AP Syllabus focus: ‘Relate the solubility of ionic and molecular compounds in aqueous and nonaqueous solvents to the intermolecular interactions between solute and solvent particles.’
Solubility depends on whether new solute–solvent attractions formed during dissolving are strong enough to compete with the attractions already present in the pure solute and pure solvent. Comparing aqueous and nonaqueous solvents highlights how interaction type and strength control dissolving.
Core idea: what “dissolving” requires
To dissolve, particles must separate and then become stabilized by the solvent.
Separate solute particles: overcome ion–ion attractions (ionic solids) or intermolecular forces (molecular solids/liquids)
Separate solvent particles: create space in the solvent
Form solute–solvent interactions: release energy and stabilise dispersed particles
Key term: solvation
Solvation: The process in which solvent particles surround and stabilise solute particles through intermolecular attractions; in water, solvation is called hydration.
Solubility is higher when solvation interactions are strong and numerous relative to the interactions that must be disrupted.
Solubility of ionic compounds in different solvents
Ionic compounds contain cations and anions held by strong Coulombic attractions in a lattice. Whether they dissolve depends strongly on solvent polarity.
Why many ionic compounds dissolve in water (aqueous solvents)
Water is highly polar and can orient to stabilise ions via ion–dipole interactions:
Oxygen end () points toward cations
Hydrogen ends () point toward anions
Multiple water molecules form a hydration shell around each ion
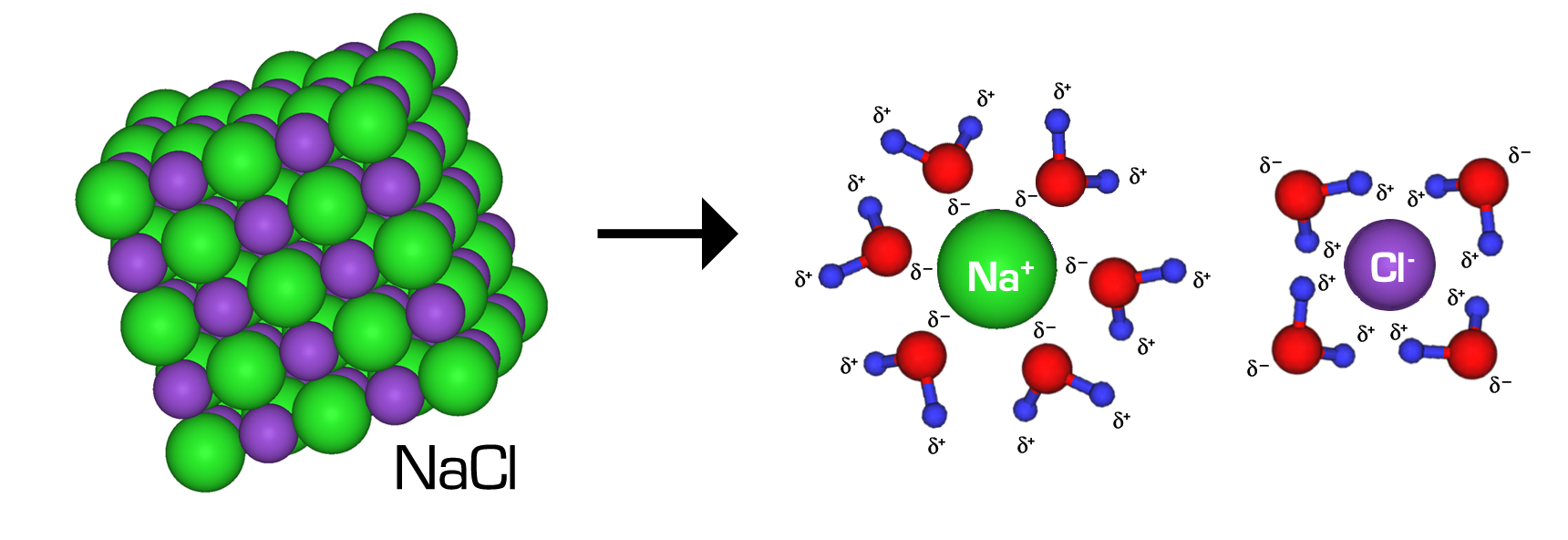
Water molecules orient their partial charges toward ions, creating ion–dipole attractions that stabilize separated and . The clustered water molecules around each ion represent hydration shells, which help keep ions dispersed in solution rather than re-forming the ionic lattice. Source
Strong hydration can compensate for breaking the ionic lattice and disrupting some water–water interactions (including hydrogen bonding). As a result, many salts are soluble in water.
Why many ionic compounds do not dissolve in nonpolar solvents
Nonpolar solvents (e.g., hexane) provide mainly London dispersion forces and cannot stabilise separated ions effectively.
Ion–dipole attractions are absent or very weak
The energy cost to separate ions is not repaid by strong solvation Therefore, many ionic solids are essentially insoluble in nonpolar liquids.
Ionic charge and size: “harder to pull apart” vs “easier to stabilise”
Two competing ideas help you reason qualitatively:
Higher ionic charge and smaller ionic radius generally increase ion–ion attraction in the solid (harder to separate).
Small, highly charged ions can also interact very strongly with polar solvents (easier to stabilise once separated).
A solvent must provide sufficiently strong stabilising interactions to make dispersed ions favourable overall.
Key term: lattice energy (qualitative use)
Lattice energy: A measure of the strength of attraction holding ions together in an ionic solid; larger magnitude indicates a more strongly bound lattice that is harder to separate.
In solubility arguments, “large lattice energy” points toward lower solubility unless the solvent provides exceptionally strong ion-stabilising interactions.
Solubility of molecular compounds in different solvents
Molecular substances dissolve when the solvent can form comparable intermolecular attractions with the solute.
Polar molecular solutes in water
Polar molecules (including those that can hydrogen bond) often dissolve well in water because they can form strong, specific interactions:
Dipole–dipole attractions between solute and water
Hydrogen bonding when the solute contains appropriate functional groups that can donate or accept hydrogen bonds These interactions can replace some solute–solute and water–water attractions with similarly strong solute–water attractions.
Nonpolar molecular solutes in nonpolar solvents
Nonpolar molecules tend to be soluble in nonpolar solvents because both rely mainly on London dispersion forces.
The solvent can surround the solute with many dispersion contacts
Little is lost by disrupting solvent structure because the pure solvent is also held together mostly by dispersion forces
Why many nonpolar molecules are poorly soluble in water

The two distinct liquid layers show that polar water and a nonpolar oil do not form a single homogeneous solution. This macroscopic observation reflects the underlying mismatch in intermolecular attractions: water maintains strong polar interactions, while the nonpolar liquid cannot replace them effectively. Source
Even if a nonpolar solute can experience dispersion forces with water, water’s strong network of polar interactions makes it costly to create cavities and reorganise water molecules. If the solute cannot replace those interactions with comparably strong attractions, solubility remains low.
Nonaqueous solvents: what to look for
When the solvent is not water, focus on what interactions it can offer.
Polar protic solvents (able to hydrogen bond as donors) can stabilise ions and polar solutes through strong dipole interactions and hydrogen bonding.
Polar aprotic solvents (polar but not hydrogen-bond donors) can strongly stabilise many cations via dipole interactions; solubility outcomes depend on how well both ions (or the overall solute) are stabilised.
Low-polarity solvents mainly provide dispersion forces, favouring nonpolar solutes.
Solvent polarity as a practical descriptor
Polarity (solvent): The extent to which a solvent has a permanent dipole that can stabilise charged or polar solutes through electrostatic attractions.
A more polar solvent generally better stabilises ions and polar molecules, increasing the likelihood of solubility.
How to justify a solubility claim (AP-style reasoning)
When comparing “soluble vs insoluble” across solvents, explicitly connect to particle-level attractions:
Identify dominant attractions in the solute (ion–ion, hydrogen bonding, dipole–dipole, dispersion)
Identify attractions the solvent can provide
Argue whether solute–solvent attractions can effectively replace solute–solute and solvent–solvent attractions
For ionic solutes, emphasise ion–dipole stabilisation in polar solvents vs weak stabilisation in nonpolar solvents
FAQ
Some “less polar” solvents can still stabilise ions if they have localised electron density that interacts strongly with ions.
Factors that can help:
Highly polarisable solvent molecules that interact strongly with ions
Specific coordination to cations (solvent acting like a ligand)
The salt having large, diffuse ions that are easier to separate and stabilise
Polar aprotic solvents often stabilise cations strongly via dipole interactions but cannot donate hydrogen bonds to stabilise anions as effectively.
This imbalance can:
Increase separation of ion pairs in some cases
Reduce solubility for salts whose anions need strong hydrogen-bond donation to be well solvated
A solute can hydrogen bond if it has:
A hydrogen-bond donor: typically $X-H$ where $X$ is electronegative (commonly $O-H$ or $N-H$)
A hydrogen-bond acceptor: a lone pair on an electronegative atom (commonly O, N, sometimes F)
Both donor/acceptor matching with the solvent increases solubility in hydrogen-bonding solvents.
Even in nonpolar systems, dispersion forces vary with:
Polarisability (more electrons, softer electron cloud)
Molecular shape and surface area (better contact increases attractions)
Ability to pack closely with the solvent molecules
These factors change how favourable solute–solvent dispersion contacts are.
For most solids and liquids dissolving in liquids, pressure has only a small effect because the solution’s volume changes little on mixing.
Pressure effects become more important when:
The solute is a gas
The solvent is near a phase change where compressibility is higher
Practice Questions
Q1 (2 marks) A student claims that potassium chloride dissolves readily in water but not in hexane. Explain this difference in solubility in terms of intermolecular interactions.
1 mark: Water is polar and forms strong ion–dipole attractions (hydration) with and .
1 mark: Hexane is nonpolar and can only offer weak London dispersion forces, so it cannot stabilise separated ions.
Q2 (5 marks) Two molecular solutes, A and B, are tested for solubility in water and in a nonpolar solvent. Solute A contains an group; solute B is a nonpolar hydrocarbon of similar size. Using intermolecular interactions, predict which solute is more soluble in (i) water and (ii) the nonpolar solvent, and justify both predictions.
1 mark: A is more soluble in water (prediction).
1 mark: Justification: A can hydrogen bond and/or engage in strong dipole–dipole interactions with water.
1 mark: B is less soluble in water (prediction).
1 mark: Justification: B is nonpolar; interactions with water are mainly weak dispersion and do not replace strong water–water attractions effectively.
1 mark: B is more soluble in the nonpolar solvent (prediction + justification): both are nonpolar and interact primarily via London dispersion forces (“like interacts with like”).

