AP Syllabus focus: ‘When the first elementary step is not rate limiting, a simple molecularity‑based rate law cannot be used.’
Kinetics often begins by linking the rate law to the slow step of a mechanism.
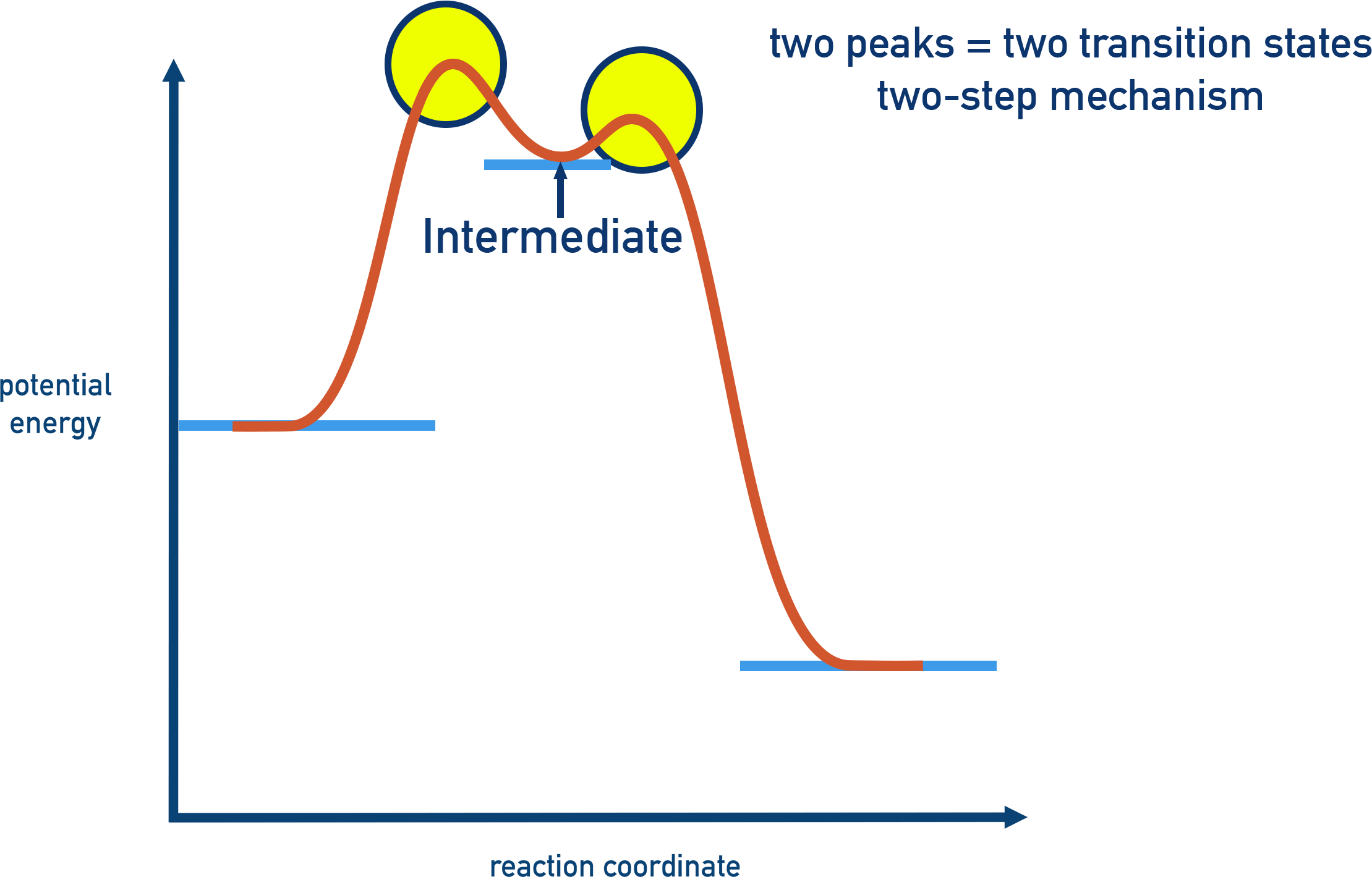
Reaction energy profiles for multistep mechanisms contain multiple peaks (transition states) separated by valleys (intermediates). The tallest peak corresponds to the largest activation barrier and typically identifies the rate-determining step, even when it is not the first elementary step. Source
That shortcut breaks down when the first step is fast, reversible, or otherwise not rate limiting.
The “molecularity shortcut” and where it works
For a mechanism made of elementary steps, each step’s molecularity (how many particles collide) can suggest that step’s rate law. If the first step is the slow step, the overall rate law can often be written directly from that first step’s reactant stoichiometry.
Why that shortcut is attractive
It avoids additional algebra or assumptions.
It uses only the written mechanism and the idea that the slowest step controls the overall pace.
When the first step is not rate limiting
When the first elementary step is fast (especially if it is reversible) and a later step is slow, the rate law for the slow step may depend on species that are not stable, not measurable, or not present in the overall equation.
Rate-determining step: The slowest elementary step in a mechanism; it limits how fast products form under the given conditions.
A key issue is that the slow step may involve an intermediate rather than only initial reactants.
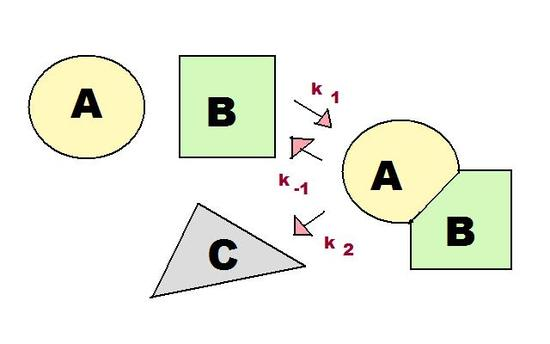
Consecutive-mechanism schematic for a fast equilibrium followed by product formation . The figure emphasizes that the slow-step rate law can depend on the intermediate concentration , which must be related back to measurable reactant concentrations to obtain an overall, testable rate law. Source
Intermediate: A species produced in one elementary step and consumed in a later step; it does not appear in the net balanced equation.
Because intermediates typically cannot be expressed directly from initial conditions without further reasoning, a simple molecularity-based overall rate law “from the slow step” is incomplete.
What the rate expression looks like (and why it’s a problem)
If a later step is slow, you can still write the rate law for that elementary step, but it may include an intermediate concentration such as , which is not an experimental “starting concentration” variable.
= rate constant for the slow elementary step (units depend on overall order)
= intermediate concentration (typically M)
= reaction orders for species in the slow step (from molecularity, for an elementary step)
A complete, testable overall rate law must be written only in terms of measurable reactant concentrations (and constants), so the appearance of signals that additional mechanistic reasoning is required.
Consequences for the observed (experimental) rate law
When the first step is not rate limiting, several outcomes are common:
The observed rate law may include a reactant that is not in the slow step (because that reactant helps establish the intermediate concentration feeding the slow step).
The observed rate law may show non-integer (fractional) orders, reflecting how intermediate concentrations depend on reactant concentrations.
Simply matching the slow step’s reactant coefficients to exponents can give a rate law that is not experimentally consistent, even if the proposed mechanism balances overall.
How to recognise this situation
You are likely in this case if:
The mechanism starts with a step labelled or implied as fast or reversible.
A later slow step uses an intermediate made earlier.
Writing the slow-step rate produces a rate law containing species that are not initial reactants.
What you do instead (conceptually)
You do not abandon the mechanism approach; you refine it:
Treat the fast early step(s) as establishing a relationship between intermediates and reactants (often an equilibrium-like constraint).
Substitute that relationship into the slow-step rate expression to eliminate intermediates.
Compare the resulting rate law form to experimental data to judge whether the mechanism is plausible.
This is why, as the syllabus states, when the first elementary step is not rate limiting, a simple molecularity‑based rate law cannot be used: the overall rate law requires connecting intermediate concentrations to measurable quantities.
FAQ
Yes. A reactant used in a fast step can control how much intermediate is available for the slow step, so it can appear in the observed rate law even if absent from the slow step.
Intermediates are typically short-lived and not part of initial conditions. Rate laws from experiments are expressed using measurable concentrations (usually initial reactants), so intermediates must be eliminated.
No. Even if it is not rate determining, it can set the intermediate concentration profile that “feeds” the slow step, indirectly influencing the overall rate dependence.
Yes. When an intermediate concentration depends on reactants through an equilibrium-like relationship, substitution can yield fractional powers in the final rate expression.
If the experimentally determined rate law contradicts the molecularity-based prediction from step 1 (e.g., different reactant dependence), it indicates a later step likely controls the rate.
Practice Questions
A proposed mechanism has a fast first step and a slow second step. State why you cannot generally write the overall rate law by using only the molecularity of the first step.
First step is not rate determining, so it does not control the overall rate (1).
The slow-step rate law may involve an intermediate and must be rewritten in terms of measurable reactant concentrations; thus the first-step molecularity is insufficient (1).
Consider a two-step mechanism: Step 1 (fast): ; Step 2 (slow): .
(a) Write the rate law for the slow step.
(b) Explain why this is not yet an overall rate law in terms of measurable reactants.
(c) State, in words, what additional idea is needed to convert it into an experimentally testable rate law.
(a) (2: correct form 1, correct species/exponents 1)
(b) Includes , an intermediate not directly measurable from initial reactant concentrations / not in overall equation (1)
(c) Use a fast-equilibrium (or similar) relationship to express in terms of and , then substitute (2: mention relationship 1, substitution/elimination of 1)

