AP Syllabus focus: ‘Elementary steps requiring a simultaneous collision of three or more particles are rare.’
Reaction mechanisms are built from elementary steps, and most real steps involve one or two reacting particles. This page explains why true three-particle elementary collisions are statistically and physically unlikely.
What “termolecular” means in kinetics
Molecularity is about an elementary step, not the overall reaction
Molecularity: The number of reactant particles that must collide simultaneously in a single elementary step.
A termolecular elementary step would require three reactant particles to participate in the same collision event, at the same instant, to form products directly.
Termolecular (elementary step): An elementary step with molecularity 3, requiring a simultaneous collision of three reactant particles.
Why simultaneous three-particle collisions are unlikely
Statistical (probability) argument
For a reaction in a gas or solution, collisions happen constantly, but the chance that two specific particles meet at the same place and time is already limited. The chance that a third particle is also in the same tiny region at the same instant is far smaller.
A bimolecular collision depends on two particles encountering one another.
A termolecular collision effectively depends on a successful bimolecular collision occurring while a third particle is also present in the same “collision volume.”
Because this is a compounded requirement, the frequency of true three-body encounters is extremely low at typical concentrations and pressures used in laboratories.
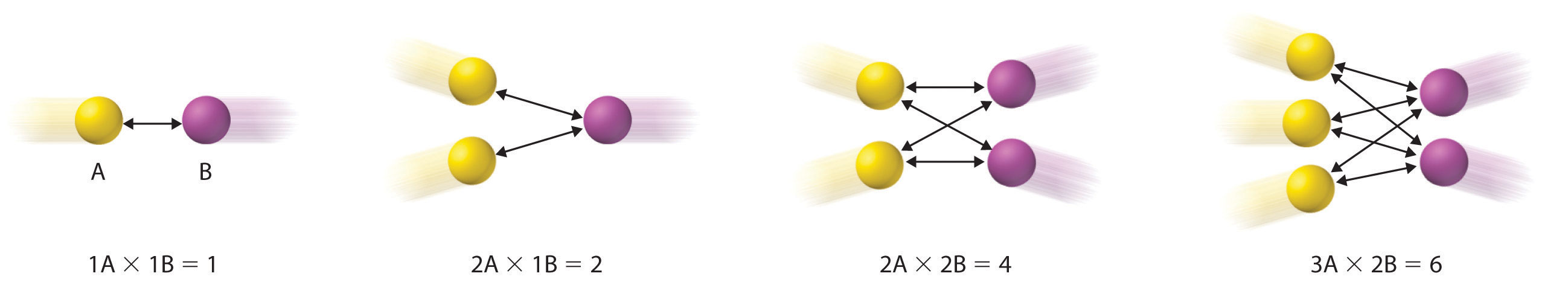
Diagram showing how the number of possible A–B collisions per unit time scales with the product of the numbers of A and B particles, not their sum. This helps explain why adding another simultaneously required reactant (moving toward a three-body encounter) makes successful events much less frequent under typical conditions. Source
Geometric (orientation) constraint
Even if three particles are momentarily close together, they must have a specific relative orientation to allow the correct bonds to break and form in a single concerted event.
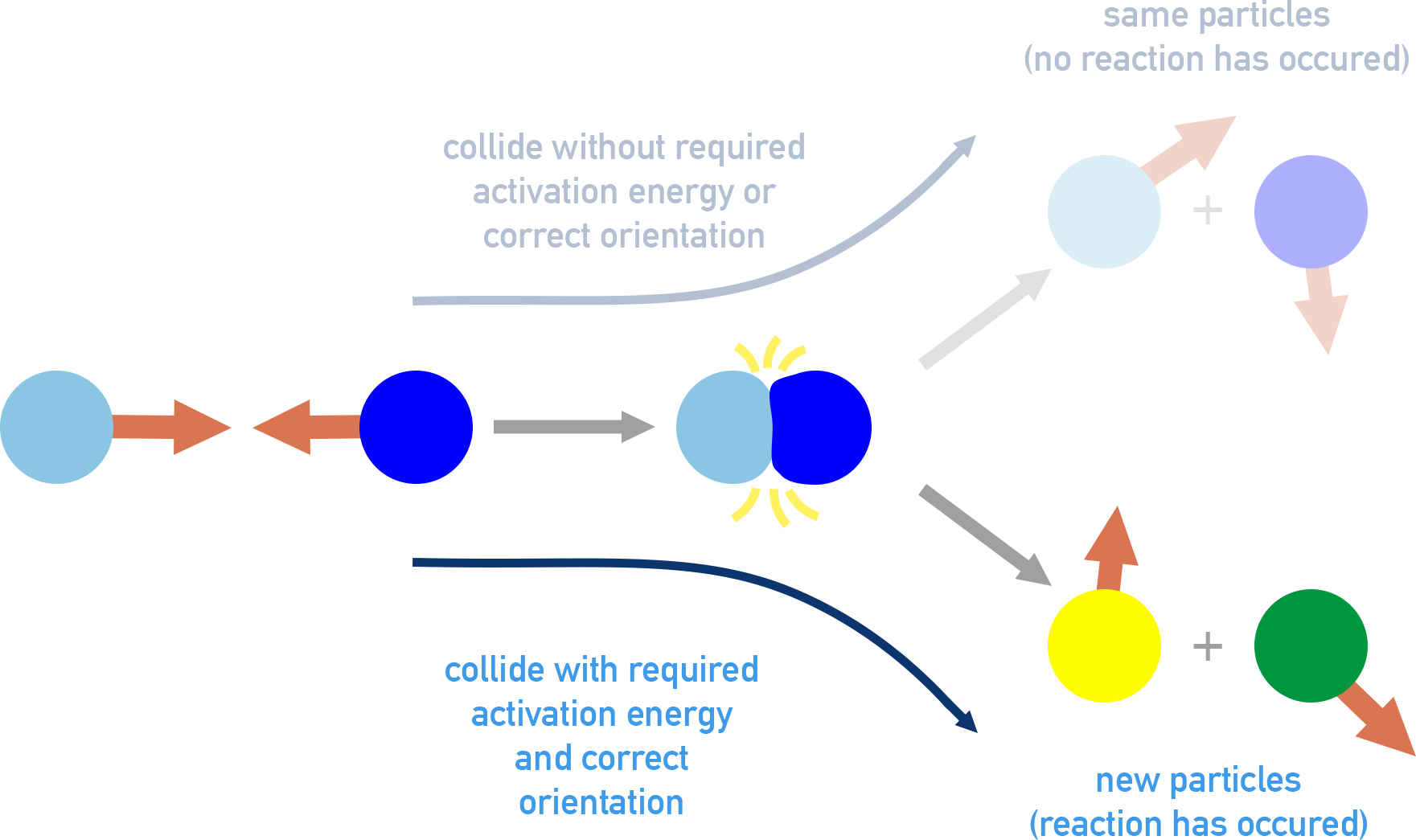
Collision-model diagram illustrating that only collisions with the correct molecular orientation lead to reaction, while misaligned collisions do not. It supports the idea that, as the number of participants increases, the fraction of encounters with the required geometry drops sharply, making termolecular elementary steps rare. Source
With two particles, there are fewer positional arrangements that matter.
With three particles, there are many more ways to be “almost right” but still nonreactive, making successful geometry rarer.
Energy and momentum complications
A single collision event must channel energy into the correct bond-making/bond-breaking motions. With three participants, energy can be redistributed among more degrees of freedom, so it is harder for the system to follow the precise pathway needed for product formation in one step.
More particles can mean more ways to “lose” the necessary energy into translation/rotation/vibration rather than reaction progress.
This further reduces the fraction of three-body encounters that are genuinely reactive.
What actually happens instead (mechanistic implication)
Apparent termolecular stoichiometry is usually multi-step
Many reactions that look like “three reactants go to products” in the overall balanced equation proceed through multiple elementary steps involving bimolecular events, often with a short-lived intermediate.
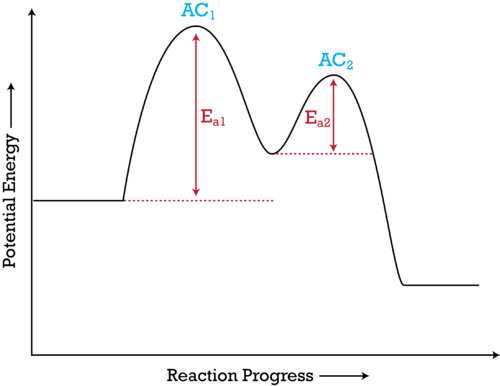
Potential-energy diagram for a two-step mechanism showing two distinct transition states (peaks) and a reaction intermediate (valley). It reinforces that a complex overall reaction can proceed through successive elementary steps, each with its own activation energy barrier, rather than as one highly unlikely concerted event. Source
A fast step may form an intermediate from two particles.
A subsequent step may have that intermediate react with a third particle.
Because each step only requires one collision between two species, the mechanism is much more plausible than a single three-body event.
Recognising “rare” vs “impossible”
The syllabus wording is deliberate: termolecular elementary steps are rare, not forbidden. Under special conditions (very high pressures, very high concentrations, or highly constrained environments), three-body collisions can occur, but they are uncommon enough that AP Chemistry typically treats them as exceptions.
Common student pitfalls
Confusing molecularity (applies to an elementary step) with stoichiometric coefficients (apply to the overall reaction).
Assuming any overall reaction with three reactant molecules must contain a termolecular elementary step.
Treating “rare” as “never,” rather than as a probability-and-conditions statement.
FAQ
Generally, higher number density helps, so increased pressure (gases) or high concentration (solutions) can raise three-body encounter frequency.
However, solvent “caging” in liquids can also alter encounter dynamics, so it is system-dependent.
$M$ is a collision partner that can absorb excess energy and/or momentum, stabilising a newly formed product.
It is regenerated, so it is not consumed overall, but it must be present for the stabilisation event.
If two fast steps occur in sequence and one involves a short-lived intermediate, the observed rate behaviour may mimic higher-order dependence under certain conditions.
This can create the illusion of a single three-reactant event.
Yes. Confinement (e.g., pores, active sites) can keep multiple species close together, effectively increasing local concentration and enforcing geometry.
This can make multi-particle encounters less improbable than in bulk phases.
Because “rare” depends on conditions and chemistry. Some specialised systems at high pressure or with strong stabilisation can involve genuine three-body events.
Mechanistic proposals weigh plausibility, not absolute impossibility.
Practice Questions
Q1 (2 marks): Explain, in terms of collision likelihood, why termolecular elementary steps are rare compared with bimolecular steps.
States that three particles must collide simultaneously in the same place and time (1 mark).
Explains that this is much less probable/frequent than two-particle collisions (1 mark).
Q2 (5 marks): A student proposes the elementary step for a mechanism. Give two distinct reasons this step is unlikely to be elementary, and describe what a more plausible alternative mechanism could involve.
Reason 1: simultaneous three-body collisions are statistically very unlikely at typical conditions (1 mark).
Reason 2: additional orientation/energy redistribution constraints make reactive three-body events even less likely (1 mark).
Describes a plausible alternative: two-step sequence of bimolecular collisions (1 mark).
Mentions formation of an intermediate from two reactants (1 mark).
Mentions the intermediate then reacting with the third reactant in a subsequent step (1 mark).

