AP Syllabus focus: ‘Approximations such as the pre‑equilibrium assumption are used to derive a rate law for mechanisms where the first step is not rate limiting.’
Pre-equilibrium kinetics connects a multistep mechanism to an experimentally testable rate law. When an early step rapidly establishes equilibrium, you can eliminate intermediates algebraically and express the rate using only reactant concentrations.
Concept: the pre-equilibrium assumption
In some mechanisms, an initial elementary step is fast and reversible, quickly reaching equilibrium, while a later step is slow and controls the overall rate.
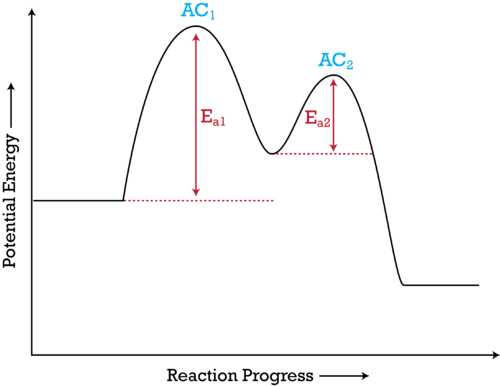
A labeled potential energy diagram for a two-step reaction, showing two activation-energy barriers and an intermediate between them. The larger barrier corresponds to the slower (rate-limiting) step, while the smaller barrier corresponds to a faster step. This visual model helps connect “fast vs. slow steps” in a mechanism to the idea of a rate-determining step. Source
The pre-equilibrium assumption treats the fast step as being at equilibrium throughout most of the reaction progress.
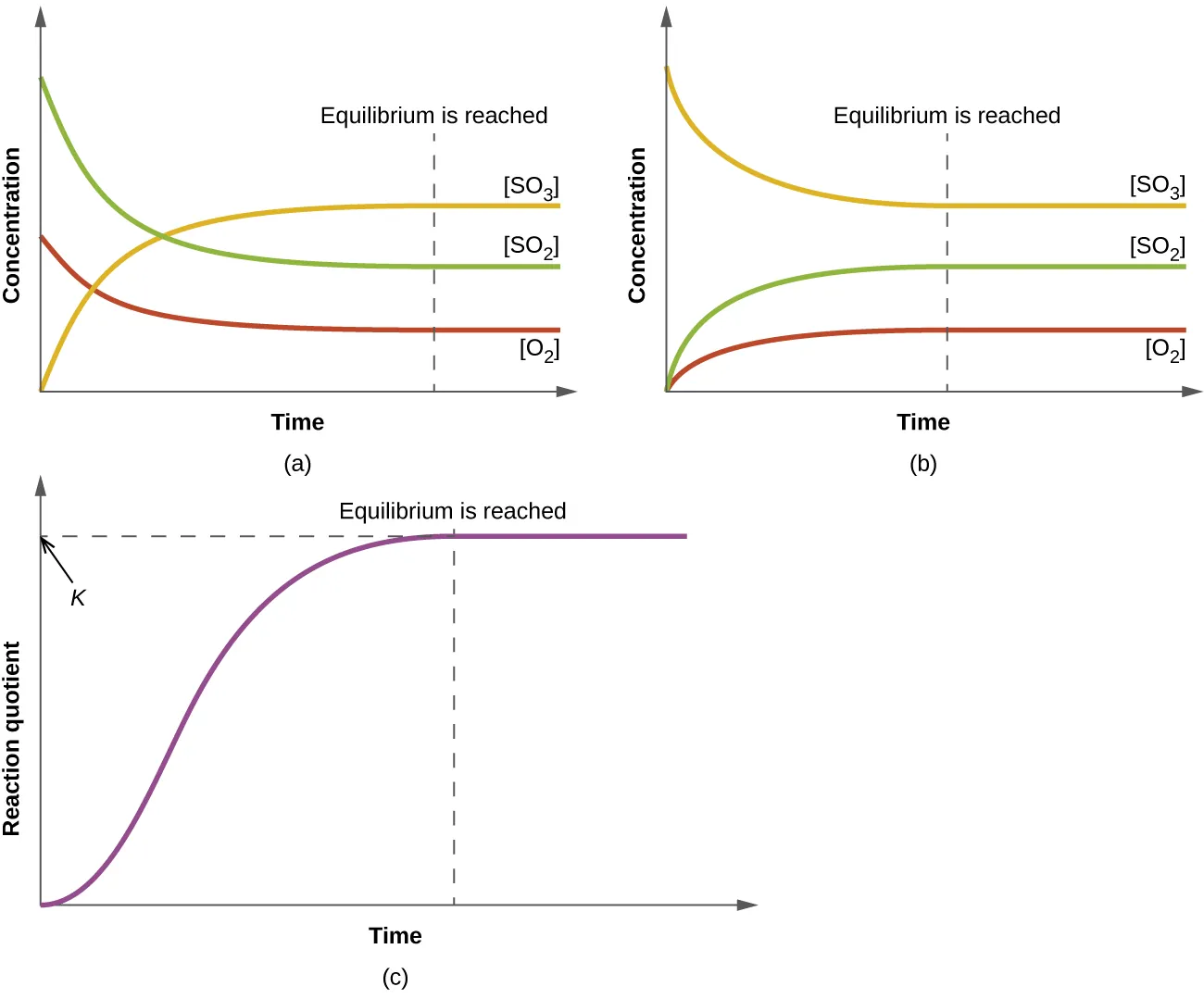
Concentration–time curves illustrating how reactant and product concentrations change and then become constant once equilibrium is reached. The dashed line marks the time after which the system is effectively at equilibrium, so the ratio defining no longer changes. This is the graphical intuition behind treating a fast reversible step as maintaining equilibrium while a slower step controls product formation. Source
Pre-equilibrium assumption: An approximation where a fast reversible step is assumed to maintain equilibrium, allowing an intermediate’s concentration to be written using an equilibrium constant and substituted into the slow-step rate law.
This approach is used specifically when a simple “slow step gives the rate law directly” would leave the rate in terms of an intermediate (a species that is produced and then consumed within the mechanism), which is typically not measurable.
When it is reasonable to use
The first step is much faster (forward and reverse) than the subsequent slow step.
The intermediate produced in the fast step does not build up significantly; it is formed and consumed while the fast equilibrium persists.
Concentrations change slowly enough that the fast step can continually re-establish equilibrium.
How to derive the rate law (workflow)
1) Identify the fast equilibrium and the slow step
Label the fast reversible step (the pre-equilibrium).
Identify the rate-determining step (the slow step) that forms product (or commits reactants to product).
2) Write the rate expression for the slow elementary step
Because the slow step is treated as an elementary step, its rate depends on the concentrations of the reactant species in that step (including any intermediate if it appears there). At this stage, the expression may still contain an intermediate concentration.
3) Write the equilibrium-constant expression for the fast step
Use the balanced equation for the fast reversible step to write in terms of concentrations. Then solve for the intermediate concentration.
4) Substitute to eliminate intermediates
Replace the intermediate concentration in the slow-step rate expression with the expression obtained from the equilibrium step. The final result should involve only:
concentrations of initial reactants (and sometimes products, if they appear in the relevant steps)
rate constants and/or the equilibrium constant
5) Simplify and interpret orders
After substitution, the observed reaction orders can be:
non-integer (less common in AP-style problems but possible in principle)
different from the stoichiometric coefficients of the overall reaction The key requirement is that the final rate law matches what could be measured experimentally.
Algebraic substitution using an equilibrium expression
Typical pattern: fast equilibrium forms an intermediate, slow step consumes it
A common pre-equilibrium mechanism has the form:
Fast:
Slow: products
The slow step suggests the instantaneous rate depends on and , but must be eliminated using the fast equilibrium.
= equilibrium constant for the fast step (unitless when written in activities; often treated as unitless in AP contexts)
= concentration of the intermediate (mol L)
= rate constant for the slow step (units depend on overall order of this step)
rate = overall reaction rate expressed without intermediates (mol L s)
This derived law shows how a mechanism can produce a rate law with dependence on species (, ) that are not in the slow step, because they control the equilibrium level of the intermediate.
What the constants mean after substitution
The product often behaves like an effective rate constant for the overall reaction under the pre-equilibrium approximation.
Temperature or conditions that change (by shifting the equilibrium composition) can change the observed rate even if is unchanged.
Practical checks and common pitfalls
Checks to perform
No intermediates should appear in the final rate law.
The mechanism steps should be consistent with the intended use of :
the step used for must be written as an equilibrium (reversible) step
The derived rate law should be physically sensible:
increasing a reactant concentration that helps form the intermediate should not decrease the rate (unless products appear in the derived expression in an inhibiting way, which requires careful mechanistic justification).
Common pitfalls
Writing the equilibrium expression with incorrect powers (forgetting stoichiometric coefficients in the fast step).
Solving for the wrong species (e.g., expressing instead of and not substituting properly).
Using the overall reaction equation to write ; the used here must correspond to the fast elementary step treated as an equilibrium.
Treating the pre-equilibrium step as irreversible; if it is not reversible, you cannot use an equilibrium constant to relate concentrations in this way.
FAQ
If an early step is labelled fast and reversible and it produces an intermediate that appears in the slow step, pre-equilibrium is usually intended.
Look for wording like “rapid equilibrium” or a given $K$ value for the first step.
You can invert the equilibrium constant.
If $A+B \rightleftharpoons I$ has $K=\frac{[I]}{[A][B]}$, then for $I \rightleftharpoons A+B$, $K'=\frac{1}{K}$.
Yes, if the product appears in the fast equilibrium expression for the intermediate.
Then substituting for the intermediate can introduce a product term in the denominator, corresponding to product inhibition under that mechanism.
It can fail if the “fast” step is not fast enough compared with the slow step, or if conditions change rapidly so equilibrium cannot be maintained.
It also becomes less reliable when multiple coupled equilibria shift significantly during the reaction.
Pre-equilibrium assumes an intermediate is governed by an equilibrium relationship (forward and reverse rates approximately balance).
Steady-state assumes the intermediate’s concentration is approximately constant because its rate of formation roughly equals its rate of consumption, without requiring an equilibrium constant description.
Practice Questions
(3 marks) The mechanism is: Step 1 (fast): Step 2 (slow): products
Derive the rate law in terms of , , and .
Writes rate for slow step: (1)
Writes equilibrium relationship: or (1)
Substitutes to give (1)
(6 marks) The mechanism is: Step 1 (fast): Step 2 (slow): products
(a) Write an expression for for step 1. (b) Obtain an expression for in terms of and . (c) Hence derive the overall rate law in terms of and only.
(a) (2)
correct form (1)
correct square on (1)
(b) Rearranges to (1)
(c) Rate for slow step (1)
Substitution gives (2)
substitution step (1)
final simplified rate law (1)

