AP Syllabus focus: ‘In surface catalysis, reactants or intermediates bind to a surface, forming bound intermediates and introducing additional elementary steps.’
Surface catalysis explains how solid surfaces (often metals or metal oxides) accelerate reactions in gases or solutions by temporarily binding reactants. This binding creates new, surface-specific steps that can make the overall pathway faster.
What surface catalysis is (and why it changes the mechanism)
Surface catalysis is typically heterogeneous catalysis: the catalyst is in a different phase than the reactants. The key idea is that the surface provides binding sites that stabilize reactants/intermediates and reorganise reactants into a more reactive arrangement.
Surface catalysis: A catalytic process in which reactants (or intermediates) adsorb to a solid surface, react via surface-bound steps, then desorb as products, leaving the surface available again.
Because adsorption and desorption are real chemical events, additional elementary steps appear in the mechanism that do not exist in the uncatalysed (gas-phase or solution-phase) pathway.
Core steps in a surface-catalysed pathway
1) Adsorption: reactants bind to the surface
Adsorption creates surface-bound intermediates and increases the chance of productive interactions.
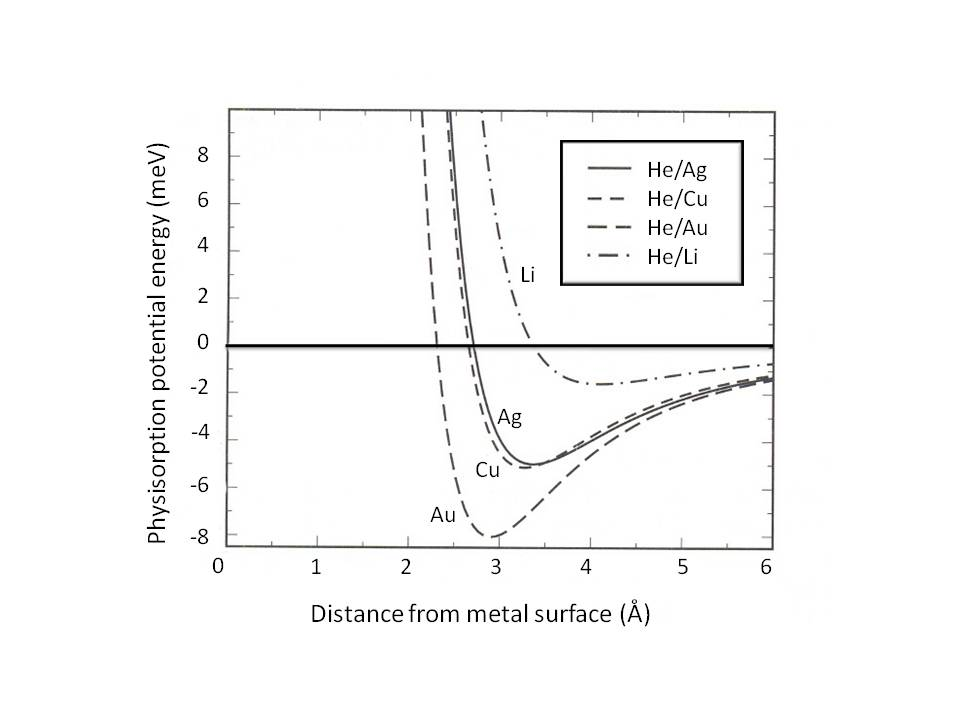
Physisorption potential-energy curve showing how interaction energy varies with distance from a surface, producing an attractive well at intermediate separation. The minimum corresponds to a bound (adsorbed) state, illustrating how adsorption can trap reactants at the surface and set up surface-bound intermediates for subsequent elementary steps. Source
Adsorption can be:
Physisorption (weak intermolecular attractions; usually less specific)
Chemisorption (stronger bonding; often essential for catalysis)
Binding can:
bring reactants into close proximity
weaken specific bonds in the reactant (making reaction easier)
hold molecules in a favourable orientation for bond rearrangement
2) Surface reaction: new elementary steps occur on the surface
Once bound, reactants can react through one or more surface elementary steps. These steps may involve:
reaction between two adsorbed species
reaction between an adsorbed species and one from the gas/solution phase
migration of adsorbed species between nearby sites before reacting
= an empty surface site
= species bound to the surface
= product released; site is regenerated
Asterisks are bookkeeping: they represent a finite number of active sites, so surface coverage can limit the rate.
3) Desorption: products leave and sites are regenerated
For catalysis to continue, products must detach so active sites reopen.
If products bind too strongly, site blocking occurs and the catalyst can “poison” itself.
If reactants bind too weakly, adsorption is insufficient and surface reactions are rare.
What controls the rate in surface catalysis
Active sites and surface area
Only atoms at/near the surface can act as catalytic sites.
Increasing surface area (e.g., finely divided metal, porous supports) typically increases the number of accessible sites and can increase rate.
Not all surface atoms are equally reactive; defects, edges, and corners often act as higher-activity sites.
Surface coverage and competition for sites
Multiple species may compete for the same sites.
Strongly adsorbing impurities can cause catalyst poisoning by occupying sites without producing product.
At high reactant pressure/concentration, the surface may become saturated; then increasing reactant amount may not increase rate much because few empty sites remain.
Energy landscape on the surface
A surface pathway can be faster because adsorption and surface bonding can stabilise key high-energy arrangements (surface-bound intermediates), effectively lowering barriers for bond breaking/making along the new sequence of steps.
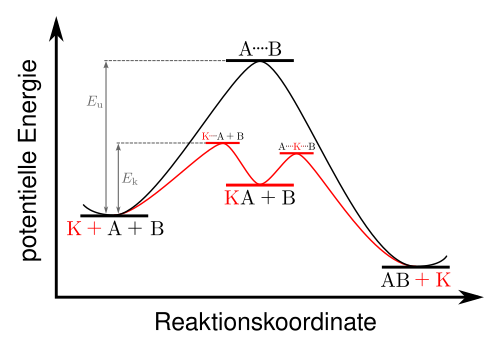
Potential-energy diagram comparing an uncatalyzed pathway to a catalyzed pathway with a lower activation barrier. In surface catalysis, adsorption can stabilize key configurations along the reaction coordinate, effectively reducing the energy required to reach the transition state and speeding up the overall rate. Source
Common features in real systems (AP-relevant examples)
Many industrial catalysts rely on surface catalysis, such as:
metal catalysts (Pt, Pd, Ni) for hydrogenation and pollution control
metal oxides for oxidation reactions
These systems illustrate the syllabus focus: reactants/intermediates bind to a surface, creating bound intermediates and additional elementary steps before products desorb.
FAQ
Adsorption is binding at the surface (sites become occupied).
Absorption is uptake into the bulk of the material. Surface catalysis relies on adsorption because the reactive events occur at surface sites.
Active sites are locations where adsorption and reaction are especially favourable.
They often occur at defects, steps, edges, or particular crystal faces that stabilise key surface-bound intermediates.
Activity can drop if sites are blocked faster than they are freed.
Common causes include poisoning (impurities bind strongly), coking (carbonaceous deposits), or products that desorb too slowly.
Supports disperse metal into small particles, increasing accessible surface area.
They can also reduce particle sintering and sometimes modify adsorption strength by altering the metal’s electronic environment.
They use surface-sensitive methods such as infrared spectroscopy of adsorbates, temperature-programmed desorption, and X-ray photoelectron spectroscopy.
These techniques can distinguish reactants, intermediates, and poisons by their characteristic signals.
Practice Questions
(2 marks) Explain how a solid catalyst can increase reaction rate via surface catalysis, referring to what happens to reactant particles on the surface.
Reactants adsorb/bind to the catalyst surface forming surface-bound species/intermediates. (1)
The surface provides an alternative pathway with additional steps (e.g., surface reaction then desorption) that increases the rate (e.g., more effective collisions/orientation or lower barrier). (1)
(5 marks) A reaction is catalysed by a metal surface. Describe a plausible surface-catalysed mechanism using adsorption and desorption steps, and explain two reasons why the rate might decrease over time during operation.
Includes adsorption of to form . (1)
Includes adsorption of to form . (1)
Includes a surface reaction step forming from adsorbed species (or equivalent). (1)
Includes desorption step showing site regeneration. (1)
Two valid causes of rate decrease, any two of: site blocking by strongly adsorbed product (), poisoning by impurities occupying sites, surface saturation reducing empty sites, sintering/aggregation reducing surface area/active sites. (1)

