AP Syllabus focus: ‘In redox, electrons move from the species that is oxidized to the species that is reduced.’
Redox reactions are best understood by tracking electrons. This page clarifies what oxidation and reduction mean, how electrons flow between reacting species, and how to represent that flow clearly in chemical equations.
Oxidation vs. reduction: the electron perspective
Redox (oxidation–reduction) chemistry centers on electron transfer between chemical species. Even when electrons are not explicitly written in the overall equation, a redox process still involves one species losing electrons and another gaining them.
Oxidation: Loss of one or more electrons by a species.
Reduction: Gain of one or more electrons by a species.
These processes are always coupled: if electrons are lost somewhere, they must be gained elsewhere, because electrons are conserved.
Key relationship: where the electrons go
The syllabus statement can be applied as a strict directional rule for any redox process:
Electrons move from the species that is oxidized to the species that is reduced.
The oxidized species is the electron donor.
The reduced species is the electron acceptor.
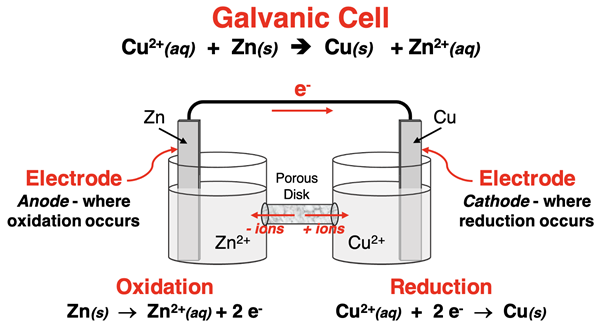
Schematic of a Zn/Cu galvanic cell showing oxidation at the anode and reduction at the cathode, with electron flow () through the external circuit from donor to acceptor. The diagram pairs the overall reaction with the corresponding half-reactions to emphasize that oxidation produces electrons while reduction consumes them. Source
Representing electron flow with half-reaction style equations
A useful way to make electron flow visible is to write oxidation and reduction in a form that shows electrons explicitly. This is not a different reaction; it is an accounting tool that clarifies electron movement.
= number of electrons lost (mol )
= number of electrons gained (mol )
In this notation, electrons appear as products for oxidation (they are released) and as reactants for reduction (they are consumed). For a complete redox process, the electrons released and consumed must match in amount.
How to identify which species is oxidized or reduced
Focus on electron bookkeeping rather than memorising reaction types. Practical cues include:
A species that forms a cation (or increases its positive charge) is typically being oxidized because it has effectively lost electron density.
A species that forms an anion (or increases its negative charge) is typically being reduced because it has effectively gained electron density.
If a neutral element becomes part of an ion, the direction is often clear:
implies oxidation (electrons lost).
implies reduction (electrons gained).
Electron flow is not “atom flow”
Electrons transfer between species, but atoms usually do not jump between particles in the same way. Many redox reactions involve:
Bond changes that redistribute electrons (electron density shifts)
Ion formation in solution
Electron transfer at a surface (common in metal–ion reactions)
Interpreting electron flow in equations and particle terms
When you see a redox reaction written as an overall balanced equation, electron flow is implied:
The oxidized species is the source of the electrons (electron donor).
The reduced species is the destination for the electrons (electron acceptor).
To keep the interpretation consistent:
Treat electrons as moving from donor to acceptor, not both ways.
Avoid the common confusion between charge movement and electron flow: in an equation, charge balance reflects electron transfer, but the electrons themselves may not be written.
Common student pitfalls
Mixing up the terms: oxidation is loss and reduction is gain of electrons.
Assuming oxygen must be involved: oxidation can occur without oxygen present.
Forgetting the directionality: electrons always go from oxidized to reduced species, never the reverse.
FAQ
No. Conventional current is defined as the direction positive charge would move.
Electron flow is the movement of $e^-$, so it is opposite to conventional current direction in many contexts.
Overall equations often omit $e^-$ because they cancel when oxidation and reduction are combined.
You can still infer transfer by identifying which species loses electron density (donor) and which gains it (acceptor).
Yes, in disproportionation reactions.
One reactant is simultaneously an electron donor and acceptor, forming two different products where it ends up at higher and lower electron “ownership” than before.
Electrons cannot be created or destroyed in chemical reactions.
If one species releases $e^-$, another species must take them up, so oxidation and reduction are inseparable parts of one redox process.
It typically means electron density is transferred through collisions, bonding changes, or contact with a conducting surface.
The species ends up with lower charge (or more negative charge) consistent with added $e^-$.
Practice Questions
Q1 (2 marks) In the reaction , identify which species is oxidised and which is reduced.
1 mark: is oxidised (loses electrons).
1 mark: is reduced (gains electrons).
Q2 (5 marks) A student claims that in a redox reaction, “electrons move from the reduced species to the oxidised species.” Using the ideas of oxidation and reduction, assess this claim and state the correct direction of electron flow. Your answer should refer to electron loss/gain and donor/acceptor language.
1 mark: Oxidation is loss of electrons.
1 mark: Reduction is gain of electrons.
1 mark: The oxidised species is the electron donor.
1 mark: The reduced species is the electron acceptor.
1 mark: Therefore electrons move from the oxidised species to the reduced species (claim is incorrect).

