AP Syllabus focus: ‘In a catalytic mechanism, the catalyst’s net concentration remains constant: it may be consumed in the rate‑determining step and regenerated in a later step.’
Catalysts participate directly in reaction mechanisms without being used up overall.
Understanding how a catalyst can be temporarily consumed yet regenerated clarifies why small amounts can speed reactions repeatedly and why it does not appear in the net chemical change.
Core idea: net amount constant, even if used in a step
What “regenerated” means in mechanisms
Catalyst: A substance that increases reaction rate by participating in the mechanism but is regenerated so its net amount remains constant after a complete catalytic cycle.
A catalyst can appear as a reactant in one elementary step and as a product in a later step. Because mechanisms are added stepwise to obtain an overall reaction, the catalyst cancels algebraically and is absent from the net equation.
How the net amount stays constant
During the reaction, some catalyst molecules may exist in “bound” or “altered” forms (often as catalyst-containing species).
After completion of each catalytic cycle, those catalyst-containing species release or re-form the original catalyst.
Therefore, catalyst is not a limiting reagent for the overall stoichiometry (though it can limit rate if very scarce).
Stoichiometric bookkeeping: why catalysts cancel
When you sum elementary steps, species that are produced and then later consumed cancel, leaving only reactants and products of the overall reaction. Catalysts behave like this by design.
= catalyst species (same chemical identity before and after a full cycle)
= catalyst-containing species formed and later consumed
= (cancels when steps are added)
In this bookkeeping sense, “net concentration remains constant” means the catalyst is not consumed overall, even if it is transiently tied up in intermediate catalyst-containing species during the reaction.
Rate-determining step: “consumed” does not mean “used up”
The syllabus emphasises that the catalyst may be consumed in the rate-determining step and regenerated in a later step. Interpreting “consumed” correctly is crucial:
In a single elementary step, “consumed” means it is on the reactant side of that step, so its free form decreases at that instant.
Later step(s) return it on the product side, restoring free catalyst.
Over many cycles, individual catalyst molecules are repeatedly consumed/regenerated, so the macroscopic amount of catalyst remains approximately constant provided it is not lost to side processes.
Practical implications for experiments (catalyst amount vs. reaction progress)
Adding catalyst typically changes the time scale of reaching a given extent of reaction, but does not change the overall stoichiometric amounts of reactants converted to products at completion.
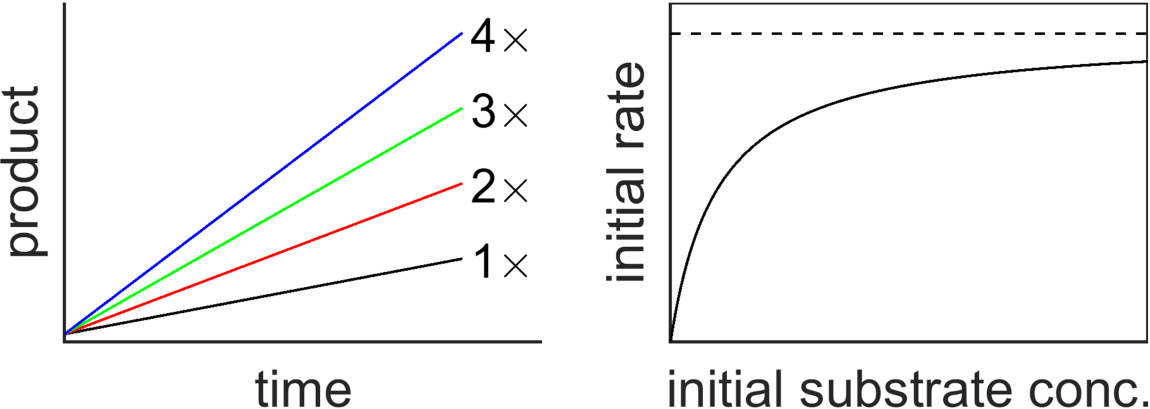
Kinetics plots illustrating how more catalyst increases reaction rate. On the left, larger catalyst (enzyme) concentration produces product faster (greater slope of product vs. time). On the right, the initial rate rises with substrate concentration and approaches a saturation limit, emphasizing that catalysts affect rate behavior rather than changing the net stoichiometry. Source
Because the catalyst is regenerated, a small initial amount can facilitate formation of a much larger amount of product over time.
If catalyst is removed from the system (e.g., separated, poisoned, or otherwise deactivated), the reaction rate can drop because fewer active catalyst molecules are available to cycle, even though the catalyst is not a net reactant.
Common student pitfalls (mechanism interpretation)
Do not include the catalyst in the overall balanced equation (it cancels).
Do not treat catalyst as a reactant that determines the final yield by stoichiometry; it affects rate, not the net reaction change.
“Regenerated” does not require the catalyst to be free at every moment—temporary sequestration is consistent with a constant net amount over complete cycles.
FAQ
“Net constant” refers to the catalyst’s amount after complete cycles, not its instantaneous free concentration.
Some catalyst can be temporarily stored as catalyst-containing species, then released back as the original catalyst.
The reaction continues at an enhanced rate without needing stoichiometric amounts of catalyst.
Also, the catalyst can often be recovered at the end in nearly the same amount (if not deactivated or lost).
Not if it is truly catalytic in the proposed mechanism.
If it appears in the overall equation, it is a reactant or product overall (or the mechanism is incomplete/incorrect regarding catalyst regeneration).
It can indicate catalyst deactivation or loss, such as:
poisoning by impurities
irreversible side reactions involving the catalyst
physical loss (e.g., adsorption to vessel walls)
These prevent full regeneration of active catalyst.
No. Regenerated means it returns to its original chemical form after later steps.
It may be chemically transformed temporarily within the mechanism, provided the cycle returns it at the end.
Practice Questions
Question 1 (2 marks) A proposed catalytic mechanism is: Step 1: Step 2: Identify the catalyst and state why it is a catalyst.
Catalyst is . (1)
It is consumed in one step and regenerated in a later step, so its net amount is unchanged / it cancels from the overall reaction. (1)
Question 2 (5 marks) A catalytic mechanism is proposed: Step 1 (slow): Step 2 (fast): (a) Show the overall reaction. (2 marks) (b) Explain, using the mechanism, what is meant by “the catalyst’s net concentration remains constant,” even though it may be consumed in the rate-determining step. (3 marks)
Add steps and cancel . (1)
Overall: and does not appear. (1) (b)
In the slow step, free is consumed to form . (1)
In the later step, is regenerated as a product. (1)
Therefore over a complete cycle the net change in amount of is zero / it is not used up overall. (1)

