AP Syllabus focus: ‘The proportionality constant k is the rate constant; its value depends on temperature, and its units depend on overall reaction order.’
Kinetics links measured rates to chemical conditions using the rate constant, k. This page explains what k represents, how its units are determined from a rate law, and why temperature changes its value.
Meaning of the rate constant, k
In a rate law, k is the proportionality factor that connects the measured reaction rate to reactant concentrations. For a specific reaction (and a specific mechanism), k is constant only when conditions are fixed, especially temperature.
Rate constant (k): The proportionality constant in a rate law that sets the scale of the reaction rate for given conditions, particularly temperature.
Key interpretation points:
A larger k means a faster reaction at the same reactant concentrations.
k is not determined from the balanced overall equation alone; it is obtained experimentally from rate data.
k does not change when concentrations change during a single run; instead, the rate changes because concentrations appear in the rate law.
k can change if conditions change; for AP Chemistry, the most emphasized dependence is temperature.
How k appears in a rate law
A general experimental rate law has the form below; the exponents come from experiment, not stoichiometric coefficients (unless the step is elementary, which is treated elsewhere).
= reaction rate (typically )
= rate constant (units depend on overall order)
= molar concentrations,
= experimentally determined reaction orders (unitless)
The overall reaction order is the sum of the concentration exponents in the rate law (here, m + p). This matters because k must supply whatever units are needed so that the right side has the same units as the rate.
Units of k (depend on overall reaction order)
Because rate is commonly reported in , the units of k depend on the overall order, n. In general:
Rate has units
The concentration term has units
Therefore, k must have units that make
= units of the rate constant
= overall reaction order (sum of exponents in the rate law)
Common AP Chemistry cases (rate in ):
Zero order (n = 0): k has units
First order (n = 1): k has units
Second order (n = 2): k has units
Third order (n = 3): k has units
Practical notes:
Always check the units of the reported rate; if time is in minutes, k will carry (first order) or include with appropriate concentration powers (other orders).
Concentration may also be expressed as ; and are equivalent.
Temperature dependence of k
The rate constant k is temperature-dependent: increasing temperature typically increases k, often dramatically.
At higher temperature, a greater fraction of collisions (or molecular encounters) have sufficient energy to proceed through the highest-energy point along the pathway, so successful events occur more frequently per unit time.
What to know qualitatively for AP Chemistry:
At a higher temperature, k is usually larger, so the reaction is faster even at the same concentrations.
The relationship is not linear; modest temperature increases can cause large increases in k.
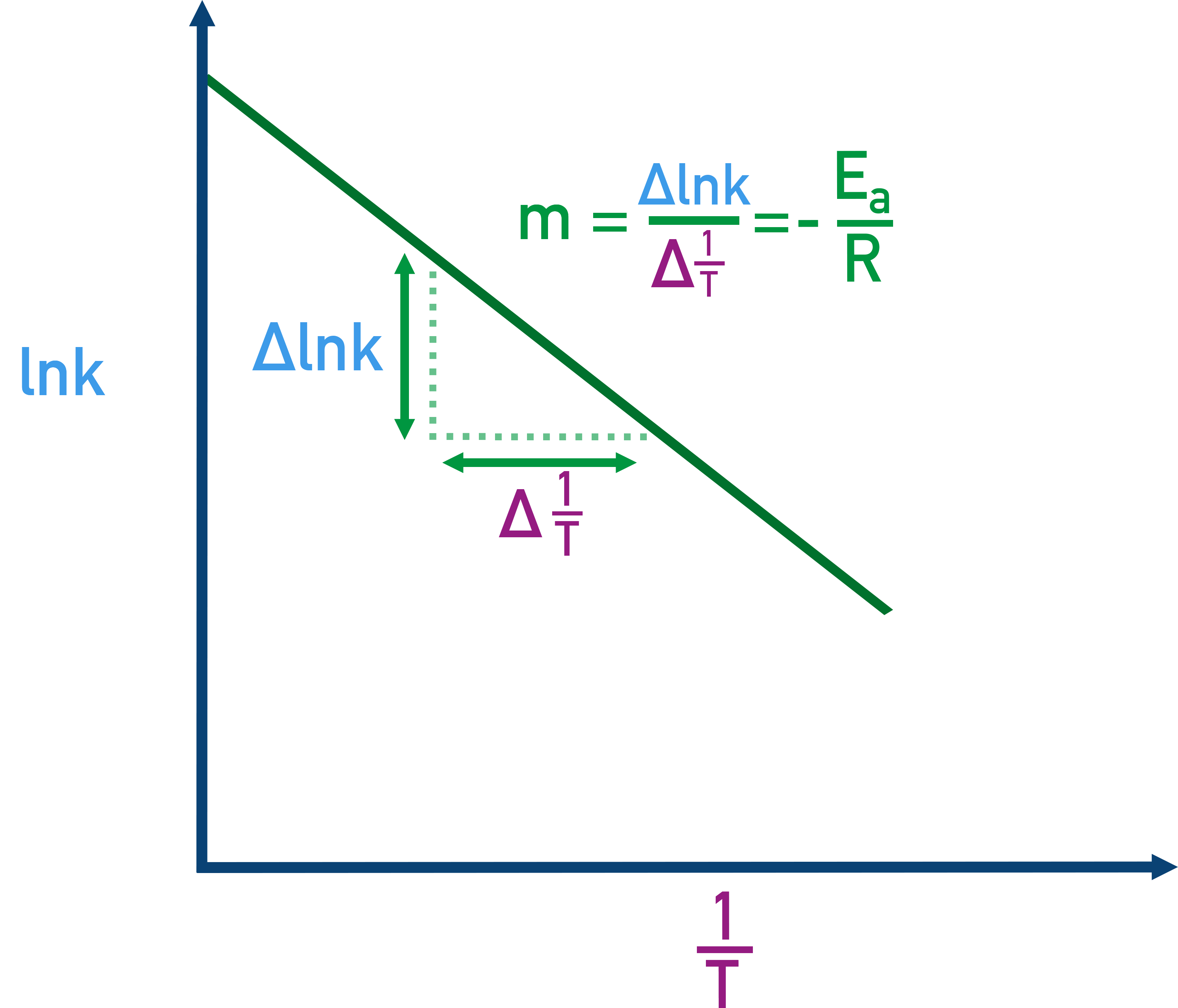
Arrhenius plot schematic showing that plotting versus yields a straight line. The diagram highlights how the slope is calculated and that the slope equals , linking temperature sensitivity of to activation energy. Source
When comparing kinetic data (or k values), temperature must be stated; k values at different temperatures should not be treated as directly comparable without noting that change.
Common pitfalls when using k
Treating k as a universal constant: it is constant only for a particular reaction at a particular temperature (and stated conditions).
Assuming the balanced equation gives the units of k: the experimental rate law determines n, which determines k’s units.
Mixing time units: seconds vs minutes changes the numerical value of k and must be tracked in units.
FAQ
k reflects the specific reaction pathway and energetic barriers.
Different reactants/bond changes lead to different probabilities of successful events per unit time, even if the concentration dependence looks similar.
Only the time unit changes.
For first order: $k(\mathrm{min^{-1}})=60,k(\mathrm{s^{-1}})$. For other orders, the concentration part is unchanged; only the $\mathrm{s^{-1}}$ factor converts.
Because $\mathrm{M}=\mathrm{mol,L^{-1}}$.
So $\mathrm{M^{-1},s^{-1}}=\mathrm{L,mol^{-1},s^{-1}}$; they are equivalent ways to express the same unit.
Not directly.
If temperature and concentrations are the same, k is unchanged; changing volume only matters insofar as it changes concentrations or conditions.
At fixed temperature, k is typically treated as independent of pressure for an elementary ideal-gas description.
However, changing total pressure can change reactant concentrations (via partial pressures), which changes the rate even if k is unchanged.
Practice Questions
(2 marks) For the rate law , determine the units of if rate is measured in .
States overall order (1)
Gives (or ) (1)
(5 marks) A reaction has rate law . In one experiment at constant temperature, doubling doubles the rate. In a separate experiment, the temperature is increased while is kept the same and the measured rate increases. Explain, using k, why each change affects the rate, and state the units of k.
Identifies first-order dependence: rate proportional to (1)
Explains doubling doubles rate because is to the first power (1)
States k remains constant when only concentration changes (same temperature/conditions) (1)
Explains increasing temperature increases k, causing a higher rate at the same (1)
Gives units of k for first order: (1)

