AP Syllabus focus: ‘A rate law relates reaction rate to reactant concentrations, each raised to an exponent.’
Reaction rates vary dramatically with conditions. A rate law is the key mathematical link between measured reaction speed and reactant concentrations, allowing chemists to predict how changing conditions affects how fast a reaction proceeds.
What a Rate Law Represents
A rate law expresses how the reaction rate depends on the concentrations of species involved (most often reactants). It is an experimentally determined relationship, not something you can reliably deduce from the overall balanced equation.
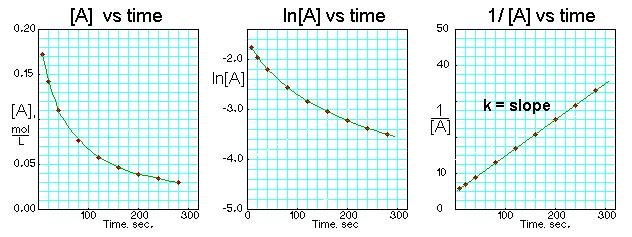
These three plots illustrate how reaction order is identified experimentally using “linearized” integrated-rate-law graphs. For a second-order reaction in , the vs. time plot is linear and its slope gives , while vs. time and vs. time are non-linear. This is a visual reminder that the order (the exponent in the rate law) is determined from data, not from the balanced equation. Source
Rate law: An equation that relates the reaction rate to the concentrations of reactants (and sometimes other species), each raised to a power determined by experiment.
Rate laws are most useful because they let you:
Predict how rate responds when a concentration changes.
Compare how strongly different reactants influence the rate.
Provide constraints on possible molecular-level explanations (mechanisms) without requiring you to know the mechanism to write the law.
General Form and Meaning of Each Part
Differential Rate Law Form
= reaction rate (typically expressed as a concentration change per unit time)
= rate constant (a proportionality constant for a given reaction at a fixed temperature)
= molar concentrations of reactants
= experimentally determined exponents that describe how sensitively the rate depends on each concentration
The exponents show concentration dependence:
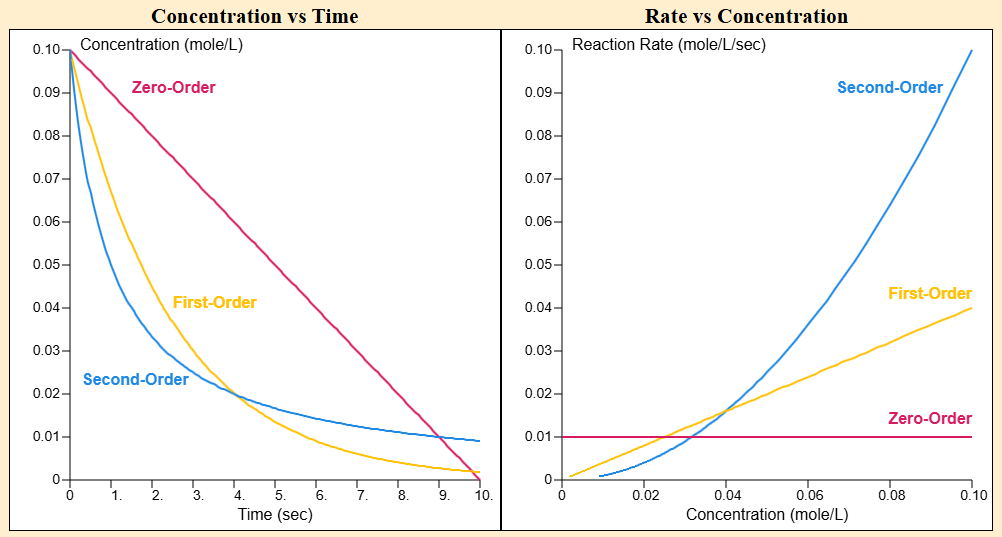
This diagram compares how zero-, first-, and second-order rate laws change the shape of kinetic data. On the rate–concentration plot, a zero-order law is flat, a first-order law is linear through the origin, and a second-order law curves upward, visually encoding the dependence . Paired with concentration–time curves, it helps you connect an exponent in the rate law to an experimentally observable pattern. Source
If , the rate is directly proportional to .
If , doubling increases the rate by a factor of .
If , changing does not change the rate (the factor ).
What to Include (and Not Include) in a Rate Law
Rate laws are commonly written using reactant concentrations.
Do not assume the exponents match stoichiometric coefficients in the overall equation; they often do not.
A species that does not appear in the overall equation can still affect rate (for example, if it participates in the pathway), but any such inclusion must be justified by experimental evidence.
Interpreting Rate Laws as Proportional Relationships
A rate law is a compact statement of proportionality:
The rate is proportional to each reactant concentration raised to its exponent.
Changing one concentration scales the rate by a predictable multiplicative factor, holding other concentrations constant.
Useful ways to reason from a rate law (without doing full calculations):
If changes by a factor , the rate changes by a factor (with all else constant).
If multiple reactants change simultaneously, multiply the separate factors (for example, ).
Experimental Nature of Exponents
The exponents in a rate law must come from data, because real reactions may involve multiple steps, temporary species, or pathways that make the observed concentration dependence non-obvious from the overall reaction alone. In practice, a proposed rate law is acceptable only if it matches measured rates across changing concentrations under controlled conditions.
Common student pitfalls to avoid:
Using the balanced equation coefficients as exponents by default.
Forgetting that changing concentration affects rate multiplicatively, not additively.
Treating as a universal constant; it is constant only for a specific reaction under specific conditions (especially temperature).
FAQ
For many reactions measured under typical conditions, the forward rate is controlled by how often reactant particles encounter each other in the relevant pathway.
Product terms may become important in situations such as strong reverse reactions or inhibition, but this must be supported by data.
Yes, if experiments show the rate depends on that species.
This can happen when a substance participates in the pathway but is regenerated later (for example, behaving like a catalyst) or affects the reactive form of a reactant.
A zero exponent means changing that species’ concentration does not change the measured rate under those conditions.
This often indicates the reaction is limited by something else (for example, saturation of a reactive site), but the rate law alone does not specify the cause.
Either can be used, provided you are consistent and the constant is defined accordingly.
For gases, partial pressures are often proportional to concentration at fixed temperature, so a pressure-based form can be written with a correspondingly defined $k$.
Yes, but the numerical value of $k$ depends on the unit choices used in the rate law.
To avoid confusion, keep concentration units consistent within a problem and interpret $k$ only within that chosen unit system.
Practice Questions
Q1 (1–3 marks) For the reaction with rate law , what happens to the rate if is doubled while is held constant?
Doubling changes rate by (1)
Rate increases by a factor of 4 (1)
Q2 (4–6 marks) A reaction has rate law . (a) Write an expression for in terms of the measured rate, , and . (b) State the factor by which the rate changes if is tripled and is halved simultaneously. (c) State whether the exponents in the rate law can be assumed to match coefficients in the overall balanced equation, and justify your answer.
(a)
(1)
(b)
Tripling gives factor (1)
Halving gives factor (1)
Combined factor (1) (c)
(c)
Cannot assume a match (1)
Exponents are determined experimentally and reflect the rate’s concentration dependence, which may not follow overall stoichiometry (1)

