AP Syllabus focus: ‘Components of a liquid solution cannot be separated by filtration; alternative methods rely on differences in intermolecular interactions between components.’
Filtration is a powerful separation technique for heterogeneous mixtures, but it fails for true liquid solutions because dissolved particles are molecular-scale and move with the solvent through any ordinary filter medium.
What filtration can and cannot separate
Key idea: particle size vs pore size
Filtration works when one component exists as insoluble solid particles suspended in a fluid (a heterogeneous mixture). A filter traps particles that are larger than the filter pores.
In a liquid solution, the solute is dispersed as individual ions or molecules throughout the solvent, so there are no solid particles to mechanically trap.
Filtration: A physical separation method that uses a porous barrier to retain solid particles while allowing a fluid (and any dissolved species) to pass through.
A filter paper, cloth, or sand bed is designed to stop particles such as precipitates, not dissolved species.
What “dissolved” means at the particle level
In a solution, solute particles are surrounded by solvent particles and are carried along with the flowing liquid.
Solution: A homogeneous mixture in which solute particles are uniformly dispersed among solvent particles at the molecular/ionic scale.
Because the solution is homogeneous, the composition of the liquid entering the filter and leaving the filter is the same (ignoring minor losses like liquid hold-up in the filter).
Why a filter does not “catch” dissolved solute
Dissolved particles are far too small
Typical filter pores are orders of magnitude larger than dissolved species:
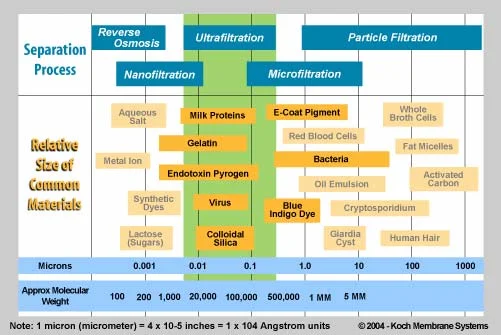
Log-scale chart comparing what different filtration/membrane processes can block based on particle size. It visually contrasts micron-scale particles (e.g., cells, bacteria) with much smaller dissolved species, clarifying why ordinary filtration cannot change the concentration of a true solution. This supports the size-exclusion argument in a quantitative, scale-aware way. Source
Dissolved ions/molecules are roughly nanometre-scale.
Common filter media have pores large enough that solvent flows readily (micrometre-scale or larger).
So both solvent and solute pass through together, and filtration cannot change concentration.
Solute–solvent attractions keep solute in the liquid phase
A dissolved solute is stabilised by intermolecular (or ion–dipole) attractions between solute and solvent. These attractions:
keep solute particles dispersed rather than clumped into filterable particles
allow solute to move wherever the solvent moves
Even if some solute weakly sticks to a filter surface, that is not reliable separation; it is typically a minor, uncontrolled loss.
What must be different for separation to work
Filtration fails because it relies on size exclusion. To separate components of a liquid solution, you must instead exploit differences in how strongly components interact with other phases or with each other.
“Alternative methods rely on differences in intermolecular interactions”
Separation approaches for solutions work by creating a situation where components experience different strengths of attraction and therefore distribute differently, for example:
one component preferentially enters the gas phase while another remains in the liquid (based on how strongly particles attract in the liquid)
one component preferentially associates with a different liquid phase or a surface (again governed by relative attractions)
The essential AP idea is to connect the failure of filtration to the molecular-scale uniformity of solutions and to recognise that successful separation requires differential intermolecular interactions rather than a simple mechanical barrier.
FAQ
Only if the “solution” actually contains larger aggregates (e.g., colloids). True molecular solutions require membranes with extremely small effective pores, not ordinary filtration.
Colloid particles can be smaller than filter pores and remain suspended due to constant motion and weak settling, so they behave unlike easily trapped precipitates.
Sometimes a small amount can adsorb, especially with charged or porous materials, but it is typically incomplete and non-quantitative, so it is not a dependable separation method.
Temperature can change how strongly particles attract and how readily a component enters another phase (e.g., more particles escaping to the gas phase), enabling separation routes based on those interaction changes.
Before precipitation, ions are dispersed and pass through; after precipitation, ions are in large solid particles (an insoluble phase) that are bigger than the pores and can be retained.
Practice Questions
Explain why a saltwater solution cannot be separated into salt and water by filtration. (2 marks)
States that dissolved ions are too small to be trapped by filter pores / are not solid particles (1)
States that both solute and solvent pass through so composition is unchanged (1)
A student tries to separate a blue aqueous dye solution using filter paper but observes no change in colour intensity of the filtrate. Using particle-level reasoning, explain why filtration fails and state what general type of property must differ for an alternative separation to succeed. (5 marks)
Identifies the mixture as a homogeneous solution with uniformly dispersed dye particles (1)
Explains dye particles are molecular/ionic scale and pass through pores with water (1)
Links dissolution to stabilising attractions between solute and solvent (intermolecular interactions) (1)
States filtration separates based on particle size/insolubility and therefore cannot change concentration for solutions (1)
States alternatives must rely on differences in intermolecular interactions/affinities between components and phases/surfaces (1)

