AP Syllabus focus: ‘Explain separation results by linking macroscopic observations to differential intermolecular interactions among solution components and with surfaces or phases.’
Separating components of a mixture is often possible because different particles experience different attractions to solvents, surfaces, or alternative phases. Those interaction differences produce observable outcomes like layering, selective dissolution, or different evaporation behavior.
Core principle: separation follows the strongest attractions
A separation “works” when one component experiences more favorable intermolecular interactions in one location (a phase or a surface) than another component does. The key is comparing:
Solute–solvent attractions (how well each solute is stabilized in a given solvent)
Solute–solute attractions (whether particles prefer to stay together)
Solute–surface attractions (whether particles “stick” to a solid surface)
Solvent–solvent attractions (how readily a solvent can carry particles away or form layers)
What “differential interactions” means
When two components experience unequal attraction strengths to the available phases/surfaces, they distribute unevenly, enabling separation.
Partitioning: the distribution of a solute between two immiscible phases based on which phase provides more favorable intermolecular attractions for that solute.
Linking particle interactions to macroscopic observations
AP explanations should explicitly connect the type/strength of attractions to what you see.
Observation: two liquid layers form and a solute concentrates in one layer
This indicates the solute has stronger attractions to that phase:
A polar/ionic solute is more stabilized by a polar phase via dipole–dipole, hydrogen bonding, or ion–dipole attractions.
A nonpolar solute is more stabilized in a nonpolar phase where London dispersion forces dominate and there is little energetic penalty for disrupting the solvent.
Macroscopic link: the solute appears mostly in the layer where its stabilizing interactions are strongest (greater “like-with-like” attraction).
Observation: one component remains on a solid while another is rinsed away
This indicates stronger solute–surface attractions for the retained component:
If the surface has polar sites, a polar solute may be retained by dipole interactions or hydrogen bonding with the surface.
If the surface is relatively nonpolar, retention may be driven more by dispersion and shape/area of contact.
Macroscopic link: the more strongly attracted component moves more slowly or remains attached; the weaker-interacting component is carried away.
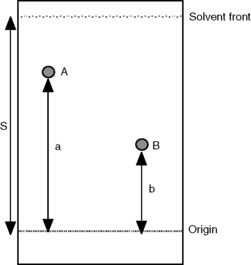
Schematic thin-layer chromatography (TLC) plate showing the origin line, solvent front, and two analyte spots that traveled different distances. A component that interacts more strongly with the solid stationary phase (e.g., polar silica) spends less time in the mobile phase and therefore migrates a shorter distance, producing separation from a less strongly retained component. Source
Observation: separation by vaporization (one component leaves first)
When separating by evaporation/boiling, the component that enters the gas phase more readily is the one with weaker effective attractions in the liquid:
Stronger intermolecular forces (especially extensive hydrogen bonding or strong dipoles) reduce volatility.
Weaker forces increase volatility, so that component contributes more to the vapor.
Macroscopic link: the collected vapor (or remaining liquid) becomes enriched in the component whose attractions best match the phase it occupies.
How to write high-scoring separation explanations
Name the relevant particles (each component, each phase, any surface).
State which interactions are strongest for each component (e.g., ion–dipole, hydrogen bonding, dipole–dipole, LDF).
Assert the direction of preference (which phase/surface better stabilizes which component).
Tie preference to the observation (layering, retention, or differing rates of vaporization).
FAQ
Higher temperature weakens the impact of attractions relative to thermal motion.
This can reduce differences in phase preference or surface retention, making separation less selective.
Mixing increases contact area between phases.
That speeds up partitioning because more solute molecules can cross the interface per unit time before the layers re-form.
Polar functional groups and available sites for hydrogen bonding increase retention.
Greater surface area also increases total interaction strength by providing more contact points.
Cations and anions can associate into less effectively charged pairs.
That reduces ion–dipole stabilisation by the solvent and can shift where the species prefers to reside (phase or surface).
Salt ions strongly attract water molecules, reducing water’s ability to stabilise some neutral solutes.
This can “salt out” the solute, increasing its preference for a less polar phase.
Practice Questions
(2 marks) A mixture contains solute P (polar) and solute N (non-polar). The mixture is shaken with water and a non-polar solvent, forming two layers. Predict which solute is mainly found in each layer and justify using intermolecular forces.
P mainly in water; N mainly in non-polar solvent (1)
Justification linking P to dipole–dipole/hydrogen bonding (or ion–dipole if stated ionic) with water and N to London forces with the non-polar solvent (1)
(5 marks) A solution contains two neutral liquids, X and Y. When gently heated, the vapour collected early in the process is richer in X than in Y. Explain this separation result by linking macroscopic observations to differences in intermolecular interactions and phase preference.
Early vapour enriched in the more volatile component X (1)
Volatility linked to weaker intermolecular attractions in the liquid for X than for Y (1)
Identify plausible interactions (e.g. Y has stronger dipole–dipole or hydrogen bonding; X dominated by LDF) (1)
Explain that stronger attractions lower tendency to enter gas phase (lower vapour pressure) for Y (1)
Connect to observation: X enters gas phase more readily so collected vapour is richer in X (1)

