AP Syllabus focus: ‘Assign oxidation numbers in reactants and products to identify which species are oxidized and reduced in a redox reaction.’
Redox analysis becomes systematic when you track oxidation numbers (oxidation states) across reactants and products. This page focuses on assigning oxidation numbers accurately and using changes to identify which species are oxidized and reduced.
Core idea: oxidation numbers reveal electron transfer
Oxidation number (oxidation state): A bookkeeping charge assigned to an atom in a substance, assuming bonding electrons are assigned to the more electronegative atom; used to track redox changes.
Oxidation numbers are not real charges in most covalent compounds, but they reliably indicate where electrons are effectively lost or gained when comparing reactants to products.
Rules for assigning oxidation numbers (AP essentials)
Stepwise assignment rules
Use these rules in order; once an atom’s oxidation number is fixed by a rule, treat it as known.
Free elements: An atom in its elemental form has oxidation number 0 (e.g., Na(s), O(g), S(s)).
Monatomic ions: Oxidation number equals the ion charge (e.g., Fe is +3).
Group 1 metals: Always +1 in compounds (Li, Na, K, etc.).
Group 2 metals: Always +2 in compounds (Mg, Ca, etc.).
Fluorine: Always −1 in compounds.
Hydrogen: Usually +1 with nonmetals; −1 in metal hydrides (e.g., NaH).
Oxygen: Usually −2; important exceptions include:
Peroxides (O–O single bond): oxygen is −1 (e.g., HO)
Superoxides: oxygen is −1/2 on average (e.g., KO)
With fluorine (e.g., OF): oxygen is positive (since F is −1)
Halogens (Cl, Br, I): Usually −1, except when combined with oxygen or a more electronegative halogen.
The accounting constraint (always true)
= total of each atom’s oxidation number multiplied by its subscript (unitless)
= net charge on the species (0 for a neutral compound; ionic charge for a polyatomic ion)
Apply this constraint after assigning the “fixed” elements (like O, H, alkali metals) to solve for unknown oxidation numbers.
Using oxidation numbers to identify what is oxidized and reduced
What counts as oxidation vs reduction (oxidation-number test)
Oxidation / Reduction (via oxidation numbers): Oxidation is an increase in oxidation number; reduction is a decrease in oxidation number.
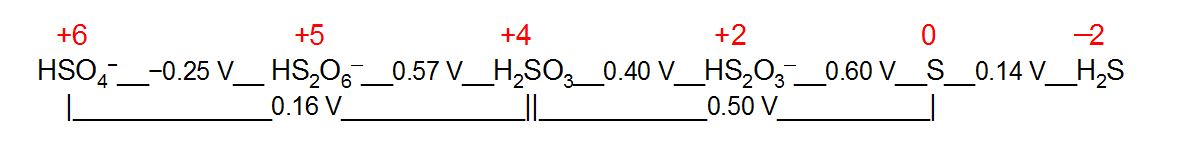
Electrochemical oxidation-state diagram showing oxidation numbers laid out on a single scale so that changes in oxidation state can be interpreted directionally. Movement toward more positive oxidation numbers corresponds to oxidation (increase in oxidation number), while movement toward more negative oxidation numbers corresponds to reduction (decrease in oxidation number). Source
In a redox reaction:
The element whose oxidation number increases is the one that is oxidized (it has effectively lost electron density).
The element whose oxidation number decreases is the one that is reduced (it has effectively gained electron density).
A practical checklist for “species changed”
To answer “which species are oxidized and reduced,” do the following:
Write the reactants and products clearly (including charges for ions).
Assign oxidation numbers to every element in each reactant species.
Assign oxidation numbers to every element in each product species.
Compare the oxidation number of each element from reactant side to product side.
Identify:
Oxidized element/species: contains an element whose oxidation number increases
Reduced element/species: contains an element whose oxidation number decreases
Be precise about wording:
The element undergoes oxidation or reduction (based on its oxidation number change).
The species (molecule/ion) that contains that element is described as being oxidized or reduced.
Common interpretation pitfalls (avoid losing points)
Do not confuse a coefficient change with an oxidation-number change; redox identification depends on oxidation numbers, not amounts.
If an element appears in multiple species, compare the correct before/after pairing based on the balanced equation.
If oxidation numbers do not change for any element, the reaction is not redox (even if it involves ions or precipitation).
FAQ
Fractional oxidation numbers arise when the oxidation state is an average over equivalent atoms (often due to delocalised bonding), such as O in $\text{O}_2^-$ being −1/2 on average.
They are acceptable for bookkeeping, but you should still use the stated exception rules (peroxide vs superoxide) before applying the sum constraint.
Treat common ligands as having typical charges (e.g., CN$^-$ is −1), then use the overall ion charge to solve for the metal.
Be careful to distinguish ligand formal charge from oxidation number assignment on the central atom.
Assign oxidation numbers to elements with fixed rules (e.g., O usually −2, H usually +1), then apply $\sum(\text{ox. no.})=\text{charge}$.
Solve algebraically for the unknown, remembering to multiply by subscripts.
Primarily when bonded to fluorine, because fluorine is always −1. For example, in OF$_2$, oxygen must be +2 to make the molecule neutral.
Look for formulas containing both O and F.
Use bond-by-bond assignment to carbon: assign bonding electrons to the more electronegative atom (O > C > H). Each C–H bond contributes −1 to C; each C–O bond contributes +1 to C (double bonds count twice).
This gives a consistent carbon oxidation number without relying on memorised patterns.
Practice Questions
(2 marks) In the reaction , assign oxidation numbers to Mg and O in reactants and products, and identify which element is oxidised and which is reduced.
1 mark: Correct oxidation numbers: Mg is in Mg(s) and in MgO; O is in O and in MgO.
1 mark: Correct identification: Mg is oxidised (0 to +2); O is reduced (0 to −2).
(5 marks) For the reaction : (a) Assign oxidation numbers to Cl in each chlorine-containing species.
(b) Identify which species is oxidised and which is reduced.
(c) State whether this is disproportionation, with a reason.
(a) 2 marks: Cl in is ; Cl in is ; Cl in is (all correct).
(b) 2 marks: Reduced species: (0 to −1). Oxidised species: (0 to +1).
(c) 1 mark: Yes, disproportionation because chlorine is both oxidised and reduced from oxidation number 0 to two different oxidation numbers.

