chemical systems): energy stored due to particle positions and interactions (especially electrostatic attractions/repulsions), not due to particle motion.
A useful mental model is that a stable bond corresponds to a “low” potential-energy arrangement.
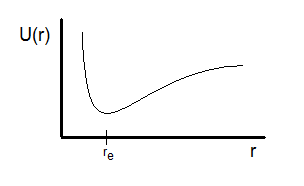
Potential-energy curve for a diatomic interaction as a function of internuclear distance, with a minimum at the equilibrium bond length . The left side rises sharply due to strong electron-cloud/nucleus repulsions at very short distances, while the right side approaches the separated-atoms limit as attractions weaken. The depth of the well corresponds qualitatively to bond strength and the energy required to separate the atoms. Source
Changing bonding means moving between potential-energy states.
Why potential energy changes during reactions
Reactants and products have different sets of bonds (different interactions and arrangements).
Different bond arrangements correspond to different electrostatic potential energies.
Therefore, as bonds are broken and formed, the system’s potential energy changes.
Bond breaking: energy input to separate interacting particles
Breaking a bond means pulling bonded atoms apart far enough that the attractive interaction that defines the bond is overcome.
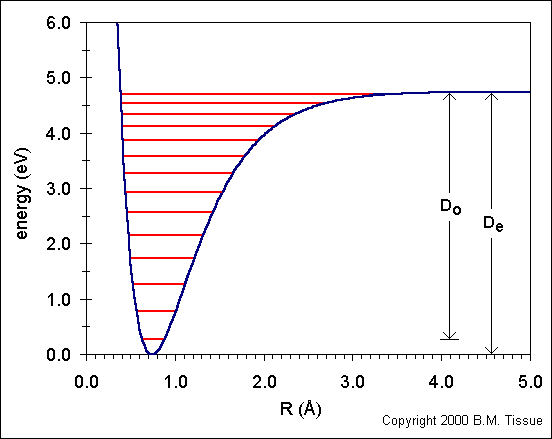
Morse potential energy well for a diatomic molecule, showing discrete vibrational energy levels within the well and the approach to dissociation at large separation. The minimum corresponds to the most stable (lowest potential-energy) bond length, while moving upward reflects added energy stored in bond stretching. Dissociation occurs only after enough energy is supplied to reach the top of the well, consistent with bond breaking requiring energy input. Source
What happens energetically when a bond breaks
As atoms separate, attractions between nuclei and shared electrons are reduced.
The system moves to a higher potential-energy state relative to the bonded state.
Energy must be transferred into the system to accomplish this separation.
Key phrasing for AP Chemistry: bond breaking is endothermic at the step level (it requires energy), even if the overall reaction may be exothermic.
Common misconceptions to avoid
“Bonds contain energy that is released when broken” is incorrect. If energy were released by breaking bonds, molecules would spontaneously fall apart.
The energy “stored” in a bond is better understood as the energy difference between the bonded, low-potential-energy state and the separated, higher-potential-energy state.
Bond forming: energy release when attractions are established
Forming a bond means bringing atoms together into a configuration where net electrostatic attractions lower the system’s potential energy.
What happens energetically when a bond forms
As atoms approach, attractive interactions dominate over repulsions at the bonding distance.
The system moves to a lower potential-energy state (more stable arrangement).
The decrease in potential energy is released as energy to the surroundings, often appearing as increased particle motion (thermal energy).
Key phrasing for AP Chemistry: bond formation is exothermic at the step level (it releases energy), even if the overall reaction may be endothermic.
Physical interpretation of the released energy
When potential energy decreases during bond formation, energy must be conserved. The “lost” potential energy is transferred out of the system, typically as:
Heat (increased random motion of nearby particles)
Occasionally other pathways (e.g., light in chemiluminescence), but the central AP idea is energy transfer associated with potential-energy changes.
Reactions combine both processes simultaneously
In most reactions, some bonds are broken and others are formed. The system’s potential energy changes because the reaction replaces one set of interactions with another set.
Thinking in terms of competing contributions
Breaking reactant bonds tends to raise potential energy (requires energy input).
Forming product bonds tends to lower potential energy (releases energy).
The overall change in the system’s potential energy depends on the balance between these two effects.
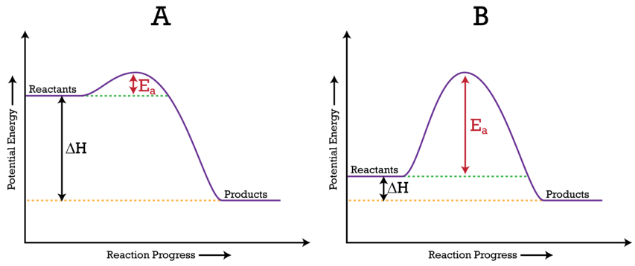
Reaction-coordinate diagram showing reactants rising to a transition state (activation energy barrier) and then falling to products. The vertical separation between reactants and products represents the net energy change ( or , depending on context), while the peak height above reactants corresponds to . This helps distinguish the energy barrier (kinetics) from the overall energy change (thermodynamics). Source
This directly supports the syllabus statement: “bonds are broken and/or formed, changing the system’s potential energy.” Even without calculation, you should be able to reason qualitatively that a reaction producing “stronger/more stabilising” bonding arrangements tends to end at lower potential energy than it started.
Molecular stability and relative bond strengths (qualitative)
Different bonds lower potential energy by different amounts. Qualitatively:
A stronger bond corresponds to a larger drop in potential energy upon formation.
A weaker bond corresponds to a smaller drop in potential energy upon formation.
Stronger bonds therefore generally require more energy to break and release more energy when formed.
This is why the identity and number of bonds matter: changing from one bonding pattern to another changes how far down (or not) the products sit in potential energy relative to reactants.
Connecting microscopic energy changes to observable effects
Because bond changes alter chemical potential energy, the energy difference can be transferred between the reacting system and its surroundings. When energy is transferred as heat:
If more energy is released during bond formation than is required for bond breaking, the surroundings can warm.
If more energy is required for bond breaking than is released during bond formation, the surroundings can cool.
These temperature observations are consequences of potential-energy changes caused by bond breaking and bond forming, not the cause of them.
FAQ
As atoms approach, attraction between nuclei and electrons increases, lowering potential energy, but at very short distances nucleus–nucleus and electron–electron repulsions rise sharply.
The stable bond length occurs near the minimum of the potential-energy curve where attractions and repulsions balance.
Not always. Often it becomes thermal energy via collisions, but it can also be emitted as light (e.g., in some excited-state processes) or transferred into other molecular motions.
In typical AP-level thermochemistry contexts, treating it as heat transfer is usually appropriate.
Multiple bonds generally involve more shared electron density and stronger attraction, typically lowering potential energy more than a single bond.
Therefore, forming a multiple bond often releases more energy, and breaking it often requires more energy, though exact values depend on the atoms involved.
Bond strength depends on factors like atomic size (orbital overlap), electronegativity differences (bond polarity), and electron density distribution.
Better overlap and stronger electrostatic attraction usually correspond to a deeper potential-energy well (stronger bond).
At a given temperature, particles have a distribution of kinetic energies. A fraction of collisions can have enough energy and proper orientation to begin disrupting bonds.
Once some new bonds begin forming, the released energy can help sustain further reaction progress in exothermic processes.
Practice Questions
(2 marks) Explain, in terms of potential energy, why breaking a chemical bond requires an input of energy.
States that separating bonded atoms increases the system’s potential energy / moves to a higher potential-energy state. (1)
States that energy must be transferred into the system to overcome attractive interactions (bond breaking is endothermic at the step level). (1)
(5 marks) A reaction converts reactant molecules into product molecules with a different set of bonds. Describe how bond breaking and bond forming change the system’s potential energy, and how these changes can lead to either heating or cooling of the surroundings.
Identifies that reactions involve both bond breaking and bond forming. (1)
States bond breaking increases potential energy and requires energy input. (1)
States bond forming decreases potential energy and releases energy. (1)
Links overall potential-energy change to the balance between energy required to break bonds and energy released on forming bonds. (1)
Connects energy transfer to surroundings as heat: net release can warm surroundings; net absorption can cool surroundings. (1)

