AP Syllabus focus: ‘An elementary reaction occurs only when reactant particles collide effectively, initiating bond breaking and bond making.’
Collision theory connects microscopic particle motion to macroscopic reaction rates. It explains why reactions require collisions and why only a small subset of collisions actually produce products in an elementary step.
Collision theory: the core idea
Collision theory treats reacting particles (atoms, ions, or molecules) as constantly moving and colliding. A reaction can occur only during a collision, because collisions are the moments when electron clouds interact strongly enough to rearrange bonds.
Collision theory: A model stating that reactions occur when reactant particles collide, and that the reaction rate depends on how often collisions occur and how many collisions lead to product formation.
In this subtopic, the emphasis is on effective collisions: collisions that directly result in chemical change (products), rather than particles simply bouncing apart unchanged.
What makes a collision “effective”?
An elementary reaction is a single molecular event (one step). In such a step, reactants must physically meet; however, most collisions are ineffective and do not form products.
Effective collision: A collision between reactant particles that results in product formation by initiating bond breaking and bond making.
An effective collision is the instant where the system begins to move from “reactants exist” to “new substances exist.” In molecular terms, an effective collision:
Brings reactants close enough for significant electron cloud overlap
Enables the weakening and breaking of certain bonds in reactants
Simultaneously enables the formation of new bonds that define products
Because bond breaking and making occurs on extremely short time scales, effective collisions are rare compared with total collisions.
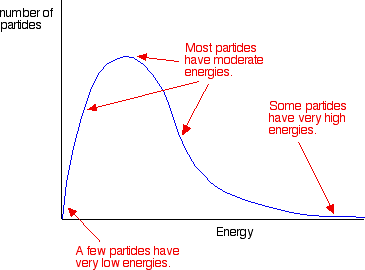
A Maxwell–Boltzmann distribution shows how particle kinetic energies are spread out at a given temperature. The vertical line marks the minimum energy needed for reaction; only the small area to the right represents collisions that can be energetically “effective.” This makes the “fraction of effective collisions” idea concrete: most collisions simply do not have enough energy to initiate bond changes. Source
Connecting collisions to reaction rate
Reaction rate increases when the number of effective collisions per unit time increases. Conceptually, the rate is controlled by two linked ideas:
Collision frequency: how often particles collide
Effectiveness fraction: what portion of those collisions actually cause reaction
= Reaction rate (e.g., )
= Collisions per unit time (depends on particle density and motion)
= Unitless proportion of collisions that form products
This relationship is qualitative for AP Chemistry: it tells you what must increase for a reaction to speed up, without requiring detailed kinetic theory mathematics.
Why collision frequency changes (without going beyond the syllabus)
Even before learning the detailed reasons collisions fail, collision theory still supports several core interpretations:
More particles in a given volume means more opportunities to collide, so reaction rate tends to increase when reactant particle density is higher.
Faster-moving particles collide more often, so conditions that increase particle motion generally increase collision frequency.
In many real systems, changing the physical state or mixing can change how often reactants encounter one another, altering the effective collision count per second.
These ideas are used to justify rate changes using particle-level reasoning, while later topics address the specific requirements that decide whether a collision is effective.
What “initiating bond breaking and bond making” means
During an effective collision, reactants reach a configuration where bonds are unstable enough to begin changing.
From a particle perspective:
Existing bonds are stressed as atoms are pushed into new proximity
Electron density shifts, allowing new attractions to develop between different atoms
A short-lived, high-energy arrangement forms at the instant bonds begin to reorganise (the detailed energetic picture is developed elsewhere)
The key takeaway for this page is the syllabus statement: an elementary reaction occurs only when reactant particles collide effectively, and that effectiveness is defined by the collision’s ability to start the bond changes required to make products.
FAQ
It is most straightforward in gases, where particles move freely and collisions are well-defined.
In liquids and solids, “encounters” can involve solvent cages, diffusion limits, or surface contacts, so collision ideas still apply but are less idealised.
A collision implies a brief, direct impact event.
An encounter can mean any close approach long enough for interaction (especially in solution), which may include multiple micro-collisions before separation.
At constant temperature, higher pressure increases number density.
More particles per volume increases collision frequency, which can increase the number of effective collisions per second.
It is a correction factor representing the fraction of collisions with suitable geometry to react.
It helps quantify why only some collisions are effective, beyond collision frequency alone.
Stirring reduces concentration gradients and improves mixing.
This increases reactant encounters at the reaction sites (especially at interfaces), raising the number of effective collisions occurring per unit time.
Practice Questions
(1–3 marks) In terms of collision theory, state what must happen for an elementary reaction to produce products.
Reactant particles must collide (1)
The collision must be effective / lead to product formation (1)
Effective collision initiates bond breaking and bond making (1)
(4–6 marks) A student claims: “If you double the number of collisions per second, the reaction rate must double.” Using collision theory, assess this claim.
Rate depends on number of effective collisions per unit time, not total collisions alone (1)
State that many collisions are ineffective (1)
Explain that doubling collision frequency could increase rate only if the fraction of effective collisions remains constant (1)
Note that if the fraction effective changes, rate may increase by more or less than double (1)
Use a clear proportionality statement such as (1)

